Barium chloride
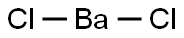
- CAS No.
- 10361-37-2
- Chemical Name:
- Barium chloride
- Synonyms
- BaCl2;Barium Chloride Solution, 10% w/w, neutralized;ba0108e;Ba 0108E;sba0108e;SBA 0108E;NCI-C61074;Dichlorobarium;BARIUM CHLORIDE;bariumdichloride
- CBNumber:
- CB8398703
- Molecular Formula:
- BaCl2
- Molecular Weight:
- 208.23
- MOL File:
- 10361-37-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/2/18 17:28:16
| Melting point | 963 °C (lit.) |
|---|---|
| Boiling point | 1560°C |
| Density | 3.856 g/mL at 25 °C (lit.) |
| storage temp. | 2-8°C |
| solubility | H2O: soluble |
| form | beads |
| color | White |
| Specific Gravity | 3.9 |
| PH | 5-8 (50g/l, H2O, 20℃) |
| Water Solubility | Soluble in water and methanol. Insoluble in acids, ethanol, acetone and ethyl acetate. Slightly soluble in nitric acid and hydrochloric acid. |
| Sensitive | Hygroscopic |
| Merck | 14,971 |
| Exposure limits |
ACGIH: TWA 0.5 mg/m3 NIOSH: IDLH 50 mg/m3; TWA 0.5 mg/m3 |
| Dielectric constant | 9.4(0.0℃) |
| Stability | Stable. |
| CAS DataBase Reference | 10361-37-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Barium dichloride(10361-37-2) |
| EPA Substance Registry System | Barium chloride (10361-37-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H319-H332 | |||||||||
| Precautionary statements | P261-P264-P270-P301+P310-P304+P340+P312-P305+P351+P338 | |||||||||
| Hazard Codes | T,Xi,Xn | |||||||||
| Risk Statements | 22-25-20-36/37/38-36/38-36 | |||||||||
| Safety Statements | 45-36-26-36/37/39 | |||||||||
| RIDADR | UN 3264 8/PG 3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | CQ8750000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2827 39 85 | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| Toxicity | LD50 orally in Rabbit: 118 mg/kg | |||||||||
| NFPA 704 |
|
Barium chloride price More Price(29)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 449644 | Barium chloride AnhydroBeads?, ?10?mesh, 99.999% trace metals basis | 10361-37-2 | 5G | ₹5434.15 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 449652 | Barium chloride AnhydroBeads?, ?10?mesh, 99.95% trace metals basis | 10361-37-2 | 10G | ₹7913.08 | 2022-06-14 | Buy |
| Sigma-Aldrich | 342920 | Barium chloride 99.9% trace metals basis | 10361-37-2 | 10G | ₹5152.7 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 342920 | Barium chloride 99.9% trace metals basis | 10361-37-2 | 10G | ₹5152.7 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 449644 | Barium chloride AnhydroBeads?, ?10?mesh, 99.999% trace metals basis | 10361-37-2 | 25G | ₹19084.48 | 2022-06-14 | Buy |
Barium chloride Chemical Properties,Uses,Production
Description
Barium dichloride is a white solid, odorless, hygroscopic chemical substance. Barium dichloride is used in the manufacture of pigments, in the manufacture of other barium salts and in fireworks to give a bright green color. It is one of the most common watersoluble salts of barium. Like other barium salts, it is toxic and imparts a yellow-green coloration to a flame. Barium chloride has wide application in the laboratory.
Chemical Properties
Barium chloride,BaCI2, is a colorless toxic salt with a melting point of 963°C. It is soluble in water. Barium chloride is used in metal surface treatment and as a rat poison.
Physical properties
Barium chloride has the formula, BaCl2 and is an ionic chemical compound. It is one of the most important water-soluble salts of barium-containing compounds. Like other barium salts, it is toxic and imparts a yellow-green coloration to a flame. It is also hygroscopic. Barium chloride was the by-product of the discovery of radium by Madame Curie (1898).When refining radium, the final separation resulted in barium chloride and radium chloride. BaCl2 crystallizes in both the cubic “fluorite” and “lead chloride” crystal structures, both of which accommodate the preference of the large Ba2+ ion for coordination numbers greater than six.
