| Identification | More | [Name]
VINYL FLUORIDE | [CAS]
75-02-5 | [Synonyms]
FLUOROETHYLENE
Monofiuoroethylene
MONOFLUOROETHYLENE
VINYL FLUORIDE
Vinyl fluoride,inhibied
1-Fluoroethylene
C2H3F
ethene,fluro-
Ethylene, fluoro-
ethylene,fluoro-
fluoroalkenes,vinylfluoride
fluoro-ethen
Fluoroethene
fluoro-ethene
fluoro-ethylen
monofluoroethene
Vinyl fluoride, inhibited
vinylfluoride(vf)
vinylfluoride,inhibited
Fluoroethene~Fluoroethylene | [EINECS(EC#)]
200-832-6 | [Molecular Formula]
C2H3F | [MDL Number]
MFCD00042113 | [Molecular Weight]
46.04 | [MOL File]
75-02-5.mol |
| Safety Data | Back Directory | [Hazard Codes ]
F | [Risk Statements ]
R12:Extremely Flammable.
R40:Limited evidence of a carcinogenic effect. | [Safety Statements ]
S9:Keep container in a well-ventilated place .
S16:Keep away from sources of ignition-No smoking .
S23:Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer) .
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection . | [OEB]
D | [OEL]
TWA: 1 ppm, Ceiling: 5 ppm [use 1910.1017] | [RIDADR ]
1860 | [Hazard Note ]
Flammable | [TSCA ]
TSCA listed | [REACH Registrations]
Active | [HazardClass ]
2.1 | [HS Code ]
2903290000 | [Safety Profile]
Confirmed carcinogen.
A poison. Mutation data reported. A very
dangerous fire hazard. To fight fire, stop
flow of gas. When heated to decomposition
it emits toxic fumes of F-. See also
FLUORIDES. | [Hazardous Substances Data]
75-02-5(Hazardous Substances Data) |
| Raw materials And Preparation Products | Back Directory | [Raw materials]
Sodium hydroxide-->Hydrogen fluoride-->Silica gel-->Potassium dichromate-->CHLOROETHANE-->Cupric oxide-->difluoroethane-->(1Z)-1,2-Dichloro-3,3,3-trifluoroprop-1-ene-->(1E)-1,2-Dichloro-3,3,3-trifluoroprop-1-ene-->2-Chloro-1,1,1,2-tetrafluoropropane-->2-IODO-1,1-DIFLUOROETHANE-->2,3,4,5,6-PENTAFLUOROSTYRENE-->2,3,3,3-TETRAFLUOROPROPENE | [Preparation Products]
potassium ω-hydroperfluoyope largonate-->trans-1-Fluoro-1-propene-->2-CHLORO-3,3,3-TRIFLUOROPROPENE-->Trifluoromethane |
| Hazard Information | Back Directory | [General Description]
A colorless gas with a faint ethereal odor. Shipped as a confined liquid under its vapor pressure. Any leak can either be liquid or vapor. Contact with the liquid can cause frostbite. Easily ignited. Vapors are heavier than air. Can asphyxiate by the displacement of air. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket. | [Reactivity Profile]
VINYL FLUORIDE is light sensitive, peroxidizable monomer may initiate exothermic polymerization of the bulk material [Handling Chemicals Safely 1980. p. 958]. Sensitive to many oxidants. | [Air & Water Reactions]
Highly flammable, reacts with air to form peroxides | [Health Hazard]
Inhalation of vapor causes slight intoxication, some shortness of breath. Liquid may cause frostbite of eyes or skin. | [Potential Exposure]
Vinyl fluoride’s primary use is as a
chemical and polymer intermediate; used to make polyvinyl
fluoride (Tedlar) film. Polyvinyl fluoride film is characterized
by superior resistance to weather, high strength; and a
high dielectric constant. It is used as a film laminate for
building materials and in packaging electrical equipment.
Polyvinyl fluoride film poses a hazard, so it is not recommended
for food packaging. Polyvinyl fluoride evolves
toxic fumes upon heating. | [First aid]
If this chemical gets into the eyes, remove any
contact lenses at once and irrigate immediately for at least
15 minutes, occasionally lifting upper and lower lids. Seek
medical attention immediately. If this chemical contacts
the skin, remove contaminated clothing and wash immediately
with soap and water. Seek medical attention immediately.
