- Ethyl Acetate
-

- $8.00 / 1KG
-
2024-09-19
- CAS:141-78-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10 ton
- Ethyl acetate
-

- $0.00 / 200kg
-
2024-09-13
- CAS:141-78-6
- Min. Order: 20kg
- Purity: 99.0%
- Supply Ability: 20 tons
- Ethyl acetate
-
- $1055.00 / 1ton
-
2024-08-29
- CAS:141-78-6
- Min. Order: 1ton
- Purity: 99%
- Supply Ability: 5000ton
|
| Product Name: | Ethyl acetate | | Synonyms: | GLASS TUBE + SCREW CAP 16 X 115 MM;KARL FISCHER LIPO SOLVENT HM NON;DIMETHYL SULPHOXIDE DESHYDRATED WITH LES;POTASSIUM IODATE SOLUTION 8.8GL;SODIUM ACETATE 2M PH 4.2;Ethyl acetate puriss. p.a., ACS reagent, reag. ISO, reag. Ph. Eur., >=99.5% (GC);Ethyl acetate4x1L;Ethyl acetate, ACS, 99.5% min. | | CAS: | 141-78-6 | | MF: | C4H8O2 | | MW: | 88.11 | | EINECS: | 205-500-4 | | Product Categories: | Solvent;Residue Analysis (Japan only);Solvent by Application;Solvents by Special Grades (Japan Customers Only);Aluminum Bottles;Solvent Bottles;Solvent Packaging Options;Amber Glass Bottles;Analytical Reagents;Analytical/Chromatography;CHROMASOLV Plus;Chromatography Reagents &;HPLC &;Ethyl Acetate;Carthamus tinctorius (Safflower oil);Ephedra sinica;Nutrition Research;Panax ginseng;Phytochemicals by Plant (Food/Spice/Herb);Solvent by Type;Zingiber officinale (Ginger);Pharmacopoeia;Pharmacopoeia A-Z;Alphabetical Listings;Certified Food Grade Products;Certified Natural Products;Flavors and Fragrances;Kosher Certified Products;E-F;E-H;LEDA HPLC;Solvents for HPLC & Spectrophotometry;Solvents for Spectrophotometry;Aluminum Bottles;ReagentPlus(R)Semi-Bulk Solvents;Ethyl AcetateSolvent Bottles;Spectrophotometric Grade Solvents;Spectrophotometric GradeSolvents;Protein Sequencing;Protein Structural Analysis;Reagents for Protein Sequencing;Chemical Class;EQ - EZAnalytical Standards;EstersAnalytical Standards;Ethyl AcetateSolvents;Biotech SolventsSolvents;CHROMASOLV Solvents (HPLC, LC-MS);CHROMASOLV(R) HPLC Grade SolventsSolvents;Solvents;CHROMASOLV for HPLCSemi-Bulk Solvents;CHROMASOLV(R) for HPLCSolvents;Composite Drums;Drums Product Line;NOWPak(R) Products;ACS Grade SolventsSolvents;ACS GradeSolvents;Analytical Reagents for General Use;E-L, Puriss p.a. ACS;Puriss p.a. ACS;ACS GradeDrums Product Line;Closed Head Drums;Ethyl AcetateSaturated fatty acids and derivatives;Ethyl EsterMore...Close...;ACS GradeSemi-Bulk Solvents;Carbon Steel Flex-Spout Cans;Esters;Capillary GC SolventsSolvent Bottles;GC Capillary;CHROMASOLV(R) LC-MSSolvents;Ethyl AcetateSpectroscopy;CHROMASOLV PlusCHROMASOLV Solvents (HPLC, LC-MS);CHROMASOLV(R) PlusSolvents;Amber Glass Bottles;Pesticide Residue Analysis (PRA) SolventsSolvent Bottles;PRA;ACS Grade Solvents;E-H, Puriss p.a.Solvents;Ethyl AcetateChromatography/CE Reagents;Pestanal/Residue Analysis;Puriss p.a.;Solvents - GC/SH;Anhydrous Grade SolventsSolvents;AnhydrousSolvents;Returnable Container Solvents;Ethyl Acetate;Certified Natural ProductsFlavors and Fragrances;E-FSolvents;Ethyl AcetateFlavors and Fragrances;Prepackaged SamplesFlavors and Fragrances;Redi-Pack Bulk;Alphabetical Listings;Flavors and Fragrances;Hematology and Histology;Routine Histology Stains;PVC Coated Bottles;ReagentPlus(R) Solvent