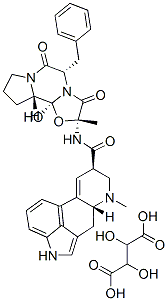
ERGOTAMINE TARTRATE
- CAS No.
- 379-79-3
- Chemical Name:
- ERGOTAMINE TARTRATE
- Synonyms
- ergomar;ergostat;etin;ergam;ergate;exmigra;lingran;secagyn;secupan;femergin
- CBNumber:
- CB9279489
- Molecular Formula:
- C70H76N10O16
- Molecular Weight:
- 1313.41
- MDL Number:
- MFCD08277639
- MOL File:
- 379-79-3.mol
| Melting point | ~195 °C (dec.)(lit.) |
|---|---|
| alpha | D25 -125 to -155° (c = 0.4 in chloroform) |
| storage temp. | 2-8°C |
| Merck | 13,3696 |
| BRN | 6173437 |
| CAS DataBase Reference | 379-79-3(CAS DataBase Reference) |
| EWG's Food Scores | 2 |
| FDA UNII | MRU5XH3B48 |
| Proposition 65 List | Ergotamine Tartrate |
| EPA Substance Registry System | Ergotamine tartrate (379-79-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|---|---|
| Signal word | Danger |
| Hazard statements | H301+H311+H331-H361 |
| Precautionary statements | P261-P280-P301+P330+P331+P310-P302+P352+P312-P304+P340+P311-P403+P233 |
| Hazard Codes | T |
| Risk Statements | 23/24/25-62-63 |
| Safety Statements | 36/37-45 |
| RIDADR | UN 1544 6.1/PG 1 |
| WGK Germany | 3 |
| RTECS | KE8225000 |
| F | 3-8-10 |
| HazardClass | 6.1(b) |
| PackingGroup | III |
| HS Code | 29396200 |
ERGOTAMINE TARTRATE Chemical Properties,Uses,Production
Description
Ergotamine and ergometrine belong to two kinds of ergot alkaloids. Ergot (Claviceps
purpurea, 麦角) is the sclerotium of ergot fungus that grows in the rye and related
cereal and forage plants. The ergot is grown in the northeast and north China and
Xinjiang, Jiangsu, Zhejiang, Sichuan, and other places. At present, the method of
growth of ergot is inoculation cultivation.
Traditional Chinese medicine believes that ergot is tasteless, tepid, toxic, liver
meridian, and kidney meridian. The traditional usage of ergot is oral administration
of extractum, 2–4 ml each time, daily amount of 6–8 ml. Its dosage forms also contain
infusion, pills, injection, etc. Pharmacology of Chinese Medicine records ergot
can be used for uterine contraction, uterine bleeding, and postpartum hemorrhage
after delivery, incomplete recovery of uterus, retrogression, etc. The Plateau
Chinese herbal medicine treatment manual records that ergot can cure migraine.
Chemical Properties
White Solid
Chemical Properties
Ergotamine tartrate is a crystalline solid.
Physical properties
Appearance: White or slightly yellow fine crystalline powder with slight hygroscopicity. Solubility: Slightly soluble in water and ethanol, insoluble in ether and chloroform.
History
As a kind of traditional Chinese medicine, ergot was originally used for hemostasis and involution of uterus by stimulating the uterine muscle powerfully and persistently. After the modernization the traditional Chinese medicine was putting forward, by making several kinds of wild ergot into extractum, we found that all of them can promote the contraction of the rabbit uterus in vivo and in vitro. In isolated rabbit uterus, the effect of ergot on postpartum uterus and pregnant uterus was stronger than nonpregnant uterus. Among a variety of active ingredients in the ergot, ergonovine has the strongest effect on uterine, followed by ergotamine. Seventeen of the 131 strains of ergot derived from the host plants of our country are able to produce ergot alkaloids in wheat culture medium. Among the strains, Claviceps microcephala Ce-3 produces the most ergonovine. Therefore, it became the main bacteria used in the process of industrialization. The study suggested that ergotamine or ergot can block the a-adrenergic receptor, so that the pressor effect of adrenaline was reversed. They do not prevent the release of sympathetic mediators. It showed that the effect of ergotamine is similar to norepinephrine on the hypogastric nerve of guinea pig in vitro. Low dose of ergot can stimulate the brain (slow heart rate, breathing, convulsions, etc.) whereas high dose can cause deaths due to the delayed paralysis. In addition, it can enhance the sedative and hypnotic effects of barbiturates, morphine, and methadone.
Uses
diuretic, inhibits Na/K/2Cl carrier system
Uses
Antimigraine.
