7081-44-9
中文名称
氯唑西林钠
英文名称
Sodium cloxacillin monohydrate
CAS
7081-44-9
分子式
C19H19ClN3NaO6S
MDL 编号
MFCD00150735
分子量
475.88
MOL 文件
7081-44-9.mol
更新日期
2026/04/20 15:25:41
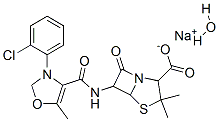 7081-44-9 结构式
7081-44-9 结构式
基本信息
中文别名
邻氯苯甲异噁唑青霉素钠邻氯青霉素
邻氯西林钠
邻氯苯唑西林钠
氯唑西林钠
邻氯苯甲异恶唑青霉素钠
邻氯苯甲异唑青霉素钠
3,3-二甲基-6-[5-甲基-3-(2-氯苯基)-4-异噁唑甲酰氨基]-7-氧代-4-硫杂-1-氮杂双环[3.2.0]庚烷-2-甲酸钠盐
氯唑西林钠
3,3-二甲基-6-[5-甲基-3-(2-氯苯基)-4-异唑甲酰氨基]-7-氧代-4-硫杂-1-氮杂双环[3.2.0]庚烷-2-甲酸钠盐
英文别名
[5-METHYL-3-(O-CHLOROPHENYL)-4-ISOXAZOLYL]PENICILLIN SODIUM SALT MONOHYDRATECLOXACILLIN SODIUM SALT HYDRATE
CLOXACILLIN SODIUM SALT MONOHYDRATE
CLOXACILLIN SODIUM SALT N-HYDRATE
bactopen
brl-1621sodiumsaltmonohydrate
cloxacillinsodiummonohydrate
orbeninsodiumhydrate
prostaphlin-amonohydrate
sodiumcloxacillinmonohydrate
staphobristol-250
thyl-4-isoxazolecarboxamido)-3,3-dimethyl-7-oxo-,sodiumsalt,monohydrate
CLOXACILLIN SODIUM SALT HYDRATE VETRANAL
CLOXACILLIN SODIUM EPC(CRM STANDARD)
CLOXACILLIN SODIUM USP(CRM STANDARD)
CLOXACILLIN SODIUM WHO(CRM STANDARD)
Cloxacillin sodium hydrate
4-Thia-1-azabicyclo3.2.0heptane-2-carboxylic acid, 6-3-(2-chlorophenyl)-5-methyl-4-isoxazolylcarbonylamino-3,3-dimethyl-7-oxo-, monosodium salt, monohydrate, (2S,5R,6R)-
3-(2-Chlorophenyl)-5-methyl-4-isoxazolylpenicillin Sodium
Cloxacillin sodium
所属类别
原料药:青霉素类药物理化学性质
外观性状白色粉末或结晶性粉末。熔点170℃(分解)。易溶于水,溶于乙醇,微溶于氯仿,有引湿性,微臭,味苦。
熔点170℃
沸点689℃
闪点>110°(230°F)
储存条件2-8°C
溶解度H2O中可溶50mg/mL
形态固体
颜色白色至橙色再至绿色
水溶解性溶于水
Merck13,2444
BRN5403885
主要应用clinical testing
InChIKeyMUYUWQFVGNJMPH-UHFFFAOYSA-M
SMILES[Na+].N12C(C(NC(C3=C(C)OCN3C3=CC=CC=C3Cl)=O)C1=O)SC(C)(C)C2C([O-])=O.[H]O[H]
安全数据
警示词危险
危险品标志Xn
危险类别码R36/37/38-R42/43
安全说明S26-S36
WGK Germany2
RTECS号XH8920000
海关编码29411099
存储类别11 - Combustible Solids
危险性类别Eye Irrit. 2
Resp. Sens. 1
Skin Irrit. 2
Skin Sens. 1
STOT SE 3
Resp. Sens. 1
Skin Irrit. 2
Skin Sens. 1
STOT SE 3
应用领域
用途一
该品为半合成抗菌素,与苯唑西林的作用特点和用途均极相似,对耐药金葡菌该品有杀菌作用。口服或肌注吸收较好,血浓度比苯唑西林高2倍,主要用于耐药金葡菌所致感染,如败血症、骨髓炎、皮肤软组织感染、心内膜炎、泌尿系统感染及脑膜炎等疗效较好。制备方法
方法一
邻甲苯胺经重氮化、置换、氯化、水解、肟化等操作。得到邻氯苯甲肟氯,然后与乙酰乙酸乙酯环合、用五氯化磷氯化,得到3-邻氯苯基-5-甲基-4异噁酰氯,最后与6-APA缩合得到邻氯青霉素碱、加异辛酸钠成盐、即为邻氯青霉素钠。氯唑西林钠一水合物价格(试剂级)
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2026/03/03 | 45530 | 邻氯西林钠一水合物 Cloxacillin sodium salt monohydrate | 7081-44-9 | 5g | 2084元 |
| 2026/03/03 | S2564 | 氯唑西林钠一水合物 Cloxacillin Sodium | 7081-44-9 | 50mg | 789.38元 |
| 2025/12/22 | HY-B0466 | 氯唑西林钠一水合物 Cloxacillin sodium monohydrate | 7081-44-9 | 100mg | 300元 |
常见问题列表
生物活性
Cloxacillin Sodium是Cloxacillin的钠盐形式,是一种耐青霉素酶,耐酸,半合成的青霉素。体外研究
Cloxacillin is an antibiotic useful for the study of a number of bacterial infections.
Cell Viability Assay
| Cell Line: | Strains M12 and M60. |
| Concentration: | 0.5 μg/mL. |
| Incubation Time: | 4-24 h. |
| Result: | Significantly reduced the bacterial numbers. |
体内研究
Cloxacillin sodium (50 mg/kg, Subcutaneously) results a significant antibiotic efficacy against S. aureus .
| Animal Model: | Mice. |
| Dosage: | 10 mg/kg ( Pharmacological Analysis ). |
| Administration: | Given subcutaneously. |
| Result: | Reached a maximal concentration in plasma of 8.4 μg/mL at 10 min and had a half-life of approximately 15 min. |
| Animal Model: | Mice injected with approximately 2 × 10 6 CFU of bacteria in 0.1 mL saline were aseptically injected into the right thigh muscle. |
| Dosage: | 0–500 mg/kg. |
| Administration: | Subcutaneously in the nuchal region at 1 h after infection. |
| Result: | Resulted in a significant decrease in the number of viable S. aureus measured 18 h thereafter. |