Preparation
Barium chloride can be prepared from barium
hydroxide or barium carbonate, the latter being found
naturally as the mineral “Witherite”. These basic salts
react to give hydrated barium chloride. On an industrial
scale, it is prepared via a two-step process from the
mineral “Baryte”:
BaSO4+4C→BaS+4CO (gas)
This first step requires high temperatures. The second
step requires fusion of the reactants:
BaS+ CaCl2→BaCl2+CaS
The BaCl2 is then be leached out from the mixture
with water. From water solutions of barium chloride,
the dihydrate can then be crystallized as white crystals,
BaCl2·2H2O, which are colorless, translucent rhomboidal
tablets or lamellae. The dihydrate is stable in
the air at room temperature, but loses one-half of its
water above 55°C(131F), and becomes anhydrous at
121°C (250 F).
General Description
Any of a variety of substances that contain barium. Most are whitish colored crystalline solids. They tend to be soluble in water and denser than water. They may be toxic by inhalation or possibly skin absorption. They are often used to make other chemicals.
Air & Water Reactions
Water soluble.
Reactivity Profile
BARIUM CHLORIDE may react violently with BrF3 and 2-furan percarboxylic acid in its anhydrous form.
Hazard
Ingestion of 0.8 g may be fatal.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Industrial uses
Barium chloride (BaCl2·2H2O) is a colorless, white powder highly soluble in water (25% at 10 °C). It is quite a toxic reagent. Barium chloride is used during borite flotation as an activator. Barium chloride also has a depressing effect on fluorite and cassiterite.
Safety Profile
A poison by ingestion, subcutaneous, intravenous, and intraperitoneal routes. Inhalation absorption of barium chloride equals 60-80%; oral absorption equals 10-30%. Experimental reproductive effects. Mutation data reported. See also BARIUM COMPOUNDS (soluble). When heated to decomposition it emits toxic fumes of Cl-.
Barium chloride Preparation Products And Raw materials
Raw materials
Preparation Products
1of6
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| ALLIPO CHEMICALS | +91-8238998187 +91-8238998187 | Gujarat, India | 32 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3758 | 58 | Inquiry |
| Otto Chemie Pvt Ltd | +91-2222070099 +91-2266382599 | Maharashtra, India | 429 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| A.B.ENTERPRISES | +91-9223381464 +91-9223381464 | Maharashtra, India | 78 | 58 | Inquiry |
| Athiappa Chemicals | 91-44-26260119 | Kerela, India | 4 | 58 | Inquiry |
| Divjyot Chemicals Private Limited | +91-8048603207 +91-9849191943 | Telangana, India | 11 | 58 | Inquiry |
| Shiv Organo Synthesis | +91-9825915701 +91-9825740398 | Gujarat, India | 7 | 58 | Inquiry |
| Ultimate Chem (India) Pvt., Ltd. | 91-22-29201582 | Maharashtra, India | 117 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| ALLIPO CHEMICALS | 58 |
| JSK Chemicals | 58 |
| Otto Chemie Pvt Ltd | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| A.B.ENTERPRISES | 58 |
| Athiappa Chemicals | 58 |
| Divjyot Chemicals Private Limited | 58 |
| Shiv Organo Synthesis | 58 |
| Ultimate Chem (India) Pvt., Ltd. | 58 |
Related articles
- Why does Barium Chloride burn green?
- When burned, barium chloride produces a bright green color in flames. It is also known to be hygroscopic, meaning it absorbs m....
- Jan 25,2024
- Property, preparation and application of barium chloride
- Barium dichloride is a white solid, odorless, hygroscopic chemical substance. Barium dichloride is used in the manufacture of ....
- Jul 7,2022