Indications
Ergometrine maleate is used in the form of extract tumor injection. As a uterotonic,
ergometrine maleate is mainly used for the prevention and treatment of uterine
bleeding caused by weak or poor uterine contraction after delivery or abortion, as
well as the incomplete recovery of postpartum uterus to accelerate the recovery of
uterus.
Ergotamine tartrate is used for the treatment of migraine in the form of tablet and
parenteral solution. It also can be used for other neurogenic headache as well.
brand name
Ergomar (Harvest);Ergostat (Parke-Davis); Wigrettes (Organon).
General Description
Powder or liquid forms available as drugs. Used in obstetrics and in the treatment of migraine headaches.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
ERGOTAMINE TARTRATE is sensitive to prolonged exposure to heat and light. . When heated to decomposition, ERGOTAMINE TARTRATE emits toxic fumes of nitrogen oxides. Protect from light and heat [EPA, 1998].
Health Hazard
High oral toxicity and a convulsant in humans.People with liver damage are at a greater risk.
Fire Hazard
When heated to decomposition, ERGOTAMINE TARTRATE emits toxic fumes of nitrogen oxides. Protect from light and heat
Pharmacology
1. The effect of uterus excitation
The effect of ergot on the uterine muscle is similar to the posterior pituitary
preparation, which directly acts on the uterine muscle with more powerful and
lasting effects. The pregnant uterus is more sensitive to ergot. Low dose of ergot
has significant effects on the uterus of labor and new postpartum with severely
tetanic contraction. The extracts from several kinds of wild ergot can promote
the contraction and increase the tension of uterus in rabbit in vivo and in vitro
. Among a variety of active ingredients in the ergot, ergonovine has the strongest
effect on uterus, followed by ergotamine.
2. The effects on cardiovascular
In the isolated vascular experiment, ergotamine can cause the contraction of
peripheral vascular smooth muscle and hypertension due to vasoconstriction and
boost and eventually produce compensatory bradycardia. Ergot has less effect on
the heart than blood vessels, but when the heart rate drops to a certain extent, it
can enhance the cardiac contractility. High dose of ergotamine can damage vascular
endothelial cells (the mechanism is unknown), leading to gangrene.
Ergotine has the strongest effect on blood vessels, and ergometrine hardly affects
blood pressure and damages to vascular endothelial cells. Ergotine can cause
vasoconstriction and decrease the arterial pulse amplitude, which may be related
to the therapeutic effect on migraine.
3. The effects on the nervous system
Ergotamine or ergot can block the a-adrenergic receptor and reverse the pressor
effect of adrenaline without preventing the release of sympathetic neurotransmitters.
Low dose of ergot stimulates the brain (slow down heart rate, breathing,
convulsions, etc.), whereas high dose causes deaths due to the delayed paralysis.
Ergotamine enhances the sedative and hypnotic effects of barbiturates, morphine,
and methadone.
Clinical Use
Ergonovine maleate is mainly used for the prevention and treatment of the weak
uterine contraction or poor contraction caused by uterine bleeding after delivery or
abortion. It is also used for incomplete recovery of postpartum uterus to accelerate
the recovery of the uterus.
Ergotamine tartrate is mainly used for the treatment of migraine by relieving the
symptoms. However, it is only suitable for short-term usage of headache attacks,
normally does not prevent migraine. The combination of ergotamine tartrate and
caffeine is better than single oral administration with the mild side effects. It also
can be used for other neurogenic headaches.
Safety Profile
Poison by ingestion, intravenous, and subcutaneous routes. An experimental teratogen. Human systemic effects by ingestion: hallucinations, dstorted perceptions, convulsions, nausea or vomiting, blood pressure elevation. Experimental reproductive effects. Human mutation data reported. Used in production of drugs of abuse. When heated to decomposition it emits toxic fumes of NOx. See also ERGOT.
Potential Exposure
(Toxicity evaluation); Suspected reprotoxic hazard, Suspected of causing genetic defects. The major uses of the ergot alkaloids fall into two categories: applications in obstetrics and treatment of migraine headaches. May be used in the production of “street” drugs
First aid
If this chemical gets into the eyes, remove any contact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin,remove contaminated clothing and wash immediately withsoap and water. Speed in removing material from skin is ofextreme importance. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure, beginrescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heartaction has stopped. Transfer promptly to a medical facility.When this chemical has been swallowed, get medical attention. Give large quantities of water and induce vomiting. Donot make an unconscious person vomit. Keep victim quietand maintain normal body temperature. Effects may bedelayed; keep victim under observation.
storage
Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with thischemical you should be trained on its proper handling andstorage. Store in tightly closed containers in a cool, well-ventilated area.
Shipping
UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required
Purification Methods
It crystallises from MeOH. [Beilstein 25 III/IV 964.]
Incompatibilities
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides, light, and heat