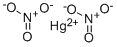MERCURIC NITRATE suppliers
MERCURIC NITRATE
- CAS:
- 10045-94-0
- MF:
- HgN2O6
- MW:
- 324.6
Properties
- Melting point:
- 79°C
- Boiling point:
- decomposes [CRC10]
- Density
- 1.025 g/mL at 25 °C
- solubility
- soluble in H2O; insoluble in ethanol
- form
- colorless hygroscopic crystals
- color
- colorless hygroscopic crystals, crystalline
- Water Solubility
- soluble H2O; insoluble EtOH [CRC10]
- Stability:
- Stable. Incompatible with strong reducing agents, combustible materials, most common metals.
Safety Information
- Symbol(GHS)




GHS03,GHS08,GHS09,GHS06
- Signal word
- Danger
- Hazard statements
- H410-H272-H373-H300+H310+H330
- Precautionary statements
- P210-P220-P221P280-P370+P378-P501-P260-P314-P501-P273-P391-P501
- Hazard Codes
- Xn
- Risk Statements
- 8-23/24/25-33-34
- Safety Statements
- 36/37
- RIDADR
- UN 3264 8/PG 3
- WGK Germany
- 3
- HazardClass
- 6.1(a)
- PackingGroup
- II
Use
MERCURIC NITRATE is noncombustible, but, as an oxidizing agent, will accelerate the burning of combustible materials. If large quantities are involved in a fire or the combustible material is finely divided, an explosion may result. Light sensitive. Mixtures with alkyl esters may explode, owing to the formation of alkyl nitrates. Mixtures with phosphorus, tin(II) chloride, or other reducing agents may react explosively [Bretherick 1979. p. 108-109]. Acetylene forms a sensitive acetylide when passed into an aqueous solution of MERCURIC NITRATE [Mellor 4:933. 1946-47]. Should not be mixed with alcohols as explosive mercury fulminates may be formed [Bahme 1961. p. 9]. Is violently reduced by hypophosphoric acid [Mellor 4:993. 1946-47]. Reacts with phosphine to give a yellow precipitate that explodes when heated or subjected to shock [Mellor 4:993. 1946-47].
According to relevant laws, regulations, policies and the provisions of this website, this website does not display the product information!
The source is "剧毒、有毒、易致癌化学品". If the information is inaccurate, please contact the platform to modify it.
contact information:info@chemicalbook.com 400-158-660