3-Chlorpropionylchlorid Chemische Eigenschaften,Einsatz,Produktion Methoden
R-Sätze Betriebsanweisung:
R14:Reagiert heftig mit Wasser.
R22:Gesundheitsschädlich beim Verschlucken.
R26:Sehr giftig beim Einatmen.
R34:Verursacht Verätzungen.
R35:Verursacht schwere Verätzungen.
R10:Entzündlich.
S-Sätze Betriebsanweisung:
S23:Gas/Rauch/Dampf/Aerosol nicht einatmen(geeignete Bezeichnung(en) vom Hersteller anzugeben).
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36/37/39:Bei der Arbeit geeignete Schutzkleidung,Schutzhandschuhe und Schutzbrille/Gesichtsschutz tragen.
S45:Bei Unfall oder Unwohlsein sofort Arzt zuziehen (wenn möglich, dieses Etikett vorzeigen).
S38:Bei unzureichender Belüftung Atemschutzgerät anlegen.
S16:Von Zündquellen fernhalten - Nicht rauchen.
S8:Behälter trocken halten.
Beschreibung
3-Chloropropionyl chloride is an important bifunctional reagent. It is capable of acylation and possesses a 2-chloro-ethyl fragment (CH2CH2Cl), which can be subjected to nucleophilic substitution and serves as a masked vinyl group. It can be used as a starting material in many reactions to construct a variety of (hetero)cyclic compounds.
Chemische Eigenschaften
Clear colorless to dark brown liquid. Soluble in ethanol, ether and chloroform. Slightly soluble in water.
Verwenden
3-Chloropropionyl chloride is a reagent used in the synthesis of Beclamide (B119400), which is a chlorinated benzylpropanamide used as an anticonvulsant drug. It is used in the treatment of tonic-clonic seizyres and has sedative properties.
synthetische
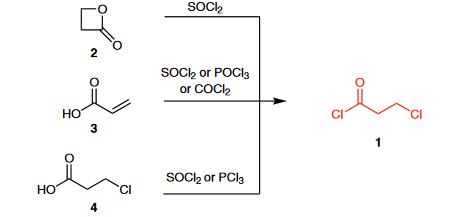
3-Chloropropionyl chloride (1) is commercially available and can be prepared from β-propiolactone (2) and thionyl chloride.1 Other standard methods available for the preparation of acyl chlorides can also be applied: the reaction of acrylic acid (3) or 3-chloropropionic acid (4) with thionyl chloride, phosphoryl chloride, phosgene, or phosphorus trichloride.
Reaktionen
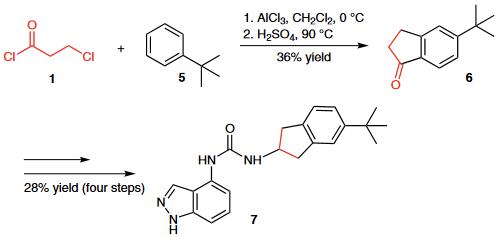
(1) The Friedel¨CCrafts acylation of tert-butylbenzene (5) with 3-chloropropionyl chloride (1) followed by cyclization provid- ed indanone 6, which was further transformed into urea derivative 7, a potent TRPV1 antagonist.
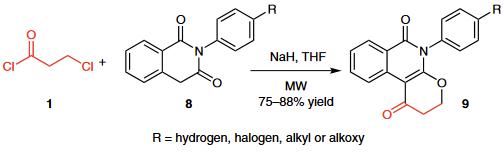
(2) A novel high-yielding one-pot microwave-assisted synthesis of condensed 5-substituted pyranoisoquinoline-1,6-diones 9 from 2-substituted isoquinoline-1,3-diones 8 and 3-chloropropionyl chloride (1) was reported.
(3) Acylation of 2-aminophenol (12) with 3-chloropropionyl chloride (1), followed by cyclization in the presence of polyphosphoric acid (PPA), gave benzoxazole 13, which was further reacted with 4-chlorophenyl-1-piperazine to yield the target benzo[d]oxazole analogue 14, a selective dopamine D4 receptor ligand.
3-Chlorpropionylchlorid Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