Grade ProductsSolvents;ReagentPlus(R)Solvents;Solvent Bottles;Sure/Seal? Bottles;Alpha Sort;E;E-LAlphabetic;EQ - EZ;Volatiles/ Semivolatiles;HPLC Plus Grade Solvents (CHROMASOLV);HPLC/UHPLC Solvents (CHROMASOLV);UHPLC Solvents (CHROMASOLV);ACS and Reagent Grade Solvents;Carbon Steel Flex-Spout Cans;ReagentPlus;ReagentPlus Solvent Grade Products;Semi-Bulk Solvents;Analytical Reagents for General Use;Puriss p.a.;PVC Coated Bottles;Sure/Seal Bottles;Anhydrous;Anhydrous Solvents;Products;Returnable Containers;GC Solvents;Pesticide Residue Analysis (PRA) Solvents;Solvents for GC applications;Solvents for Organic Residue Analysis;Trace Analysis Reagents &;Biotech Solvents;CHROMASOLV for HPLC;Composite Drums;Drums Product Line;HPLC Grade Solvents (CHROMASOLV);NOWPak Products;ACS Grade;ACS Grade Solvents;Organics;Alcohol;Intermediates;Analytical Chemistry;LC-MS SolventsProteomics;Solvents and Solutions for Mass Spectrometry;CHROMASOLV LC-MSCHROMASOLV Solvents (HPLC, LC-MS);LC-MS Plus and Gradient;Mass Spectrometry;Mass Spectrometry (MS)&LC-MS;Chromatography/CE Reagents;141-78-6 | | Mol File: | 141-78-6.mol |  |
| | Ethyl acetate Chemical Properties |
| Melting point | −84 °C(lit.) | | Boiling point | 76.5-77.5 °C(lit.) | | density | 0.902 g/mL at 25 °C(lit.) | | vapor density | 3 (20 °C, vs air) | | vapor pressure | 73 mm Hg ( 20 °C) | | refractive index | n20/D 1.3720(lit.) | | FEMA | 2414 | ETHYL ACETATE | | Fp | 26 °F | | storage temp. | Store at +2°C to +25°C. | | solubility | Miscible with ethanol, acetone, diethyl ether and benzene. | | pka | 16-18(at 25℃) | | form | Liquid | | Specific Gravity | 0.902 (20/20℃) | | color | APHA: ≤10 | | Relative polarity | 0.228 | | Odor | Pleasant fruity odor detectable at 7 to 50 ppm (mean = 18 ppm) | | explosive limit | 2.2-11.5%, 38°F | | Odor Threshold | 0.87ppm | | Odor Type | ethereal | | Water Solubility | 80 g/L (20 ºC) | | λmax | λ: 256 nm Amax: ≤1.00
λ: 275 nm Amax: ≤0.05
λ: 300 nm Amax: ≤0.03
λ: 325-400 nm Amax: ≤0.005 | | JECFA Number | 27 | | Merck | 14,3757 | | BRN | 506104 | | Henry's Law Constant | 0.39 at 5.00 °C, 0.58 at 10.00 °C, 0.85 at 15.00 °C, 1.17 at 20.00 °C, 1.58 at 25.00 °C (column
stripping-UV, Kutsuna et al., 2005) | | Exposure limits | TLV-TWA 400 ppm (~1400 mg/m3)
(ACGIH, MSHA, and OSHA); IDLH 10,000
ppm (NIOSH). | | Dielectric constant | 23.0(Ambient) | | Stability: | Stable. Incompatible with various plastics, strong oxidizing agents. Highly flammable. Vapour/air mixtures explosive. May be moisture sensitive. | | InChIKey | XEKOWRVHYACXOJ-UHFFFAOYSA-N | | LogP | 0.68-0.73 at 20-25℃ | | CAS DataBase Reference | 141-78-6(CAS DataBase Reference) | | NIST Chemistry Reference | Ethyl acetate(141-78-6) | | EPA Substance Registry System | Ethyl acetate (141-78-6) |
| | Ethyl acetate Usage And Synthesis |
| organic ester compound | Ethyl Acetate is an organic ester compound with a molecular formula of C4H8O2 (commonly abbreviated as EtOAc or EA), appears as a colorless liquid. It is highly miscible with all common organic solvents (alcohols, ketones, glycols, esters), which make it a common solvent for cleaning, paint removal and coatings.
