- Sorafenib tosylate
-

- $44.00 / 5g
-
2024-09-25
- CAS:475207-59-1
- Min. Order: 5g
- Purity: 0.98
- Supply Ability: 25kg
- Sorafenib Tosylate
-

- $0.00 / 1g
-
2024-09-25
- CAS:475207-59-1
- Min. Order: 1g
- Purity: More Than 99%
- Supply Ability: 100kg/Month
- Sorafenib Tosylate
-

- $0.00 / 1kg
-
2024-09-25
- CAS:475207-59-1
- Min. Order: 1kg
- Purity: 99%,single impurity<0.1
- Supply Ability: 1 ton
Related articles - About Sorafenib tosylate
- Sorafenib is used to treat kidney, liver, and thyroid cancer. It is a chemotherapy drug that works by slowing or stopping the ....
- Aug 14,2019
|
| Product Name: | Sorafenib tosylate | | Synonyms: | SORAFENIB TOSYLATE;Sorafenib & its intermediates;Sorafinib mesylate;4-Methyl-3-((4-(3-pyridinyl)-2-pyrimidinyl)amino)-N-(5-(4-methyl-1H-imidazol-1-yl)-3-(trifluoromethyl)phenyl)benzamide monomethanesulfonate;Sorafenib Tosylate(Bay 43-9006,Nexavar);2-PyridinecarboxaMide, 4-[4-[[[[4-chloro-3-(trifluoroMethyl)phenyl]aMino]carbonyl]aMino]phenoxy]-N-Methyl-, 4-Methylbenzenesulfonate;Sorafenib Tosylate (Nexavar);Sorafenib (Nexavar) | | CAS: | 475207-59-1 | | MF: | C21H16ClF3N4O3.C7H8O3S | | MW: | 637.03 | | EINECS: | 641-758-8 | | Product Categories: | Bay 43-9006;Inhibitors;MAPK;API | | Mol File: | 475207-59-1.mol | 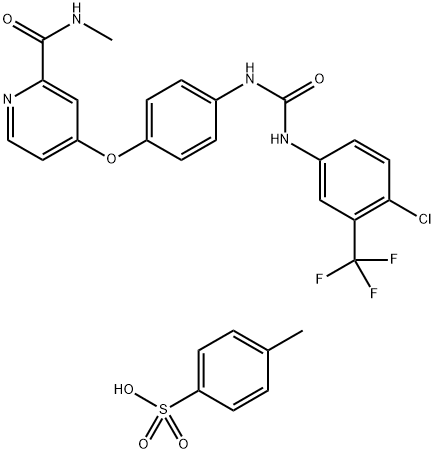 |
| | Sorafenib tosylate Chemical Properties |
| Melting point | 229-232°C | | storage temp. | 2-8°C(protect from light) | | solubility | Soluble in DMSO (up to 200 mg/ml) or in Ethanol (up to 3 mg/ml). | | form | solid | | color | Off-white | | Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 1 month. | | InChIKey | IVDHYUQIDRJSTI-UHFFFAOYSA-N | | SMILES | S(C1C=CC(C)=CC=1)(O)(=O)=O.O(C1C=CC(NC(=O)NC2C=CC(Cl)=C(C(F)(F)F)C=2)=CC=1)C1=CC=NC(C(=O)NC)=C1 | | CAS DataBase Reference | 475207-59-1(CAS DataBase Reference) |
| | Sorafenib tosylate Usage And Synthesis |
| Indications and Usage | Sorafenib tosylate is a new type of multi-target antitumor drug, was developed by the German Bayer Pharmaceuticals, and displayed expansive antitumor activity in preclinical animal tests. Sorafenib tosylate is mainly used in advanced liver cancer treatment and is well tolerated.
| | Mechanisms of Action | Sorafenib tosylate can simultaneously affect tumor cells and tumor blood vessels. It has a double antitumor effect: it can block the cell signal transduction pathways mediated by RAF/MEK/ERK to directly inhibit tumor cell growth, while also inhibiting VEGF and platelet derived growth factors (PDGF) receptors to prevent the formation of new tumor blood vessels, thus indirectly inhibiting tumor cell growth.
| | Clinical Research | In a stage three randomized clinical study of American and European treatments for advanced kidney cancer, 903 cases advanced kidney patients who had experienced one unsuccessful systematic treatment (biological immunity or chemotherapy) were randomly split into two groups. One group received sorafenib tosylate, while the other received a placebo. Mid-term analysis found already 222 deaths and the objective efficacy of the two groups to be 10% and 2%, while 74% and 53% of patients had stabilized conditions. The sorafenib tosylate group’s survival period was longer than that of the placebo group, with the risk ratio as 0.72. However, this different does not have statistical significance, as this is only a mid-term analysis, and final analysis after treatment will yield the correct survival period comparison.
602 advanced liver cancer patients selected from the USA, Europe, Australia, and other countries and areas, who did not receive systematic treatment, were included in a study. Results showed that compared to the placebo, taking sorafenib tosylate could extend their total survival period of patients with late stage or primary liver cancer by about 44% and their extend disease progression period by about 73%.
