- Ursolic acid
-

- $5.00 / 25kg
-
2024-04-26
- CAS:77-52-1
- Min. Order: 1kg
- Purity: ≥99%
- Supply Ability: 200mt/year
- Ursolic acid
-

- $30.00 / 1g
-
2024-04-26
- CAS:77-52-1
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 300tons
- Ursolic acid
-
- $0.00 / 1kg
-
2024-04-24
- CAS:77-52-1
- Min. Order: 0.10000000149011612kg
- Purity: ≥98%HPLC
- Supply Ability: 20tons
Related articles - Sources of usolic acid
- Ursolic acid (UA) is a well-studied natural pentacyclic triterpenoid found in herbs, fruit and a number of traditional Chinese....
- Apr 24,2022
|
| Product Name: | Ursolic acid | | Synonyms: | 3BETA-HYDROXY-12-URSEN-28-OIC ACID;3B-HYDROXYURS-12-EN-28-OIC ACID;Ursolic acid 3beta-Hydroxyurs-12-en-28-oic acid;(1S,2R,4aS,6aS,6bR,12aR,12bR,14bS)-10-hydroxy-1,2,6a,6b,9,9,12a-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylic acid;(3beta)-urs-12-en-28-oicaci;3-hydroxy-,(3.beta.)-Urs-12-en-28-oicacid;URSOLIC ACID hplc;Urs-12-en-28-oic acid, 3-hydroxy-, (3)- | | CAS: | 77-52-1 | | MF: | C30H48O3 | | MW: | 456.71 | | EINECS: | 201-034-0 | | Product Categories: | Natural Plant Extract;natural product;chemical reagent;pharmaceutical intermediate;phytochemical;reference standards from Chinese medicinal herbs (TCM).;standardized herbal extract;Intermediates & Fine Chemicals;Pharmaceuticals;chiral;Antineoplastic;oleanolic acid;Ursolic acid;Inhibitors;Triterpenoids;Plant extract;Organics;Tri-Terpenoids;Pentacyclic Triterpenes;77-52-1 | | Mol File: | 77-52-1.mol | 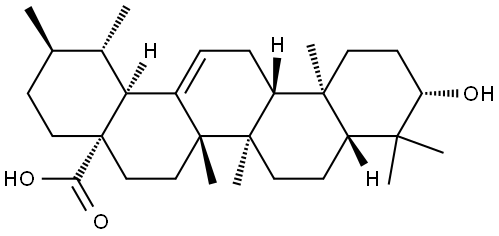 |
| | Ursolic acid Chemical Properties |
| Melting point | 292 °C (dec.) (lit.) | | alpha | 59 º (c=0.3, pyridine) | | Boiling point | 502.79°C (rough estimate) | | density | 1.0261 (rough estimate) | | refractive index | 1.4940 (estimate) | | storage temp. | 2-8°C | | solubility | Soluble in DMSO (up to 25 mg/ml with warming) | | form | Crystalline Powder or Needles | | pka | 4.68±0.70(Predicted) | | color | White to off-white | | Water Solubility | insoluble | | Merck | 14,9890 | | BRN | 2228563 | | Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 2 months. | | LogP | 8.731 (est) | | CAS DataBase Reference | 77-52-1(CAS DataBase Reference) | | EPA Substance Registry System | Urs-12-en-28-oic acid, 3-hydroxy-, (3.beta.)- (77-52-1) |
| | Ursolic acid Usage And Synthesis |
| Overview |
Ursolic acid (UA,77-52-1) (3β-hydroxy-urs-12-en-28-oic-acid) is a pentacyclic triterpenoid carboxylic compound (C30H48O3) which may occur in the free acid form or as aglycones for triterpenoid saponins. Its isomer, oleanolic acid (OA) (3β-hydroxy-olea-12-en-28-oic-acid), presents different substitution of the methyl group, but they have similar molecular structures and pharmacological activity[1]. Oleanolic acid and ursolic acid are ubiquitous triterpenoids in plant kingdom, medicinal herbs, and are integral part of the human diet, once they are found in fruits and medicinal herbs.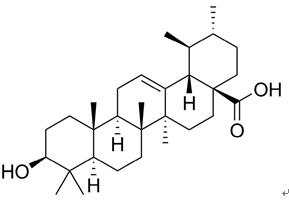
Figure 1 the chemical structure of Ursolic acid
In recent years they became the subject of many publications because of their various activities and low toxicity. Many beneficial effects such as antioxidative, antimicrobial, anti-inflammatory, anticancer, anti-hyperlipidemic, analgesic, hepatoprotectory, gastroprotective, anti-ulcer, anti-HIV, cardiovascular, antiatherosclerotic and immunomodulatory effects have been reported[2, 3]. Several works have shown that the ursolic acid can also stimulate muscle growth, reduce fat gain and enhance the epidermal permeability barrier recovery in the skin[4], therefore it has been proposed as a skin therapeutic agent and it could be introduced in sport supplements[5], cosmetics[6] and health products[7].
