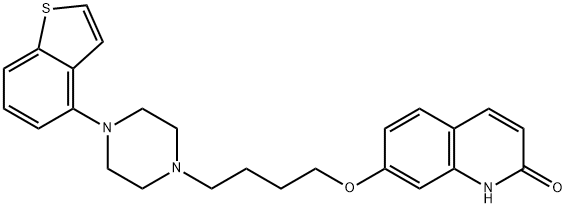
브렉스피프라졸
|
|
브렉스피프라졸 속성
- 녹는점
- 179 - 181oC
- 끓는 점
- 675.2±55.0 °C(Predicted)
- 밀도
- 1.245±0.06 g/cm3(Predicted)
- 저장 조건
- Refrigerator
- 용해도
- 디클로로메탄 (약간 용해됨), 클로로포름 (약간 용해됨), 메탄올 (약간 용해됨)
- 산도 계수 (pKa)
- 11.22±0.70(Predicted)
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 색상
- 흰색에서 황백색까지
- InChI
- InChI=1S/C25H27N3O2S/c29-25-9-7-19-6-8-20(18-22(19)26-25)30-16-2-1-11-27-12-14-28(15-13-27)23-4-3-5-24-21(23)10-17-31-24/h3-10,17-18H,1-2,11-16H2,(H,26,29)
- InChIKey
- ZKIAIYBUSXZPLP-UHFFFAOYSA-N
- SMILES
- N1C2=C(C=CC(OCCCCN3CCN(C4=C5C=CSC5=CC=C4)CC3)=C2)C=CC1=O
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
브렉스피프라졸 C화학적 특성, 용도, 생산
개요
Brexpiprazole is a novel antipsychotic drug which serves as a serotonin ® dopamine activity modulator and has demonstrated efficacy as an adjunctive treatment in patients with major depressive disorder (MDD). The drug exhibits a unique pharmacological profile, acting as a partial agonist of serotonin 5-HT1A and dopamine D2 receptors and as a full antagonist of 5-HT2A and noradrenaline α1B/2C receptors, with similar subnanomolar binding affinity. The drug, which was developed by Otsuka and Lundbeck, was approved in 2015 by the FDA for the treatment of schizophrenia and as an adjunctive treatment for depression. Brexpiprazole is widely considered to be a successor to Otsuka’s antipsychotic drug aripiprazole (trade name Abilify) whose patent expired in August 2014.용도
Brexpiprazole is a kind of atypical antipsychotic. It is a dopamine D2 receptor partial agonist. As a novel serotonin-dopamine activity modulator, it can be used for the treatment of schizophrenia, and for the adjunctive treatment for depression. It can also provide efficacy and tolerability over established adjunctive treatments formajor depressive disorder(MDD).Recent study has also suggested it can ameliorate PCP-induced cognitive deficits in mice via 5-HT1A receptors.정의
ChEBI: Brexpiprazole(913611-97-9) is a N-arylpiperazine. It is a novel D2 dopamine and serotonin 1A partial agonist, called serotonin-dopamine activity modulator (SDAM), and a potent antagonist of serotonin 2A receptors, noradrenergic alpha 1B and 2C receptors. Brexpiprazole is a drug candidate useful in treatment and prevention of mental disorders including CNS disorders.Synthesis
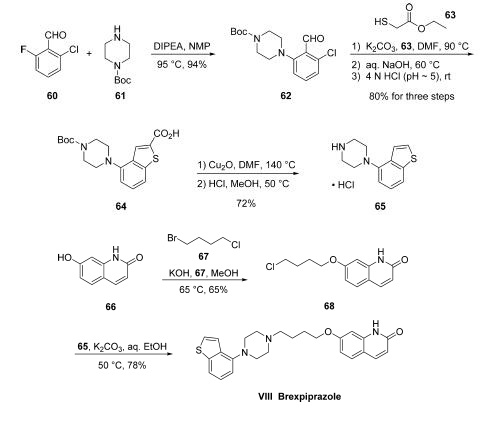
Commercially available fluorobenzaldehyde (60) underwent a substitution reaction with commercial tert-butyl piperazine-1- carboxylate (61) under basic conditions to afford the piperazinyl benzaldehyde 62 in excellent yield. Next, the construction of the benzothiophene was affected by initial condensation of thioglycolic acid ethyl ester 63 with ochlorobenzaldehyde 62 under mildly basic conditions at elevated temperatures. Treatment with aqueous base and adjustment of pH to roughly 5 through the use of 4 N HCl furnished the 2-carboxylic acid benzothiophene 64 in 80% yield across the three-step operation. Next, decarboxylation through the use of cuprous oxide using conditions slightly modified from those originally described by Goosen followed by acidic removal of the Boc protecting group on the terminal piperazine nitrogen secured the key piperazinyl benzothiophene subunit 65 as the corresponding hydrochloride salt.The hydroxyquinolone and linker component synthesis began with alkylation of commercially available quinolone 66 with 1,4-bromochlorobutane (67) under basic conditions to furnish chloroalkoxyquinolone 68. A subsequent alkylation with hydrochloride salt 65 using potassium carbonate and warm aqueous ethanol followed by recrystallizative workup resulted in clean conversion to brexpiprazole (VIII) in 78% yield from 68.
