62-57-7
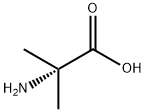 62-57-7 结构式
62-57-7 结构式
基本信息
2-甲基丙氨酸
N-甲基-L-丙氨酸
2-氨基异酷酸
DL-2-甲基丙氨酸
DL-α-氨基异丁酸
2-氨基异酪酸
Α-氨基异丁酸
Α-甲基丙氨酸
2-氨基异丁酸,99%
N-甲基丙氨酸
Β-氨基异丁酸
2-AMINOISOBUTYRIC ACID
2-METHYLALANINE
A-AMINO ISO-BUTYRIC ACID
AIB
ALPHA-AMINOISOBUTYRIC ACID
ALPHA-METHYLALANINE
DL-2-AMINO-ISO-BUTYRIC ACID
H-2-AMINOISOBUTYRIC ACID
H-AIB-OH
H-ALA(ME)-OH
H-ALPHA-METHYLALANINE
H-(ME)ALA-OH
N-ME-ALANINE
N-ME-ALA-OH
N-METHYL-L-ALANINE
N-METHYL-L-AMINOPROPANOIC ACID
RARECHEM EM WB 0051
2-Amino-2-methylpropanoic acid
2-amino-2-methylpropanoicacid
物理化学性质
安全数据
常见问题列表
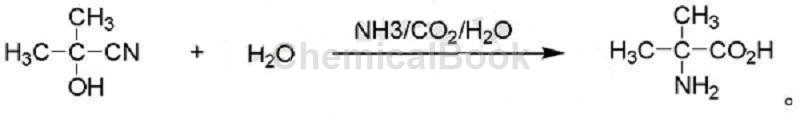
2-甲基丙氨酸的制备方法,包括步骤:丙酮氰醇与碳酸铵在水介质中,加 压加热反应合成2-甲基丙氨酸;所述碳酸铵可用碳酸氢铵代替,或者用氨和二 氧化碳代替;所述反应式如下:
|
Human Endogenous Metabolite
|
NSC 16590 (α-Aminoisobutyric acid , AIB) inhibits the production of endogenous ethylene in the cotyledonary segments of cocklebur ( Xanthium pennsylvanicum Wallr.) seeds most strongly. NSC 16590 at 4 mM inhibits the formation of ethylene by about 50%, although the O 2 uptake of the segments is not affected even at 20 mM. NSC 16590 also inhibits ethylene formation in the stem segments of etiolated pea ( Pisum satiuum L. cv . Alaska) seedlings. Kinetic analysis with cell free extracts from etiolated pea shoots reveals that NSC 16590 competitively inhibits the conversion of ACC into ethylene.
知名试剂公司产品信息
2-Aminoisobutyric acid, 99+%(62-57-7)
2-Aminoisobutyric Acid,>98.0%(T)(62-57-7)