Ethyl acetate is found in alcoholic beverages, cereal crops, radishes, fruit juices, beer, wine, spirits etc. It has a fruity characteristic odor that is commonly recognized in glues, nail polish remover, decaffeinating tea and coffee, and cigarettes. Due to its agreeable aroma and low cost, this chemical is commonly used and manufactured in large scale in the world, as over 1 million tons annually.
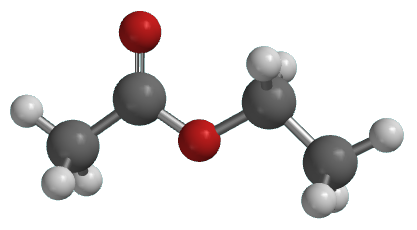
ethyl acetate structure | | Chemical Properties | Ethyl acetate (structure shown above) is the most familiar ester to many chemistry students and possibly the ester with the widest range of uses. Esters are structurally derived from carboxylic acids by replacing the acidic hydrogen by an alkyl or aryl group. Ethyl acetate itself is a colourless liquid at room temperature with a pleasant "fruity" smell, b.p. 77°C.

Ethyl acetate has many uses, such as artificial fruit essences and aroma enhancers, artificial flavours for confectionery, ice cream and cakes, as a solvent in many applications (including decaffeinating tea and coffee) for varnishes and paints (nail varnish remover), and for the manufacture of printing inks and perfumes.
| | Purification and water removal methods | Ethyl acetate generally has a content of 95% to 98% containing a small amount of water, ethanol and acetic acid. It can be further purified as following: add 100mL of acetic anhydride into 1000mL of ethyl acetate; add 10 drops of concentrated sulfuric acid, heat and reflux for 4h to remove impurities such as ethanol and water, and then further subject to distillation. Distillate is oscillated by 20~30g of anhydrous potassium carbonate and further subject to re-distillation. The product has a boiling point of 77 °C and purity being over 99%.
| | Uses |
Industry
Applications
Role/Benefit
Flavor and essence
Food flavor
Used largely to prepare bananas, pears, peaches, pineapple and grape scent food flavors, etc
Alcoholic essence
Used slightly as fragrance volatile
Perfume essence
Used slightly as fragrance volatile
Chemical manufacture
Production of acetamide, acetyl acetate, methyl heptanone, etc
Organic chemical raw materials
Production of organic acid
Extracting agent
Laboratory
Dilution and extraction
Supply excellent dissolving capacity
Chromatographic analysis
Standard material
Column chromatography and extractions
Main component of mobile phase
Reaction solvent
Be prone to hydrolysis and transesterification
Chemical analysis
Thermometer calibration for sugar separation
Standard material
Determination of bismuth, boron, gold, molybdenum, platinum and thallium
Solvent
Entomology
Insect collecting and study
Used as effective asphyxiant to kill the collected insect quickly without destroying it
Textile industry
Cleaning agent
Supply excellent dissolving capacity
Printing
Flexographic and rotogravure printing
Dissolve the resin, control the viscosity and modify the drying rate
Electronics industry
Viscosity reducer
Reduce the viscosity of resins used in photoresist formulations
Paint manufacture
Solvent
Dissolve and dilute the paints
Health & personal care products
The formulation of nail polish, nail polish removers and other manicuring products
Supply excellent dissolving capacity
Pharmaceutical
Medicine manufacturing
Extraction agent; intermediate
Cosmetics
Aroma enhancer
In perfume to enhance aroma
Others
Tanning extracts
Used for desulfurization of tanning, cigarette materials, oil field drilling, metal flotation, descaling, etc
Production of adhesive
Solvent
Extract many compounds (phosphorus, cobalt, tungsten, arsenic) from aqueous solution
Extracting agent
| | Production | Industrial production of ethyl acetate is mainly classified into three processes.