효소 저해제
This serotonin-dopamine activity modulator, or SDAM (FW = 433.60 g/mol; CAS 913611-97-9), also known as OPC-34712, Rexulti, 7-{4-[4-(1- benzothiophen-4-yl)piperazin-1-yl]butoxy}quinolin-2(1H)-one, is an atypical antipsychotic drug that acts as a dopamine D2L (Ki = 1.1 nM) and D3 (Ki = 0.3 nM) receptor partial agonist and a partial agonist of 5-HT1A receptors (Ki = 0.12 nM). Brexpiprazole is also an antagonist of the 5-HT2A (Ki = 0.47 nM), 5-HT2B (Ki = 1.9 nM), 5-HT7 (Ki = nM), α1B-adrenergic (Ki = 0.17 nM), α2C-adrenergic (Ki = 0.59 nM), and histamine H1 receptors (Ki = 19 nM). It has negligible affinity for the mACh receptors. Rexulti is an FDA-approved add-on medication for major depressive disorder in adults. See AripiprazoleMode of action
Brexpiprazole's suggested mechanism of action is based on its impact on dopamine and serotonin receptors. As a serotonin-dopamine activity modulator (SDAM), it acts as a novel partial agonist for D2 dopamine and serotonin 1A receptors while effectively blocking the serotonin 2A receptors, as well as noradrenergic alpha 1B and 2C receptors.참고 문헌
https://en.wikipedia.org/wiki/Brexpiprazolehttps://www.drugbank.ca/drugs/DB09128
Maeda, K, et al. "Brexpiprazole I: in vitro and in vivo characterization of a novel serotonin-dopamine activity modulator." Journal of Pharmacology & Experimental Therapeutics 350.3(2014):589-604.
Maeda, K, et al. "Brexpiprazole II: antipsychotic-like and procognitive effects of a novel serotonin-dopamine activity modulator." Journal of Pharmacology & Experimental Therapeutics 350.3(2014):605.
Yoshimi, N, et al. "Effects of brexpiprazole, a novel serotonin-dopamine activity modulator, on phencyclidine-induced cognitive deficits in mice: a role for serotonin 5-HT1A receptors. " Pharmacology Biochemistry & Behavior 124(2014):245.
브렉스피프라졸 준비 용품 및 원자재
원자재
4-BROMO-BENZO[B]THIOPHENE
1-BENZOTHIEN-4-YLAMINE
7-Hydroxyquinolinone
7-(4-BROMOBUTOXY)-QUINOLINE-2(1H)-ONE
(1-Benzothiophen-4-yl)boronic acid
Benzothiazole, 4-bromo- (7CI,8CI)
4-chloro- Benzo[b]thiophene
준비 용품
브렉스피프라졸 공급 업체
글로벌( 359)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Huadong Medicine (Xi'an)Bodyguard Pharmaceutical Co.,Ltd. | +86-029-86185165 +8618629664246 |
guoyuan@eastchinapharm.com | China | 1615 | 58 |
| Changzhou Rokechem Technology Co., Ltd. | 18758118018 |
sales001@rokechem.com | China | 255 | 58 |
| Seasons Biotechnology Co., Ltd. | +86-0576-89232655 +86-13566878689 |
info@seasonsbio.com | China | 46 | 58 |
| TAIZHOU YUXIN BIOTECHNOLOGY CO,.LTD | +86-576-88902229;+86-0576-88902229 +8613968687450 |
yuxin@yuxchem.com | China | 122 | 58 |
| Dorne Chemical Technology co. LTD | +86-13583358881 +86-18560316533 |
Ethan@dornechem.com | China | 294 | 58 |
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 |
sales@sdperfect.com | China | 294 | 58 |
| Zibo Hangyu Biotechnology Development Co., Ltd | +86-0533-2185556 +8617865335152 |
Mandy@hangyubiotech.com | China | 11013 | 58 |
| Shandong Hanjiang Chemical Co., Ltd | +86-0533-2066820 +8618369939125 |
hanson@sdhanjiang.com | China | 1527 | 58 |
| Sigma Audley | +86-18336680971 +86-18126314766 |
nova@sh-teruiop.com | China | 525 | 58 |
| Shanghai Affida new material science and technology center | +undefined15081010295 |
2691956269@qq.com | China | 359 | 58 |
브렉스피프라졸 관련 검색:
트라조돈
Tetrabenazine
Ramelteon
ETOFIBRATE
VILAZODONE
Posaconazole
Eszopiclone
Brexpiprazole sulfoxide impurity
Brexpiprazole-d8
Brexpiprazole Impurity 34
4-(4-benzo[b]thiophen-4-yl-piperazin-1-yl)butan-1-ol
Empagliflozin
Epalrestat
Brexpiprazole Impurity 44
4-chloro- Benzo[b]thiophene
Brexpiprazole Impurity
Brexpiprazole Impurity
Brexpiprazole Impurity