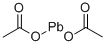아세트산 납
|
|
아세트산 납 속성
- 녹는점
- 75 °C (dec.)(lit.)
- 끓는 점
- decomposes at >280℃ [KIR78]
- 밀도
- 3.3 g/cm3
- 증기압
- 15.7hPa at 25℃
- 저장 조건
- 2-8°C
- 용해도
- DMSO(약간 용해됨), 메탄올(약간 용해됨)
- 물리적 상태
- 액체
- 색상
- 무색의
- 수용성
- g/100g H2O: 19.7(0°C), 55.2(25°C); 평형 고체상, Pb(CH3COO)2·3H2O [KRU93]; g/100mL H2O: 44.3(20°C), 221(50°C) [KIR78]
- 최대 파장(λmax)
- 260nm(H2O)(lit.)
- Merck
- 14,5397
- Solubility Product Constant (Ksp)
- pKsp: 2.75
- Dielectric constant
- 2.5(0.0℃)
- LogP
- -0.17
- CAS 데이터베이스
- 301-04-2(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,N | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 61-33-48/22-50/53-62 | ||
| 안전지침서 | 53-45-60-61 | ||
| 유엔번호(UN No.) | UN 1616 6.1/PG 3 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | OF8050000 | ||
| HS 번호 | 2915.29.5000 | ||
| 위험 등급 | 6.1(b) | ||
| 포장분류 | III | ||
| 유해 물질 데이터 | 301-04-2(Hazardous Substances Data) | ||
| 독성 | LD50 i.p. in rats: 15 mg Pb/100g (Bradley, Fredrick) | ||
| 기존화학 물질 | KE-21888 | ||
| 유해화학물질 필터링 | 97-1-9 | ||
| 중점관리물질 필터링 | 별표1-78 | ||
| 암, 돌연변이성물질 필터링 | 355 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 사산화납(lead tetraoxide), 황산납(lead sulfate), 염기성탄산납(basic lead carbonate)을 제외한 납화합물(Lead compounds) 및 이를 25% 이상 함유한 혼합물. 다만, 초산납(lead acetate), 알킬화납(lead alkyls), 아지드화납(lead azide), 이초산납(lead di(acetate)), 메탄술폰산납(lead(Ⅲ) methansulfonate), 인산납(lead phosphate(3:2)), 스티핀산납(lead styphate)의 경우는 이를 0.5% 이상 함유한 혼합물 |
아세트산 납 C화학적 특성, 용도, 생산
개요
Lead acetate is stable under ordinary conditions of use and storage. Lead acetate is incompatible with bromates, phenol, chloral hydrate, sulphides, hydrogen peroxide, resorcinol, salicylic acid, sulphites, vegetable infusions, alkalis, tannin, phosphates, citrates, chlorides, carbonates, tartrates, and acids. Lead (II) acetate, as well as white lead, has been used in cosmetics throughout history, though this practice has ceased in Western countries. It is still used in men’s hair colouring. Lead (II) acetate paper is used to detect the poisonous gas hydrogen sulphide. The gas reacts with lead (II) acetate on the moistened test paper to form a grey precipitate of lead (II) sulphide.화학적 성질
Lead acetate is a white, flaky crystalline substance with a slight odor of acetic acid. Commercial grades may be powdered granules, or brown or gray lumps. Diacetate: Powder.용도
2 – 1 - SweetenerLike other lead (II) salts, lead (II) acetate has a sweet taste, which has led to its use as a sugar substitute throughout history. The ancient Romans, who had few sweeteners besides honey, would boil must (grape juice) in lead pots to produce a reduced sugar syrup called defrutum, concentrated again into sapa. This syrup was used to sweeten wine and to sweeten and preserve fruit. It is possible that lead(II) acetate or other lead compounds leaching into the syrup might have caused lead poisoning in anyone consuming it . Lead acetate is no longer used in the production of sweeteners in most of the world because of its recognized toxicity. Modern chemistry can easily detect it, which has all but stopped the illegal use that continued decades after legal use as a sweetener was banned .
2 – 1 - Sweetener2 – 1 – 1 - Resultant deaths
Pope Clement II died in October 1047. A toxicologic examination of his remains conducted in the mid – 20 th century confirmed centuries-old rumors that he had been poisoned with lead sugar.It is not clear if he was assassinated.
In 1787 painter Albert Christoph Dies swallowed, by accident, approximately 21 g of lead acetate. His recovery from this poison was slow and incomplete. He lived with illnesses until his death in 1822 .
Although the use of lead (II) acetate as a sweetener was already illegal at that time, composer Ludwig van Beethoven may have died of lead poisoning caused by wines adulterated with lead acetate.
Mary Seacole applied lead (II) acetate, among other remedies, against an epidemic of cholera in Panama.
생산 방법
Lead acetate is made by dissolving lead monoxide (litharge) or lead carbonate in strong acetic acid. Several types of basic salts are formed when lead acetates are prepared from lead monoxide in dilute acetic acid or at high pH. The basic salts of lead acetate are white crystalline compounds, which are highly soluble in water and dissolve in ethyl alcohol.Lead acetate can be made by boiling elemental lead in acetic acid and hydrogen peroxide.
정의
ChEBI: A lead coordination entity in which a central lead(2+) atom is coordinated to two acetate ions.잠재적 노출
Lead acetate is used as a color additive in hair dyes; as a mordant in cotton dyes, in the lead coating of metals; as a drier in paints; varnishes and pigment inks; and in medicinals, such as astringents. Incompatibilities: A strong reducing agent. Reacts violently with strong oxidizers, bromates, strong acids; chemically active metals; phosphates, carbonates, phenols. Contact with strong acids forms acetic acid. Incompatible with strong bases: ammonia, amines, cresols, isocyanates, alkylene oxides; epichlorohydrin, sulfites, resorcinol, salicylic acid, and chloral hydrat운송 방법
UN1616 Lead acetate, Hazard Class: 6.1; Labels: 6.1-Poisonous materialsPurification Methods
Crystallise it twice from anhydrous acetic acid and dry it under vacuum for 24hours at 100o. The solubility in H2O is 63% (at ~20o) and 200% (at boiling point). [Beilstein 2 IV 118.]비 호환성
A strong reducing agent. Reacts violently with strong oxidizers, bromates, strong acids; chemically active metals; phosphates, carbonates, phenols. Contact with strong acids forms acetic acid. Incompatible with strong bases: ammonia, amines, cresols, isocyanates, alkylene oxides; epichlorohydrin, sulfites, resorcinol, salicylic acid, and chloral hydrate폐기물 처리
Convert to nitrate using nitric acid; evaporate, then saturate with H2S; wash and dry the sulfide and ship to the supplier. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal주의 사항
Lead (II) acetate, as with any other lead salts, causes lead poisoning.아세트산 납 준비 용품 및 원자재
원자재
준비 용품
아세트산 납 공급 업체
글로벌( 184)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shenyang Simchoice Chemical Co.,Ltd | +86-024-23769576 +86-15040101888 |
sales@simchoicechem.com | China | 255 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +8618949832763 |
info@tnjchem.com | China | 2989 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8820 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
| Richest Group Ltd | 18017061086 |
oled@richest-group.com | CHINA | 5601 | 58 |
| Antai Fine Chemical Technology Co.,Limited | 18503026267 |
info@antaichem.com | CHINA | 9641 | 58 |
| Xingtai Haoxun Import and Export Trade Co., Ltd. | +8617733976525 |
lisa@xthaoxun.com | CHINA | 258 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 |
sales@tnjchem.com | China | 34571 | 58 |