Learn More About Ammonium Acetate
The passage introduces the physical and chemical properties, uses and hazards of Ammonium acetate.
Nov 22,2022 Organic ChemistryThe Side Effects of Cetearyl Alcohol
The passage introduces the side effects of Cetearyl alcohol.
Nov 22,2022 Organic reagentsAcetyl Chloride: Preparation and Hazards
The passage introduces the preparation and hazards of Acetyl chloride.
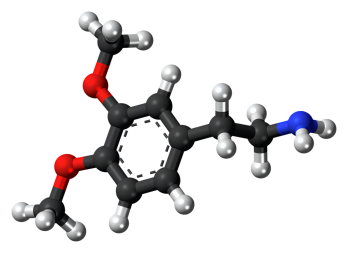
Nov 22,2022 Organic Raw MaterialThe Benefits of L-Tyrosine
The passage introduces the benefits of L-Tyrosine.
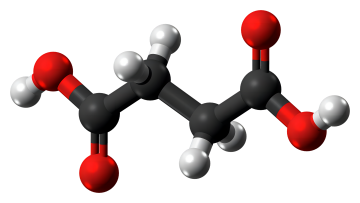
Nov 22,2022 Amino Acids and DerivativesSuccinic Acid: Solubility and Mechanism of Action
The passage introduces the solubility and mechanism of action of succinic acid.
Nov 22,2022 Organic ChemistryBioactivity of Human Growth Hormone
Human Growth Hormone (HGH) is composed of growth hormones naturally found in bacteria that are linked to signaling peptides.
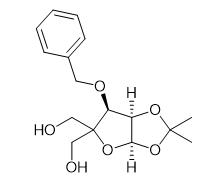
Nov 22,2022 Hormones and the Endocrine SystemSynthesis and Application of 3-O-Benzyl-4-(hydroxymethyl)-1,2-O-isopropylidene-a-D-ribofuranose
3-O-Benzyl-4-(hydroxymethyl)-1, 2-O-isopropylidene-a-d-ribofuranose is a research chemical.
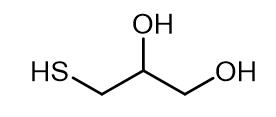
Nov 22,2022 Organic ChemistryApplication of 3-Mercapto-1,2-propanediol
3-Mercapto-1,2-propanediol is a chemical.
Nov 22,2022 Organic ChemistryLearn More About Zinc Chloride
The passage introduces the chemical and physical properties and toxicity of Zinc chloride.
Nov 21,2022 APIThe Benefits of Citric Acid Monohydrate
The passage introduces the benefits of Citric acid monohydrate.
Nov 21,2022 Organic Raw Material