The first one is a classical Fischer esterification process of ethanol with acetic acid in presence of acid catalyst. This process needs acid catalyst2 such as sulphuric acid, hydrochloride acid, ptoluene sulfonic acid etc. This mixture converts to the ester in about 65% yield at room temperature.
CH3CH2OH + CH3COOH ↔ CH3COOC2H5 + H2O
The reaction can be accelerated by acid catalysis and the equilibrium can be shifted to the right by removal of water.
The second one is Tishchenko Reaction of acetaldehyde using aluminium triethoxide as a catalyst. In Germany and Japan, most ethyl acetate is produced via the Tishchenko process.
2 CH3CHO → CH3COOC2H5
This method has been proposed by two different routes; (i) dehydrogenative process, which uses copper or palladium based catalyst and (ii) the oxidative one, which employs, PdO supported catalysts.
The third one, which has been recently commercialized, is addition of acetic acid to ethylene using clay and heteroploy acid7 as a catalyst.
CH2= CH2 + CH3COOH → CH3COOC2H5
The processes, however, have some disadvantages; both the conventional esterification and addition of acetic acid to ethylene need stock tanks and apparatus for several feed stocks. Moreover, they use acetic acid that causes apparatus corrosion. Although Teshchenko Reaction uses only one feed and it is a non-corrosive material, it is difficult to handle acetaldehyde because is not available outside of petrochemical industrial area.
In such circumstances, an improved process of ethyl acetate production is strongly desired.
| | Extinguishing agent | dry powder, dry sand, carbon dioxide, foam, and 1211 fire extinguishing agent
| | Professional standards | TWA 1400 mg/m³; STEL 2000 mg/m³
| | Description | Ethyl acetate (systematically, ethyl ethanoate, commonly abbreviated EtOAc or EA) is the organic compound with the formula CH3COOCH2CH3. This colorless liquid has a characteristic sweet smell (similar to pear drops) and is used in glues, nail polish removers, decaffeinating tea and coffee, and cigarettes (see list of additives in cigarettes). Ethyl acetate is the ester of ethanol and acetic acid; it is manufactured on a large scale for use as a solvent. The combined annual production in 1985 of Japan, North America, and Europe was about 400,000 tons. In 2004, an estimated 1.3M tons were produced worldwide. | | Chemical Properties | Ethyl acetate has a pleasant ethereal fruity, brandy-like odor, reminiscent of pineapple, somewhat nauseating in high
concentration. It has fruity sweet taste when freshly diluted in water. Ethyl acetate is probably one of the most used of all flavor
chemicals by volume. Ethyl acetate is slowly decomposed by moisture and then acquires an acid status due to the acetic acid formed. | | Physical properties | Clear, colorless, mobile liquid with a pleasant, sweet fruity odor. Experimentally determined
detection and recognition odor threshold concentrations were 23 mg/m3 (6.4 ppmv) and 48 mg/m3
(13.3 ppmv), respectively (Hellman and Small, 1974). Cometto-Mu?iz and Cain (1991) reported
an average nasal pungency threshold concentration of 67,300 ppmv. | | Occurrence | Although it has been reported present in some natural fruital aromas and in some distillates (rum, rum ether),
it has not been reported yet as a constituent of essential oils; it has been identified also in the petals of Magnolia fuscata. Reported