| | Origins |
Ursolic acid(77-52-1) can be isolated from various medicinal plants, Lamiaceae family being one of the most known sources of triterpenes, with contents up to 2.95% d.w[8]. In Rosmarinus officinalis leaves, one of the traditional commercial sources[9]. UA has also been identified in a variety of sources, particularly in leaves and flowers[10]. The triterpenic acids oleanolic and ursolic were recently detected for the first time in wild edible mushrooms[11] and in some commercial dried fruits[12].
UA content in the samples from different sources were significantly different, and the geographic variation was also observed in Paulownia fortune, Ocimum species, Argania spinosa[13]. The outer barks of Eucalyptus trees from temperate and Mediterranean zones are richer in triterpenic acids than the species from sub-tropical and tropical regions[14].
| | Properties and biological effects |
Ursolic acid(77-52-1) displayed lower toxicity than OA, 0.95 up from 0.10 mg/mL, respectively[15], but the data ranged depending on the raw material source, the extraction solvent, the type of activity, the disease, the clinical, cosmetically or pharmacological study, the study in animal or human, etc. Bioavailability studies confirmed a low plasma concentrations of UA in plasma of mice orally administered with high dose of UA, suggested either high binding activity in organs or low bioavailability or metabolism by the gut wall of the intestine and liver[16]. Despite many aspects of the biological activities of UA are not completely understood, the pharmacological effects could be attributed, in part, to their action against free radicals. Cardiovascular protection and anti-hyperlipidemic effect Cardiovascular protection, antihyperlipidemic (triglycerides, total cholesterol and lipoprotein fractions), antioxidant (glutathione peroxidase and superoxide dismutase activities), hypoglycemic effects and prevention of hypertension were observed in rats, displaying UA a low toxicity (LC50 0.95 mg/mL). Ursolic acid fed to rabbits and rats prevented the experimental atherosclerosis and lowered blood cholesterol[17]. A strong synergistic effect derived from the combination of ursolic acid and artesunate can reduce both triglyceride and cholesterol, showing more potent effects than either agent alone. Ethanolic extract from the stems and roots of Celastrus orbiculatus Thunb decreases athero-susceptibility in lipoproteins and the aorta of guinea pigs fed a high-fat diet, and increases high-density lipoprotein; likewise, it reduce lipid accumulation and promote reverse cholesterol transport in vivo and in vitro[18].
Antitumor effect
Beneficial action of ursolic acid and its derivatives was reported based on their anti-tumor, including inhibition of angiogenesis, invasion of tumor cells and metastasis, induction of apoptosis in tumor cells and prevention of malignant transformation of normal cells, and it also interferes with numerous enzymes[19]. Clinical tests suggesting the possibility of practical use of UA have already been conducted[20]. Ursolic acid was identified as active components of different plants for inhibiting mutagenicity and tumor-promotion. Ursolic acid exert anticancer effects in various cancer cell systems[20], it may have a potential application as a chemopreventive agent in gastric cancer, colorectal carcinogenesis and tumors of the colon, lung cancer, prostate cancer, UA pretreatment potentiated cell cycle arrest and UV radiation-induced apoptosis selectively in skin melanoma cells[21].
