Sodium ketoisocaproate manufacturers
- Sodium ketoisocaproate
-

- $1.00 / 1KG
-
2020-01-03
- CAS:4502-00-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10000KGS
|
| | Sodium ketoisocaproate Basic information |
| Product Name: | Sodium ketoisocaproate | | Synonyms: | Sodium ketoisocaproate;SODIUM 4-METHYL-2-OXOVALERATE;SODIUM ALPHA-KETOISOCAPRATE;2-KETOISOCAPROIC ACID SODIUM SALT;2-KETOISOHEXANOIC ACID SODIUM SALT;A-KETOISOCAPROIC ACID, SODIUM SALT;ALPHA-KETOISOCAPROIC ACID SODIUM SALT;4-Methyl-2-oxopentanoic acid monosodium salt | | CAS: | 4502-00-5 | | MF: | C6H9NaO3 | | MW: | 152.12 | | EINECS: | 224-816-3 | | Product Categories: | pharmacetical | | Mol File: | 4502-00-5.mol | 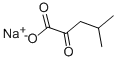 |
| | Sodium ketoisocaproate Chemical Properties |
| Melting point | 275 °C (dec.)(lit.) | | storage temp. | 2-8°C | | solubility | Water (Slightly) | | form | Solid | | color | White to Off-White | | Odor | at 100.00 %. butyric valeric buttery | | Odor Type | buttery | | JECFA Number | 633.1 | | BRN | 4239297 | | Stability: | Hygroscopic | | LogP | 0.17 | | CAS DataBase Reference | 4502-00-5(CAS DataBase Reference) |
| | Sodium ketoisocaproate Usage And Synthesis |
| Occurrence | Reported found in banana, asparagus, cheese, white wine, cocoa, blue cheese and provolone cheese. | | Uses | Sodium 4-methyl-2-oxovalerate has been used for quantifying branched-chain keto acids from cell extracts by HPLC-fluorescence method. | | Uses | 4-Methyl-2-oxovaleric Acid is an α-ketomonocarboxylic acid that triggers insulin release by acting upon receptor sites which differ from those occupied by amino acids. 4-Methyl-2-oxovaleric Acid is an intermediate in the metabolism of Leucine. | | Biochem/physiol Actions | α-Ketoisocaproic acid is a leucine metabolite that is known to induce insulin secretion from the βcells of pancreas. Enteral infusion of aα-Ketoisocaproate is found to ameliorate, endotoxemic condition. The resulting ketone bodies obtained from its conversion, might serve as an energy source. Accumulation of α-Ketoisocaproic acid is characterized in branched chain ketoaciduria, which is caused due to the lack of branched chain L-2-keto acid dehydrogenase activity. α-Ketoisocaproic acid causes dissociation in the oxidative phosphorylation reaction and acts as a metabolic inhibitor of α-ketoglutarate dehydrogenase. Thus, leads to a defect in the mitochondrial homeostasis, observed in branched chain ketoaciduria. |
| | Sodium ketoisocaproate Preparation Products And Raw materials |
|