found in many foods including fresh and cooked apple, apricot, banana (169 ppm), sweet and sour cherry, citrus peel oils and juices,
blueberry, cranberry, black currants, raspberry, blackberry, guava, passion fruit, melon, peaches, papaya, pineapple, cabbage, onion,
leek, potato, tomato (3 to 6 ppm), clove, ginger, vinegar, breads, cheeses (0.2 to 0.8 ppm), butter (2 ppm), yogurt, milk, meats, cognac,
beer (4 to 64 ppm), whiskies, cider, sherry, grape wines, rum, cocoa, coffee, tea, filberts, peanuts, popcorn, oats, honey, soybeans,
coconut, olive oil (0.02 ppm) and olive. | | Uses | Pharmaceutic aid (flavor); artificial fruit essences; solvent for nitrocellulose, varnishes, lacquers, and aeroplane dopes; manufacture of smokeless powder, artificial leather, photographic films and plates, artificial silk, perfumes; cleaning textiles, etc. | | Uses | Ethyl acetate is used primarily as a solvent and diluent, being favored because of its low cost, low toxicity, and agreeable odor. For example, it is commonly used to clean circuit boards and in some nail varnish removers (acetone and acetonitrile are also used). Coffee beans and tea leaves are decaffeinated with this solvent.It is also used in paints as an activator or hardener.[citation needed] Ethyl acetate is present in confectionery, perfumes, and fruits. In perfumes, it evaporates quickly, leaving only the scent of the perfume on the skin.
3 – 1 - Laboratory uses
In the laboratory, mixtures containing ethyl acetate are commonly used in column chromatography and extractions. Ethyl acetate is rarely selected as a reaction solvent because it is prone to hydrolysis and trans esterification.
3 – 2 - Occurrence in wines
Ethyl acetate is the most common ester in wine, being the product of the most common volatile organic acid — acetic acid, and the ethyl alcohol generated during the fermentation. The aroma of ethyl acetate is most vivid in younger wines and contributes towards the general perception of "fruitiness" in the wine.
3 – 3 - Entomological killing agent
In the field of entomology, ethyl acetate is an effective asphyxiant for use in insect collecting and study. In a killing jar charged with ethyl acetate, the vapors will kill the collected (usually adult) insect quickly without destroying it. Because it is not hygroscopic, ethyl acetate also keeps the insect soft enough to allow proper mounting suitable for a collection. | | Uses | Ethyl acetate is used as a solvent for varnishes, lacquers, and nitrocellulose; as anartificial fruit flavor; in cleaning textiles;and in the manufacture of artificial silk andleather, perfumes, and photographic filmsand plates (Merck 1996). Ethyl Acetate is generally used as a solvent in organic reactions. Environmental contaminants; Food contaminants. | | Preparation | Ethyl acetate is made by esterification of acetic acid with ethanol, from acetaldehyde, or by the direct addition of ethylene to acetic acid. BP started a 220,000 tonne/year plant in 2001 to operate the last of these processes, known as AVADA. Ethylene and acetic acid react in the presence of a heteropolyacid catalyst to give ethyl acetate at a claimed high selectivity and 99.97% purity. This is the world’s largest ethyl acetate plant and is motivated by its increasing use as a more “acceptable” solvent than hydrocarbons.