Antimicrobial
Ursolic acid and its derivatives have shown growth inhibition of gram-positive and gram-negative bacteria and fungi, and UA showed ability to control bacterial growth, biofilm formation, and elastase activity[22]. Synergistic effect of UA and two semi-synthetic derivatives and the aminoglycosides antibiotics neomycin, amikacin, kanamycin and gentamicin towards twelve bacterial pathogens strains was observed. Ursolic acid is the active components in natural extracts to inhibit the growth of some food-associated bacteria and yeast, protozoa (Trypanosoma cruzi), promoting an anti-inflammatory response during Leishmania infection[23].
Anti-diabetes, anti-inflammatory and anti-aging effect
Ursolic acid exhibits potential anti-diabetic and immunomodulatory properties by increasing insulin levels with preservation of pancreatic beta-cells and modulating blood glucose levels, T-cell proliferation and cytokines production by lymphocytes in type 1 diabetic mice fed a high-fat diet, compared to the diabetic group[24]. Type-2 diabetes is associated with obesity and ursolic acid may prevent or treat this disorder and its related comorbidities by acting as hypoglycemic and anti-obesity agent with five effects: reduction of the absorption of glucose, decrease the endogenous glucose production and increase the glycogen synthesis, increase the insulin sensitivity, improvement of lipid homeostasis, and promotion of the body weight regulation[9].
UA was twice as potent as indomethacin, and has been proposed for the treatment of rheumatism, fever and arthritis. The mechanisms of the anti-inflammatory action of ursolic acid and novel derivatives may be ascribed to inhibition of histamine release from mast cells, lipoxygenases, cyclooxygenases activity, inducible nitric oxide synthase and elastase, suppress the inflammatory cytokine-induced expression of E-selectin (an early response adhesion molecule expressed on the surface of endothelial cells during inflammation) in endothelial cells via inhibition of NF-kappa B activation[25] and suppressed the production of intracellular reactive oxygen species[26].
The epidermal permeability barrier plays a crucial role in human physical, chemical and biological cutaneous functions. Sensitive skin, associated with increased transepidermal water loss, penetrability and susceptibility to irritants, is related with impaired barrier function and reduction in ceramides. The atopic dermatitis displays impaired epidermal permeability barrier function, diminished water-holding properties and decreased ceramide levels. UA are great moisturizing candidates that do not create any cutaneous irritations[27]. Induction of ceramide synthesis is usually associated with keratinocyte differentiation. The ursolic acid increases the expression of genes required for terminal keratinocyte differentiation (involucrin, loricrin and filaggrin), improving the recovery of skin barrier function.
| | References |
- Lim, S. W et al (2007). Journal of Dermatology, 34(9), 625–634.
- Kashyap, D., Tuli, H. S., & Sharma, A. K. (2016). Life Sciences,146, 201–213.
- Wo?niak, L., Sk?pska, S., & Marsza?ek, K. (2015). Molecules, 20(11), 20,614–20,641.
- Kunkel, S. D. et al (2012). PLoS ONE, 7(6)
- Deane, C. S., et al (2017). American Journal of Physiology Endocrinology and Metabolism, 321(4), E282–E299.
- De Almeida et al (2014). Journal of Thermal Analysis and Calorimetry, 115(3), 2401–2406.
- Navina, R., Lee, Y. G., & Kim, S. M. (2017). Molecular biological roles of ursolic acid in he treatment of human diseases. Current Bioactive Compounds, 13(3), 177–185.
- Razborsek, M. I., Voncina, D. B., Dolecek, V., & Voncina, E. (2008). Determination of oleanolic, betulinic and ursolic acid in Lamiaceae and mass spectral fragmentation of their trimethylsilylated derivatives. Chromatographia, 67(5–6), 433–440
- Silva, F. S. G., Oliveira, P. J., & Duarte, M. F. (2016). Oleanolic, ursolic, and betulinic acids as food supplements or pharmaceutical agents for type 2 diabetes: Promise or Illusion? Journal of Agricultural and Food Chemistry, 64, 2991–3008.