| | Adverse reactions | Manageable diarrhea, rashes, fatigue, hand-foot syndrome, hypertension, alopecia, nausea, vomiting, and loss of appetite.
| | Description | Sorafenib (475207-59-1) was initially developed as a Raf kinase inhibitor, IC50 = 6 nM, but has been shown to inhibit many receptor tyrosine kinases including BRAF (IC50 = 22 nM); VEGFR-2 (IC50 = 90 nM); VEGFR-3 (IC50 = 20 nM); PDGFR-β (IC50 = 57 nM); Flt3 (IC50 = 58 nM); c-KIT (IC50 = 68 nM); FGFR-1 (IC50 = 580 nM).1?Paradoxically more potent in a cellular assay (IC50 = 20 nM) compared to an isolated enzyme assay (IC50 = 107 nM) for c-Fms.2?Inhibits activation of MAPK pathway and ERK phosphorylation.3?Induces caspase-independent apoptosis in melanoma cells.4?Sorafenib is a clinically useful anticancer agent. | | Uses | Sorafenib Tosylate (Bay 43-9006, Nexavar) is a small molecular inhibitor of VEGFR, PDGFR, c-Raf and B-Raf with IC50s of 18 nM, 10 nM, 3 nM and 15 nM, respectively. | | Definition | ChEBI: Sorafenib tosylate is an organosulfonate salt. It contains a sorafenib. | | Brand name | Nexavar (Bayer). | | General Description | Sorafenib is available in 200-mg tablets for oral administrationand is used in the treatment of RCC and colon cancer.The agent is classified as a multikinase inhibitor because ofits action on numerous kinase enzymes including thePDGF-R, VEGF-R, Kit, and Raf. Sorafenib is 39% to 48% bioavailable and CYP3A4-mediated metabolism giveseight identified metabolites including the N-oxide, which isequally active with the parent. However, the majority of thedrug in plasma is present as the parent compound.Sorafenib is highly protein bound (99.5%). The drug iseliminated primarily in the feces (77%) with 19% appearingin the urine as glucuronides (UGT1A1) and has a eliminationhalf-life of 24 to 48 hours. The most commonly seentoxicity is skin rash that normally occurs within the first6 weeks of therapy. Other adverse effects include hypertension,fatigue, increased wound healing time, and increasedrisk of bleeding. | | Clinical Use | Sorafenib is used to treat late-stage kidney cancer (advanced renal cell carcinoma), liver cancer (hepatocellular carcinoma) that cannot be treated by surgery, and differentiated thyroid cancer that has come back or spread to other parts of your body. Sorafenib is an antineoplastic (cancer) agent. It interferes with the growth of cancer cells, which are eventually destroyed by the body. | | Side effects | The side effects of Sorafenib tosylate include Bleeding gums, blistering, peeling, redness, or swelling of the palms of the hands or bottoms of the feet, bloating of the abdomen or stomach,
blood in the urine or stools, clay-colored stools,
coughing up blood, difficulty with breathing or swallowing, increased menstrual flow or vaginal bleeding. | | target | Raf-1 | | References | 1) Wilhelm et al. (2004), BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis; Cancer Res., 64 7099
2) Guo et al. (2006), Inhibition of phosphorylation of the colony-stimulating factor-1 receptor (c-Fms) tyrosine kinase in transfected cells by ABT-869 and other tyrosine kinase inhibitors; Mol. Cancer Ther., 5 1007
3) Wilhelm et al. (2003), The novel Raf inhibitor BAY 43-9006 blocks signaling and proliferation in BRAF mutant and wildtype melanoma and colorectal tumor cell lies; Proc. Am. Assoc. Cancer Res., 44 106609
4) Panka et al. (2006), The Raf inhibitor BAY 43-9006 (Sorafenib) induces caspase-independent apoptosis in melanoma cells; Cancer Res., 66 1611 |
| | Sorafenib tosylate Preparation Products And Raw materials |
|