In some countries, where ethanol is expensive or there is surplus acetaldehyde capacity, ethyl acetate is made by a Tishchenko reaction. Sasol in South Africa was said to be investigating such a process in the early 2000s. Ethanol is a solvent for surface coatings, cleaning preparations, and cosmetics. Industrial ethanol is aerobically fermented to white vinegar (dilute acetic acid) of the type used for pickling. Gourmet vinegars—wine vinegar, cider vinegar, and so on, made by fermentation of alcoholic beverages—are also available. Ten percent of industrial ethanol production was used for vinegar in the United States in 2001. | | Production Methods | Ethyl acetate is synthesized in industry mainly via the classic Fischer esterification reaction of ethanol and acetic acid. This mixture converts to the ester in about 65% yield at room temperature:
CH3CH2OH + CH3COOH ? CH3COOCH2CH3 + H2O
The reaction can be accelerated by acid catalysis and the equilibrium can be shifted to the right by removal of water. It is also prepared in industry using the Tishchenko reaction, by combining two equivalents of acetaldehyde in the presence of an alkoxide catalyst:
2 CH3CHO → CH3COOCH2CH3. | | Production Methods | Ethyl acetate can be manufactured by the slow distillation of a
mixture of ethanol and acetic acid in the presence of concentrated
sulfuric acid. It has also been prepared from ethylene using an
aluminum alkoxide catalyst. | | Definition | ChEBI: Ethyl acetate is the acetate ester formed between acetic acid and ethanol. It has a role as a polar aprotic solvent, an EC 3.4.19.3 (pyroglutamyl-peptidase I) inhibitor, a metabolite and a Saccharomyces cerevisiae metabolite. It is an acetate ester, an ethyl ester and a volatile organic compound. | | Reactions | Ethyl acetate can be hydrolyzed in acidic or basic conditions to regain acetic acid and ethanol. The use of an acid catalyst accelerates the hydrolysis, which is subject to the Fischer equilibrium mentioned above. In the laboratory, and usually for illustrative purposes only, ethyl esters are typically hydrolyzed in a two step process starting with a stoichiometric amount of strong base, such as sodium hydroxide. This reaction gives ethanol and sodium acetate, which is unreactive toward ethanol:
CH3CO2C2H5 + Na OH → C2H5OH + CH3CO2Na
The rate constant is 0.111 dm3 / mol.sec at 25 °C. | | Aroma threshold values | Detection: 5 ppb to 5 ppm | | General Description | Ethyl acetate, a carboxylate ester, is bio-friendly organic solvent with wide range of industrial applications. Its synthesis by reactive distillation and by acceptorless dehydrogenative dimerization of ethanol has been explored. Its utility as a less toxic alternative to diethyl ether in the formalin-ether (F-E) sedimentation procedure for intestinal parasites has been investigated. Its ability as an acyl acceptor in the immobilized lipase-mediated preparation of biodiesel from crude vegetable oils has been examined. The complete degradation of ethyl acetate to CO2 using manganese octahedral molecular sieve (OMS-2) has been investigated. | | Air & Water Reactions | Highly flammable. Slightly soluble in water. Ethyl acetate is slowly hydrolyzed by moisture. | | Reactivity Profile | Ethyl acetate is also sensitive to heat. On prolonged storage, materials containing similar functional groups have formed explosive peroxides. Ethyl acetate may ignite or explode with lithium aluminum hydride. Ethyl acetate may also ignite with potassium tert-butoxide. Ethyl acetate is incompatible with nitrates, strong alkalis and strong acids. Ethyl acetate will attack some forms of plastics, rubber and coatings. Ethyl acetate is incompatible with oxidizers such as hydrogen peroxide, nitric acid, perchloric acid and chromium trioxide. Violent reactions occur with chlorosulfonic acid. . SOCl2 reacts with esters, such as Ethyl acetate, forming toxic SO2 gas and water soluble/toxic acyl chlorides, catalyzed by Fe or Zn (Spagnuolo, C.J. et al. 1992. Chemical and Engineering News 70(22):2.). | | Health Hazard | The acute toxicity of ethyl acetate is low. Ethyl acetate vapor causes eye, skin, and
respiratory tract irritation at concentrations above 400 ppm. Exposure to high
concentrations may lead to headache, nausea, blurred vision, central nervous system
depression, dizziness, drowsiness, and fatigue. Ingestion of ethyl acetate may cause
gastrointestinal irritation and, with larger amounts, central nervous system
depression. Eye contact with the liquid can produce temporary irritation and
lacrimation. Skin contact produces irritation. Ethyl acetate is regarded as a substance
with good warning properties.