- Han, S. K., et al (2014). Oleanolic acid from Fragaria ananassa calyx leads to inhibition of alpha-MSH-induced melanogenesis in B16-F10 melanoma cells. Journal of the Korean Society for Applied Biological Chemistry, 57(6), 735–742
- Kalogeropoulos, N., Yanni, A. E., Koutrotsios, G., & Aloupi, M. (2013). Toxicology, 55, 378–385.
- Zhang, F., Daimaru, E., Ohnishi, M., Kinoshita, M., & Tokuji, Y. (2013). Food Science and Technology Research, 19(1), 113–116.
- Guinda, Á., Rada, M., Delgado, T., & Castellano, J. M. (2011). Pentacyclic triterpenic acids from Argania spinosa. European Journal of Lipid Science and Technology, 113(2), 231–237.
- Domingues, R. M. A., et al (2011). Cellulose Chemistry and Technology, 45(7–8), 475–481
- Somova, L. I., Shode, F. O., & Mipando, M. (2004). Phytomedicine, 11, 121–129.
- Wójciak-Kosior, M., Sowaa, I., Kocjan, R., & Nowak, R. (2013). Industrial Crops and Products, 44, 373–377.
- Liu, J. (1995). Pharmacology of oleanolic acid and ursolic acid. Journal of Ethnopharmacology, 49, 57–68.
- Zhang, Y., Si, Y., et al (2016). Lipids, 51, 677–692.
- Dong, H., et al (2015). Biochemical Pharmacology, 93(2), 151–162.
- Wo?niak, L., Sk?pska, S., & Marsza?ek, K. (2015). Molecules, 20(11), 20,614–20,641
- Lee, Y. H., Wang, E. X., Kumar, N., & Glickman, R. D. (2014). Ursolic acid differentially modulates apoptosis in skin melanoma and retinal pigment epithelial cells exposed to UV–VIS broadband radiation. Apoptosis, 19(5), 816–828.
- Gilabert, M., Marcinkevicius, K., Andújar, S., Schiavone, M., Arena, M. E., & Bardón, A. (2015). Sesquiand triterpenoids from the liverwort Lepidozia chordulifera inhibitors of bacterial biofilm and elastase activity of human pathogenic bacteria. Phytomedicine, 22(1), 77–85.
- Rodrigues, I. A., et al (2015). Natural products: Insights into Leishmaniasis inflammatory response. Mediators of Inflammation (Article number 835910).
- Jang, S. M., et al (2009). International Immunopharmacology, 9(1), 113–119.
- Takada, K., Nakane, T., Masuda, K., & Ishii, H. (2010). Phytomedicine, 17(14), 1114–1119
- Kim, E. S., & Moon, A. (2015). Oncology Letters, 9(2), 897–902.
- Both, D. M., Goodtzova, K., Yarosh, D. B., & Brown, D. A. (2002) Archives of Dermatological Research, 293(11), 569–575
- Gribble, G. W., Fu, L., Sporn, M. B., & Liby, K. T. (2015). Triterpenoids and compositions containing the same. USA Patent US 2015011627.
- Asami, C., & Lida, M. (2014). Lip cosmetics. Japan Patent JP 2014237594.
- Akamatsu, H., Suzuki, M., & Sakai, Y. (2007a). Cosmetic for improving skin barrier function. Japan Patent JP 2007161610.
- Akamatsu, H., Suzuki, M., & Sakai, Y. (2007b). Skin care preparation for improving skin barrier function. Japan Patent JP 200716161
- Kobayashi, K., Imamura, H., & Seto, M. (2008). Skin care preparation containing antiinflammatory component. Japan Patent JP 2008115109.
- Ryu, S. H., et al (2013). Pharmaceutical composition comprising expression or activity inhibitor of TENC1 for preventing or treating diabetes mellitus. South Korea Patent KR 20130088054.
- Junhai, L., et al (2012). Methods for extracting ursolic acid from red jujube. China Patent CN 102321144.
- Anonymous (The inventor has waived the right to be mentioned). (2013).
