Current Location: HOME >> Enterprise Certification
Contact information
| Country: | China |
|---|---|
| Tel: | |
| Mobile: | 0086-17821030819 |
| E-mail: | June.Liu@Desano.com |
| QQ: | 1546418271 |
| Skype: | Chat Now! |
| Address: | 1479 Zhangheng Road,Zhangjiang Hi-Tech Park |
Gold product
- CAS:312693-72-4
- Purity:99%
- CAS:951-78-0
- Purity:99%
- CAS:1445-07-4
- Purity:0.99
Enterprise Basic Information

Enterprise Certification
 |
|
| Type of Enterprise: | Limited Liability Company |
| Legal Representative: | Kan Ying |
| Registered Capital: | CNY100millions |
| Founded Date: | 2021-08-16 |
| Registered Address: | building 10, No.,816 Xinyang Road,Shanghai City |
| Term of Validity: | 2021-08-16—2051-08-15 |
| Business Scope: | Biotechnology,medical science and technology in the field of techncal services,technology development、consulting、transfer and promotion,sales on chemical products、mechanical equipments、electronics、instruments and apparatus、packing-materials, imports and exports,medical research and experimental development. |
| Staff number: | 100-500 People |
| Number of R & D department: | 1-5 |
| Annual turnover: | RMB 50 million-100 million |
| Main consumer market: | Pharmaceutical factory Medical materials factory Research InstituteL trading company |
| Main marketing location: | South Korea India Japan United States Germany |
| Main product or service: | P-reagents, Nuleosides,Nuleotides,Phosphoramidites,CPG |
Manufacturer
| Marketing : | SHANGHAI NOVRIBO BIOTECH |
| Address: | 1479 Zhangheng Road,Zhangjiang Hi-tech Park,Shanghai |
| Area: | Shanghai, China |
| Pictures of scenes: | |
|
|
|
| Marketing : | Puxin/ Huayao |
| Address: | Yancheng City, Jiangsu; Shanghai |
| Area: | Shanghai ,China |
| Equipment: | GC-MASS, reactors ,analytical equipments |
| Pictures of scenes: | |
|
|
| Brief description : | GMP ,FDA approved |
| Quality inspection equipment: | GC-MS HPLC |
|
|
| Certificate of accreditation: | ISO |
|
|
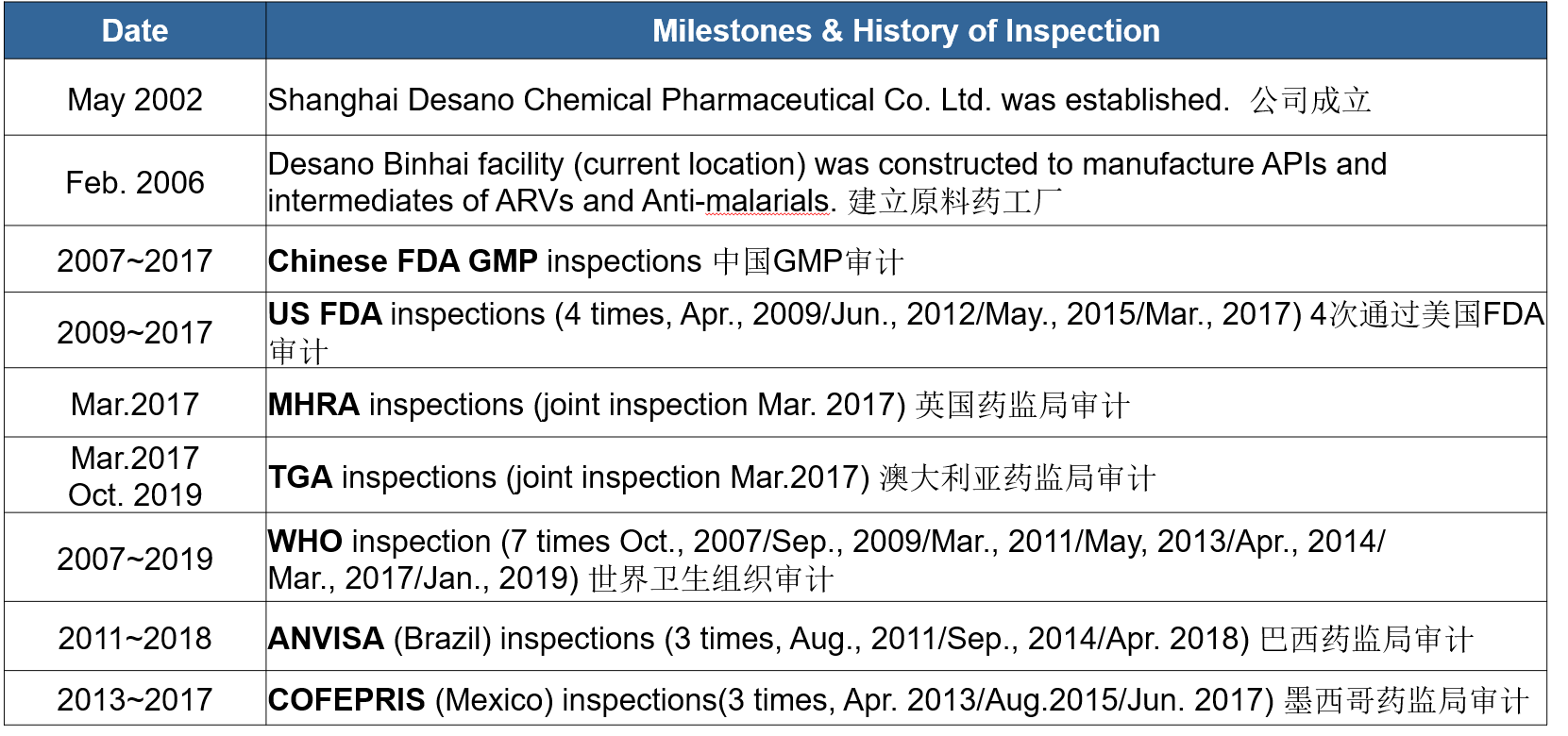
| Brief description : | 4factories with 3 GMP and FDA appoved sites ,one GMP-Equivalent facotry for CDMO.2R&D centers. |
| Pictures of scenes: | |
|
|
|
| Staff composition for example: | GMP&FDA- approved factories, 2R&D center with expertise with our 10years nucleosides experience |
| Professional skills: | CPG and nucleoside experience more then 10years |
| Pictures of scenes: | |
|
|