No chronic systemic effects have been reported in humans, and ethyl acetate has not
been shown to be a human carcinogen, reproductive, or developmental toxin | | Flammability and Explosibility | Ethyl acetate is a flammable liquid (NFPA rating = 3), and its vapor can travel a considerable distance to an ignition source and "flash back." Ethyl acetate vapor forms explosive mixtures with air at concentrations of 2 to 11.5% (by volume). Hazardous gases produced in ethyl acetate fires include carbon monoxide and carbon dioxide. Carbon dioxide or dry chemical extinguishers should be used for ethyl acetate fires. | | Chemical Reactivity | Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent. | | Pharmaceutical Applications | In pharmaceutical preparations, ethyl acetate is primarily used as a
solvent, although it has also been used as a flavoring agent. As a
solvent, it is included in topical solutions and gels, and in edible
printing inks used for tablets.
Ethyl acetate has also been shown to increase the solubility of
chlortalidone and to modify the polymorphic crystal forms
obtained for piroxicam pivalate, mefenamic acid, and fluconazole,and has been used in the formulation of microspheres. Ethyl acetate has been used as a solvent in the preparation of a
liposomal amphotericin B dry powder inhaler formulation.(9) Its use
as a chemical enhancer for the transdermal iontophoresis of insulin
has been investigated.
In food applications, ethyl acetate is mainly used as a flavoring
agent. It is also used in artificial fruit essence and as an extraction
solvent in food processing. | | Safety Profile | Potentially poisonous by ingestion. Toxicity depends upon alcohols in question, generally ethanol with methanol as a denaturant. A flammable liquid and dangerous fire hazard; can react vigorously with oxidzing materials. Moderate explosion hazard. See ETHANOL, METHYL ALCOHOL, and n-PROPYL ALCOHOL. | | Safety | Ethyl acetate is used in foods, and oral and topical pharmaceutical
formulations. It is generally regarded as a relatively nontoxic and
nonirritant material when used as an excipient.
However, ethyl acetate may be irritant to mucous membranes,
and high concentrations may cause central nervous system
depression. Potential symptoms of overexposure include irritation
of the eyes, nose, and throat, narcosis, and dermatitis.
Ethyl acetate has not been shown to be a human carcinogen or a
reproductive or developmental toxin.
The WHO has set an estimated acceptable daily intake of ethyl
acetate at up to 25 mg/kg body-weight.
In the UK, it has been recommended that ethyl acetate be
temporarily permitted for use as a solvent in food and that the
maximum concentration consumed in food should be set at
1000 ppm.
LD50 (cat, SC): 3.00 g/kg
LD50 (guinea-pig, oral): 5.50 g/kg
LD50 (guinea-pig, SC): 3.00 g/kg
LD50 (mouse, IP): 0.709 g/kg
LD50 (mouse, oral): 4.10 g/kg
LD50 (rabbit, oral): 4.935 g/kg
LD50 (rat, oral): 5.62 g/kg | | Synthesis | By reacting acetic acid and ethanol in the presence of sulfuric acid; by distillation of sodium potassium, or lead acetate
with ethanol in the presence of sulfuric acid; by polymerizatin of acetaldehyde in the presence of aluminum ethylate or aluminum
acetate as catalysts. | | Potential Exposure | This material is used as a solvent for
nitrocellulose and lacquer. It is also used in making dyes,flavoring and perfumery, and in smokeless powder
manufacture | | First aid | If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seekmedical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately.If this chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPRif heart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, getmedical attention. Give large quantities of water andinduce vomiting. Do not make an unconscious personvomit | | Carcinogenicity | Ethyl acetate was not mutagenic in bacterial
assays; it was not genotoxic in a number
of in vivo assays but did cause chromosomal
damage in hamster cells in vitro.
Ethyl acetate has a fruity odor detectable
at 10ppm.