- Ping, Z., Yong, L., & Chengbiao, X. (2011). Medical application of ursolic acid-NF (Nuclear Factor)-κB inhibitor. China Patent CN 102206243.
| | Chemical Properties | white to light yellow crystal powde | | Uses | Ursolic Acid is a Triterpene acid used in cosmetics, that also has STAT3 pathway inhibiting properties. It inhibits endothelial cell proliferation and migration (IC50=5 μM) and angiogenesis. Promotes skeletal muscle rejuvenation via enhanced SIRT1 expression. Induces apoptosis in malignant mesothelioma cells. Ursolic acid protects against muscle atrophy and boosts muscle growth (see also tomatidine T538500). | | Application | ursolic acid helps maintain the look and feel of the skin, acts as a perfume and helps mask odor. Therapeutic benefits in skin care include anti-inflammatory activity. ursolic acid has demonstrated anti-microbial, anti-bacterial, and anti-fungal action as well. | | Definition | ChEBI: Ursolic acid is a pentacyclic triterpenoid that is urs-12-en-28-oic acid substituted by a beta-hydroxy group at position 3. It has a role as a plant metabolite and a geroprotector. It is a pentacyclic triterpenoid and a hydroxy monocarboxylic acid. It derives from a hydride of an ursane. | | General Description | Ursolic acid (UA) is a hydroxyl pentacyclic triterpenoic acid (HPTA), which exhibits anti-bacterial, anti-cancer, anti-oxidative and anti-inflammatory effects. It can also promote neuroregeneration after peripheral nerve injury. UA can enhance sleep duration by activating the GABAergic neurotransmitter system. [GABA= γ-aminobutyric acid] | | Biochem/physiol Actions | Triterpenoid found in a variety of fruits, including apples. Cardioprotective and anti-tumor agent. Under study as a potential Alzheimer′s disease therapeutic due to its inhibitory effect on the interactions between amyloid-β and the CD36 receptor. | | Anticancer Research | It is a triterpenoid, derived from basil, and suppresses the activation of NF-κB byinhibiting a kinase that activates NF-κB(IKK) (Aggarwal and Shishodia 2004). It suppresses tumorigenesis, tumor promotion, and angiogenesis by downregulationof the expression of lipoxygenases (LOX), MMP-9, and COX-2. It is reported toinduce apoptosis in breast cancer, melanoma, hepatoma, prostate cancer, andacute myelogenous leukemia by preventing replication of DNA, inhibition of proteintyrosine kinases, activation of caspases, induction of calcium release, anddownregulating apoptosis gene cellular inhibitor. Ursolic acid inhibits IκBαkinase activity, degradation of IκBα, phosphorylation, NF-κB-dependent reportergene expression, p65 nuclear translocation, and p65 phosphorylation. Inhibitionof NF-κB leads to the reduction of cyclin D1, COX-2, and MMP9 expression.This downregulates STAT-3 activation and its regulated gene products such ascyclin D1, Mcl-1, Bcl-xL, survivin, Bcl-2, and VEGF. It also induces the expressionof tyrosine phosphatase SHP-1 protein and of mRNA (Aggarwal et al. 2008;Hsu et al. 2004). | | Side effects | Adverse effects of Ursolic acid include reduction of sperm viability and inhibition of spermatogenesis, leading to infertility. Ursolic acid is capable of inducing endothelial cell death at concentrations above 12.5uM, and the mechanism of death is related to apoptosis. In addition, it also appears to cause DNA strand breaks. | | Cytotoxicity | IC50 (μg/mL): 6.7 (518A2), 5.3 (A2780), 7(A549), 6.4 (FaDu), 4.8 (HT29), 5.8(MCF-7), 8.5 (NIH3T3) (Wiemann et al.2016)
IC50 (μg/mL): 12.38 (MGC-803),17.12(HCT-116), 13.39 (T24), 13.81(HepG2), 16.36 (A549), >45.7 (HL-7702)(Hua et al. 2015). |
| | Ursolic acid Preparation Products And Raw materials |
|