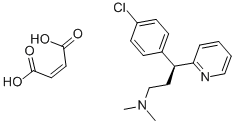
Dexchlorpheniramine Maleate
|
|
Dexchlorpheniramine Maleate 속성
- 녹는점
- 112-115 °C(lit.)
- 알파
- D25 +44.3° (c = 1 in dimethylformamide)
- 저장 조건
- 2-8°C
- 용해도
- 물에 매우 잘 녹으며, 에탄올(96%), 메탄올 및 염화메틸렌에 잘 녹습니다.
- 물리적 상태
- Solid
- 색상
- White to off-white
- 수소이온지수(pH)
- pH(10g/l, 25℃) : 4.0~5.0
- Merck
- 13,2198
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 25 | ||
| 안전지침서 | 36/37/39-45 | ||
| 유엔번호(UN No.) | UN 2811 6.1/PG 3 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | US6504000 | ||
| HS 번호 | 2933399090 | ||
| 기존화학 물질 | KE-05827 |
Dexchlorpheniramine Maleate C화학적 특성, 용도, 생산
개요
Dexchlorpheniramine Maleate is the maleate salt form of dexchlorpheniramine, an alkylamine, and first-generation histamine antagonist with anti-allergic activity. Dexchlorpheniramine maleate competitively blocks H1 receptors, thereby preventing the actions of histamine on bronchial smooth muscle, capillaries and gastrointestinal (GI) smooth muscle. This prevents histamine-induced bronchoconstriction, vasodilation, increased capillary permeability, and GI smooth muscle spasms.화학적 성질
Dexchlorpheniramine maleate is a white, odourless, crystalline powder which in aqueous solution has a pH of between 4 and 5. It is freely soluble in water, soluble in alcohol and in chloroform, but only slightly soluble in benzene or ether.용도
Dexchlorpheniramine is an antihistamine used to relieve symptoms of allergy, hay fever, and the common cold. Polaramine (dexchlorpheniramine maleate) is the dextro-isomer of chlorpheniramine maleate and the S-enantiomer of Chlorpheniramine. It is an antihistamine with anticholinergic properties. S-(+)-Chlorpheniramine maleate salt has been used to study the anticholinergic effect of Achillea millefolium and Portulaca olerace on muscarinic receptors of guinea pig tracheal smooth muscle.일반 설명
Dexchlorpheniramine(Polaramine) is the dextrorotatory enantiomer of chlorpheniramine.In vitro and in vivo studies of the enantiomers ofchlorpheniramine showed that the antihistaminic activity existspredominantly in the dextro isomer. As mentioned previously,the dextro isomer has the (S) configuration, which issuperimposable on the (S) configuration of the more activelevorotatory enantiomer of carbinoxamine.Mode of action
Dexchlorpheniramine, the d-isomer of the racemic compound chlorpheniramine, is two times more active than chlorpheniramine. Dexchlorpheniramine does not prevent the release of histamine, but rather, competes with free histamine for binding at the H1-receptor sites, and competitively antagonizes the effects of histamine on H1- receptors in the GI tract, uterus, large blood vessels, and bronchial muscle. Blockade of H1-receptors also suppresses the formation of oedema, flare, and pruritus that result from histaminic activity. Since dexchlorpheniramine binds to central and peripheral H1-receptors, sedative effects are likely to occur. H1- antagonists are structurally similar to anticholinergic agents and therefore possess the potential to exhibit anticholinergic properties of varying degrees. They also have antipruritic effects. Dexchlorpheniramine has high antihistaminic activity, moderate anticholinergic effects and minimal sedative effects. The medicine does not possess antiemetic properties.참고 문헌
[1] P. E. CHAVES. Assessment of cytotoxicity, genotoxicity, and mutagenicity of the dexchlorpheniramine reference standard and pharmaceutical formula in human peripheral blood mononuclear cells[J]. Brazilian Journal of Pharmaceutical Sciences, 2022, 18 1. DOI:10.1590/s2175-97902022e20096.[2] GUSTAVO FACCHINI MSC. Ultraviolet A photosensitivity profile of dexchlorpheniramine maleate and promethazine-based creams: Anti-inflammatory, antihistaminic, and skin barrier protection properties[J]. Journal of Cosmetic Dermatology, 2017, 16 4: e59-e67. DOI:10.1111/jocd.12349.
[3] MASAAKI TAGAWA. Neuroimaging of histamine H1-receptor occupancy in human brain by positron emission tomography (PET): a comparative study of ebastine, a second-generation antihistamine, and (+)-chlorpheniramine, a classical antihistamine.[J]. British journal of clinical pharmacology, 2001, 52 5 1: 501-509. DOI:10.1046/J.1365-2125.2001.01471.X.
[4] DONG LIANG. Identification of anthelmintic parbendazole as a therapeutic molecule for HNSCC through connectivity map-based drug repositioning[J]. Acta Pharmaceutica Sinica B, 2022, 12 5: Pages 2429-2442. DOI:10.1016/j.apsb.2021.12.005.
Dexchlorpheniramine Maleate 준비 용품 및 원자재
원자재
준비 용품
Dexchlorpheniramine Maleate 공급 업체
글로벌( 141)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49392 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354 +1-00000000000 |
marketing@targetmol.com | United States | 19892 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 |
eric@witopchemical.com | China | 23555 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 |
1026@dideu.com | China | 8232 | 58 |
| AFINE CHEMICALS LIMITED | +86-0571-85134551 |
info@afinechem.com | China | 15394 | 58 |
| Dayang Chem (Hangzhou) Co.,Ltd. | 571-88938639 +8617705817739 |
info@dycnchem.com | China | 52861 | 58 |
| GIHI CHEMICALS CO.,LIMITED | +8618058761490 |
info@gihichemicals.com | China | 50003 | 58 |
| LEAPCHEM CO., LTD. | +86-852-30606658 |
market18@leapchem.com | China | 43348 | 58 |
Dexchlorpheniramine Maleate 관련 검색:
2,2',4,4',6,6'-헥사나이트로다이페닐아민 페니라민 말레산염 말레인산클로로페니라민
2-Phenoxyethanol
Ceramides
Nicotinic acid
Nicotinamide
Arbutin
Centella asiatica extract
Squalane
Dexchlorpheniramine Maleate
2-(4-Chlorobenzyl)pyridine
DEXCHLORPHENIRAMINE
Ammonium hydrogen maleate
3-(4-CHLORO-PHENYL)-PROPYLAMINE
3-(2-PYRIDINYL)PROPYLAMINE
BROMPHENIRAMINE MALEATE IMP. B (EP) AS MALEATE: DEXCHLORPHENIRAMINE MALEATE
DEXCHLORPHENIRAMINE MALEATE IMP. A (EP) AS MALEATE: (3RS)-N,N-DIMETHYL-3-PHENYL-3-(PYRIDIN-2-YL)PROPAN-1-AMINE MALEATE