The 2003 ACGIH threshold limit valuetime-
weighted average (TLV-TWA) for ethyl
acetate is 400pm (1440mg/m3). | | Source | Identified among 139 volatile compounds identified in cantaloupe (Cucumis melo var.
reticulates cv. Sol Real) using an automated rapid headspace solid phase microextraction method
(Beaulieu and Grimm, 2001). | | Environmental fate | Biological. Heukelekian and Rand (1955) reported a 5-d BOD value of 1.00 g/g which is 54.9%
of the ThOD value of 1.82 g/g.
Photolytic. Reported rate constants for the reaction of ethyl acetate and OH radicals in the
atmosphere (296 K) and aqueous solution are 1.51 x 10-12 and 6.60 x 10-13 cm3/molecule?sec,
respectively (Wallington et al., 1988b).
Chemical/Physical. Hydrolyzes in water forming ethanol and acetic acid (Kollig, 1993). The
estimated hydrolysis half-life at 25 °C and pH 7 is 2.0 yr (Mabey and Mill, 1978). | | Metabolism | Ethyl acetate is hydrolysed to ethyl alcohol, which is then partly excreted in the expired air and urine. The rest is metabolized, the acetate fraction becoming incor porated in the body pool (Fassett, 1963). | | storage | Ethyl acetate should be stored in an airtight container, protected
from light and at a temperature not exceeding 30°C. Ethyl acetate is
slowly decomposed by moisture and becomes acidic; the material
can absorb up to 3.3% w/w water.
Ethyl acetate decomposes on heating to produce ethanol and
acetic acid, and will emit acrid smoke and irritating fumes. It is
flammable and its vapor may travel a considerable distance to an
ignition source and cause a ‘flashback’.
The alkaline hydrolysis of ethyl acetate has been shown to be
inhibited by polyethylene glycol and by mixed micelle systems. | | Shipping | UN1173 Ethyl acetate, Hazard Class: 3; Labels:
3-Flammable liquid. | | Purification Methods | The most common impurities in EtOAc are water, EtOH and acetic acid. These can be removed by washing with aqueous 5% Na2CO3, then with saturated aqueous CaCl2 or NaCl, and drying with K2CO3, CaSO4 or MgSO4. More efficient drying is achieved if the solvent is further dried with P2O5, CaH2 or molecular sieves before distillation. CaO has also been used. Alternatively, ethanol can be converted to ethyl acetate by refluxing with acetic anhydride (ca 1mL per 10mL of ester), the liquid is then fractionally distilled, dried with K2CO3 and redistilled. [Beilstein 2 III 127.] | | Toxicity evaluation | Ethyl acetate is rapidly hydrolyzed to ethanol and acetic acid.
When ethyl acetate was injected intraperitoneal at 1.6 g kg-1,
hydrolysis to acetic acid and ethanol occurred rapidly. The
biological half-life value of the conversion of ethyl acetate to
ethanol was found to be between 5 and 10 min. At doses higher
than 1.6 g kg-1 in rats the rate of hydrolysis exceeded the
ethanol oxidation leading to the ethanol accumulation in the
vascular system. | | Incompatibilities | Ethyl acetate can react vigorously with strong oxidizers, strong
alkalis, strong acids, and nitrates to cause fires or explosions. It also
reacts vigorously with chlorosulfonic acid, lithium aluminum
hydride, 2-chloromethylfuran, and potassium tert-butoxide. | | Waste Disposal | Dissolve or mix the material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal,
state, and local environmental regulations must be
observed. Consult with environmental regulatory agencies for guidance on acceptable disposal practices.
Generators of waste containing this contaminant (≧100 kg/
mo) must conform with EPA regulations governing storage,
transportation, treatment, and waste disposal. | | Regulatory Status | Included in the FDA Inactive Ingredients Database (oral tablets and
sustained-action tablets; topical and transdermal preparations).
Included in nonparenteral medicines licensed in the UK (tablets,
topical solutions, and gels). Ethyl acetate is also accepted for use in
food applications in a number of countries including the UK.
Included in the Canadian List of Acceptable Non-medicinal
Ingredients. |
| | Ethyl acetate Preparation Products And Raw materials |
|