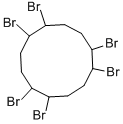
Hexabromocyclododecane
- CAS No.
- 25637-99-4
- Chemical Name:
- Hexabromocyclododecane
- Synonyms
- HBCD;Cyclododecane, hexabromo-;BETA-HEXABROMOCYCLODODECANE;GAMMA-HEXABROMOCYCLODODECANE;HBCD HT;hexabromo-cyclododecan;HEXABROMOCYCLODODECANE;HEXABROMOCYCLODODECANE HT;1,3,5,7,9,11-hexabromocyclododecane;Hexabromcyclododecan, 1,2,5,6,9,10-
- CBNumber:
- CB4363332
- Molecular Formula:
- C12H18Br6
- Molecular Weight:
- 641.7
- MDL Number:
- MFCD00003716
- MOL File:
- 25637-99-4.mol
- MSDS File:
- SDS
| Melting point | 188-191 °C |
|---|---|
| Density | 1.05 g/cm3 |
| vapor pressure | 0Pa at 21℃ |
| solubility | Benzene (Slightly), Ethyl Acetate (Slightly) |
| form | Solid |
| color | White to Off-White |
| Water Solubility | 65.6μg/L at 20℃ |
| LogP | 5.625 at 25℃ |
| CAS DataBase Reference | 25637-99-4(CAS DataBase Reference) |
| EWG's Food Scores | 1 |
| FDA UNII | 5I9835JO3M |
| EPA Substance Registry System | Hexabromocyclododecane (25637-99-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|---|---|
| Signal word | Warning |
| Hazard statements | H361-H362 |
| Precautionary statements | P201-P260-P263-P264-P270-P308+P313-P201-P202-P281-P308+P313-P405-P501 |
| Safety Statements | 22-24/25 |
| RIDADR | 3077 |
| WGK Germany | 1 |
| F | 3-9 |
| HazardClass | 9 |
| PackingGroup | III |
Hexabromocyclododecane price More Price(3)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TRC | H290720 | Hexabromocyclododecane(MixtureofDiastereomers) | 25637-99-4 | 5g | $70 | 2021-12-16 | Buy |
| TRC | H290720 | Hexabromocyclododecane(MixtureofDiastereomers) | 25637-99-4 | 25g | $120 | 2021-12-16 | Buy |
| TRC | H290720 | Hexabromocyclododecane(MixtureofDiastereomers) | 25637-99-4 | 50g | $215 | 2021-12-16 | Buy |
Hexabromocyclododecane Chemical Properties,Uses,Production
Chemical Properties
Hexabromocyclododecane (HBCD) is a white odorless nonvolatile solid compound which is used as an additive flame retardant on its own, or in combination with other flame retardants. It is used as a flame retardant mainly in building insulation composed of extruded or expanded polystyrene foam. A minor use is in flame retardant back-coats of some upholstery textiles.
Uses
The commercially available brominated flame retardant hexabromocyclododecane (HBCD) is lipophilic, has a high affinity to particulate matter and low water solubility. It is used as a flame retardant additive, with the intent of delaying ignition and slowing subsequent fire growth during the service life of vehicles, buildings or articles, as well as while materials are stored. The main uses of HBCD are in flame-retarded expanded (EPS) and extruded (XPS) polystyrene foam insulation, with smaller scale use in textile applications and electric and electronic appliances (high impact polystyrene/HIPS). In textiles HBCD is used in back-coatings for upholstery and other interior textiles, including automotive applications.
Preparation
The aliphatic flame retardant, hexabromocyclododecane (HBCD), is produced by the addition reaction of bromine to trans,trans,cis-1,5,9-cyclododecatriene, the cyclic trimer of butadiene. The HBCD that is obtained from this reaction consists of a mixture of stereoisomers of 1,2,5,6,9,10-hexabromocyclo-dodecane [3194-55-6] ranging from oils to an isomer with a 205–208℃ melting point. Commercially available HBCD contains a mixture of solid isomers and has a melting point range of 170–180 ℃ (185–195℃ ).
Addition of bromine to the cyclic triene in nonpolar solvents results in substantial amounts of free-radical substitution reactions which lead to decreased yields and lower quality product. Use of polar solvents, such as ethanol, reduces the substitution reaction but can give rise to vicinal bromoethoxy by products. Mixed solvent systems, such as benzene–tert-butyl alcohol or perchloroethylene-ethanol,can be employed to improve product purities and yields.
Another technique to reduce the formation of undesired byproducts is to add the bromine and cyclododecatriene simultaneously and sep- arately to ethanol. This process is carried out by separately feeding bromine and cyclo- dodecatriene in a molar ratio of 3 : 1 to 96 % ethanolinastirredreactor.The reaction temperature is kept at 20-30℃ and a 1-5% excess of bromine is maintained during the 2.5–3.0h addition. The precipitated product is recovered by filtration or centrifugation, and the mother liquor is recovered and reused in subsequent reactions after adding makeup ethanol. The total yield of dried HBCD from nine separate batches using recycled mother liquor is 92%.
General Description
HexabroMocyclododecane is White powder. Insoluble in water.
Air & Water Reactions
HexabroMocyclododecane is Insoluble in water.
Reactivity Profile
Halogenated aliphatic compounds, such as Hexabromocyclododecane, are moderately or very reactive. Reactivity generally decreases with increased degree of substitution of halogen for hydrogen atoms. Materials in this group may be incompatible with strong oxidizing and reducing agents. Also, they may be incompatible with many amines, nitrides, azo/diazo compounds, alkali metals, and epoxides. Hexabromocyclododecane can react vigorously with oxidizing materials.
Fire Hazard
Flash point data for Hexabromocyclododecane are not available. Hexabromocyclododecane is probably combustible.
Hexabromocyclododecane Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 7845 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 15928 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 32480 | 60 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 | sales@chemdad.com | China | 39916 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-81138252 +86-18789408387 | 1057@dideu.com | China | 3537 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 | sales@tnjchem.com | China | 34572 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 | 1026@dideu.com | China | 9320 | 58 |
| Hebei Guanlang Biotechnology Co,.LTD | +8619930503252 | daisy@crovellbio.com | China | 5964 | 58 |
View Lastest Price from Hexabromocyclododecane manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2023-12-26 | Hexabromocyclododecane
25637-99-4
|
US $100.00-1.00 / KG | 1KG | 99% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd | |
 |
2023-04-07 | Hexabromocyclododecane
25637-99-4
|
US $30.00-10.00 / kg | 10kg | 99% | 100000kg | Hebei Guanlang Biotechnology Co,.LTD | |
 |
2020-01-07 | HexabroMocyclododecane
25637-99-4
|
US $1.00 / KG | 1KG | 99% | 20T | Shaanxi Dideu Medichem Co. Ltd |
-

- Hexabromocyclododecane
25637-99-4
- US $100.00-1.00 / KG
- 99%
- Henan Fengda Chemical Co., Ltd
-

- Hexabromocyclododecane
25637-99-4
- US $30.00-10.00 / kg
- 99%
- Hebei Guanlang Biotechnology Co,.LTD
-

- HexabroMocyclododecane
25637-99-4
- US $1.00 / KG
- 99%
- Shaanxi Dideu Medichem Co. Ltd