If this chemical has been inhaled, remove from
exposure, begin rescue breathing (using universal precautions,
including resuscitation mask) if breathing has stopped
and CPR if heart action has stopped. Transfer promptly to a
medical facility. When this chemical has been swallowed,
get medical attention. Give large quantities of water and
induce vomiting. Do not make an unconscious person
vomit. If frostbite has occurred, seek medical attention
immediately; do NOT rub the affected areas or flush them
with water. In order to prevent further tissue damage, do
NOT attempt to remove frozen clothing from frostbitten
areas. If frostbite has NOT occurred, immediately and thoroughly
wash contaminated skin with soap and water. | [Shipping]
UN1860 Vinyl fluoride, inhibited, Hazard Class:
2.1; Labels: 2.1-Flammable gas. | [Incompatibilities]
May polymerize. Inhibited with 0.2%
terpenes to prevent polymerization. Violent reaction with
oxidizers. May accumulate static electrical charges. | [Description]
Vinyl fluoride (VF) was first synthesized by Frederic Swarts,
a Belgian chemist in 1901, by the reaction between zinc and
1,1-difluoro-2-bromoethane. Modern preparation involves
the reaction of acetylene and hydrogen fluoride (HF) in the
presence of a mercury- or aluminum-based catalyst. The US
Environmental Protection Agency (EPA) listed VF as a highproduction-
volume chemical in 1990. According to National
Toxicology Program (NTP), 2005, the annual production of
VF in the United States was above 1 million pounds
(454 000 kg) in 1990 and approximately 3.3 million pounds
(1.5 million kg) in 2001. | [Chemical Properties]
Colorless gas. Insoluble in
water; soluble in alcohol and ether. | [Chemical Properties]
Vinyl fluoride is a colorless gas. | [Uses]
Since the 1960s, VF has mainly been used in the production of
polyvinyl fluoride (PVF) and other fluoropolymers. Polymers
of VF have excellent resistance to degradation by sunlight,
chemical attack, and water absorption and exhibit great
strength, chemical inertness, and low permeability to air and
water. PVF is laminated with aluminum, galvanized steel, and
cellulose materials and is used as a protective surface for the
exteriors of residential and commercial buildings. PVF laminated
with various plastics has been used to cover walls, pipes,
and electrical equipments and inside aircraft cabins. PVF is sold
under the trademarks Tedlar PVF film and Dalvor. Due to
increase in demand for solar panels, the demand for photovoltaic
materials such as Tedlar is high, forcing the manufacturer
to boost VF production. | [Uses]
Vinyl fluoride is used primarily in the production of polyvinyl fluoride and other fluoropolymers. Polymers of vinyl fluoride are resistant to weather and exhibit great strength, chemical inertness, and low permeability to air and water. Polyvinyl fluoride is laminated with aluminum, galvanized steel, and cellulose materials and is used as a protective surface for the exteriors of residential and commercial buildings. Polyvinyl fluoride laminated with various plastics has been used to cover walls, pipes, and electrical equipment and inside aircraft cabins (IARC 1995). | [Definition]
ChEBI:Fluoroethene is a monohaloethene and a gas molecular entity. | [Preparation]
Vinyl fluoride may be obtained from acetylene by either of the two following
routes:
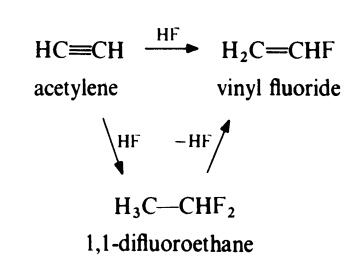
In the first method, acetylene is heated with hydrogen fluoride in the presence
of a catalyst of mercuric chloride on charcoal at about 40??C to yield vinyl
fluoride directly. In the second method, acetylene is treated with an excess of
hydrogen fluoride to form difluoroethane which is then pyrolysed at about
700??C in a platinum tube to give vinyl fluoride, which is separated by
distillation under pressure.
Vinylidene fluoride is obtained from vinylidene chloride by the
following route:
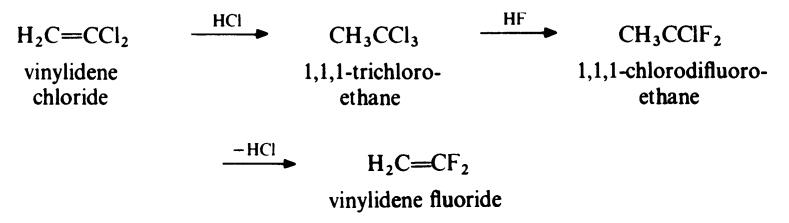
In the first stage, vinylidene chloride undergoes addition with hydrogen
chloride at about 30??C and atmospheric pressure in the presence of a Friedel�Crafts type catalyst. The resulting trichloroethane is then treated with
hydrogen fluoride at about 180??C and 3 MPa (30 atmospheres) in the
presence of antimony pentachloride to give chlorodifluoroethane. Pyrolysis of this product yields vinylidene fluoride. Vinylidene fluoride is a gas, b.p.
-84??C. | [Production Methods]
The first preparation of VF in the early 1900s was by reacting
zinc with 1,1-difluoro-2-bromomethane.
VF was considered to be a high production volume chemical
according to the U.S. Environmental Protection Agency
with annual production exceeding 1million lb in 1990.
In 2001, annual U.S. production was estimated approximately
3.3 million lb. In 1994, VF was produced by one
company each in Japan and the United States. More recently, only one U.S. manufacturer of VF was identified
. Information on European manufacturer is not
available.
The modern production is by the addition of hydrogen
fluoride to acetylene over a mercury- or aluminum-based
catalyst. | [Carcinogenicity]
Vinyl fluoride is reasonably anticipated to be a human carcinogenbased on sufficient evidence of carcinogenicity from studies in experimental animals. | [Environmental Fate]
VF is expected to exist solely as a gas in the ambient atmosphere.
The gas-phase of VF is degraded in the atmosphere by
reaction with photochemically produced hydroxyl radicals. The
half-life for this reaction in air is estimated to be 3 days as
calculated from its rate constant of 5.56 × 10�-12 cm3 molecule sec--1 at 25°�C. VF also reacts with atmospheric ozone, leading
to its atmospheric degradation (estimated half-life of about
16 days). The Henry’s Law constant of VF (0.118 atmm3
mol�1) indicates that VF is expected to volatilize rapidly
from water surfaces. Due to its volatile property, VF is not
persistent in nature and adsorption to sediment is not
considered to be a natural process for VF in water. The half-life
for volatilization from a model river (1-m deep) and a model
pond (2-m deep) are 2 and 23.5 h, respectively. VF is not expected
to bioconcentrate in aquatic organisms as it has a bioconcentration
factor (BCF) of 4.7, whereas a BCF value greater
than 1000 is required for its significant bioaccumulation. As VF
remains as a gas under normal conditions, it readily evaporates
to the atmosphere when released into soil. When dissolved in
an aqueous solution, VF is very mobile in soil. Lack of sufficient
data prevents to predict its biodegradation fate in soils. | [Toxicity evaluation]
VF is readily absorbed after administration by inhalation. Its
metabolism is saturable and dose dependent.
VF is metabolized via the same pathway as for other carcinogenic
vinyl halides like vinyl chloride (VC) and vinyl
bromide. VF is metabolized to DNA-reactive intermediates fluoroethylene oxide and fluoroacetaldehyde via a human
cytochrome P450 2E1 (CYP) dependent pathway. These reactive
metabolites react with DNA bases and form promutagenic
DNA adducts mainly 1, N6-ethenoadenine and N2,3-
-ethenoguanine and cause DNA miscoding by modifying
base-pairing sites. These cyclic etheno adducts lead to misincorporation
of bases upon replication or transcription and
cause critical lesions in VF-induced carcinogenesis. The fluoroacetaldehyde
is metabolized to fluoroacetic acid, a potent
inhibitor of the Krebs cycle. As a consequence, its incorporation
into the citric acid cycle disrupts energy metabolism and leads
to increased production of mitochondrial acetyl coenzyme A
and causes excretion of ketone bodies and free F. So, administration
of VF has been shown to increase acetone exhalation
and F excretion in urine of experimental animals. On the other
hand, fluoroacetaldehyde alkylates the prosthetic heme group
of CYP resulting irreversible inactivation of the enzyme, which
catalyzes the VF metabolism. The alkylate has been identified as
N-(2-oxoethyl) protoporphyrin IX or green porphyrin. |
|
|