|
ChemicalBook Optimization Suppliers |
|
| 化学名: | アタルレン | | 英語化学名: | Ataluren (PTC124) | | 别名: | Ptc-124;PTC-124,ataluren;Ataluren (PTC124);Ataluren PTC 124 Ataluren;Ataluren, >=98%;PTC124, 775304-57-9;3-[5-(2-Fluorophenyl)-1,2,4-oxadiazol-3-yl]benzoic acid;Ataluren [usan] | | CAS番号: | 775304-57-9 | | 分子式: | C15H9FN2O3 | | 分子量: | 284.24 | | EINECS: | | | カテゴリ情報: | Inhibitors;Ribosome binding agent;API | | Mol File: | 775304-57-9.mol | 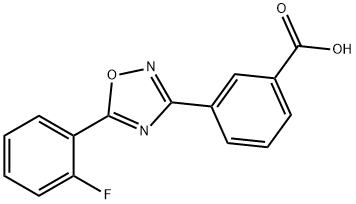 |
| 融点 | 241 - 242°C | | 沸点 | 503.7±60.0 °C(Predicted) | | 比重(密度) | 1.379 | | 貯蔵温度 | Refrigerator | | 溶解性 | DMSO (Slightly) | | 外見 | White to off-white solid. | | 酸解離定数(Pka) | 3.58±0.10(Predicted) | | 色 | White to Off-White | | InChI | InChI=1S/C15H9FN2O3/c16-12-7-2-1-6-11(12)14-17-13(18-21-14)9-4-3-5-10(8-9)15(19)20/h1-8H,(H,19,20) | | InChIKey | OOUGLTULBSNHNF-UHFFFAOYSA-N | | SMILES | C(O)(=O)C1=CC=CC(C2N=C(C3=CC=CC=C3F)ON=2)=C1 | | CAS データベース | 775304-57-9 |
| | アタルレン Usage And Synthesis |
| 効能 | デュシェンヌ型筋ジストロフィー治療薬 | | 説明 | Ataluren is a drug marketed under the trade name Translarna®
which was developed by PTC Therapeutics and approved by the
European Union in May 2014 for the treatment of Duchenne’s muscular
dystrophy (DMD) and potentially other genetic disorders.
Ataluren renders ribosomes less sensitive to premature stop or
‘read-through’ codons, which are thought to be beneficial in diseases
such as DMD and cystic fibrosis. | | 説明 | Nonsense mutations create a premature termination of mRNA translation and have been implicated in various genetic disorders, including muscular dystrophy and cystic fibrosis. PTC-124 is a nonaminoglycoside that has been reported to selectively induce ribosomes to read through premature nonsense stop signals on mRNA, thus allowing the production of full length, functional proteins. In a mouse model of cystic fibrosis caused by nonsense mutations, PTC-124 treatment (60 mg/kg s.c. injection or 0.3-0.9 mg/ml orally) has been shown to restore cystic fibrosis transmembrane conductance regulator (CFTR) protein expression and function. The target activity of PTC-124 was initially evaluated by firefly luciferase reporter cell-based nonsense codon assay (IC50 = 7 nM); however, subsequent assessments using a Renilla reniformis luciferase reporter have failed to produce nonsense codon suppression activity. Thus, while PTC-124 is in clinical testing in patients with nonsense mutations within the CFTR or dystrophin genes, controversy surrounds its exact mechanism of action. | | 使用 | Nonsense mutations create a premature termination of mRNA translation and have been implicated in various genetic disorders, including muscular dystrophy and cystic fibrosis. PTC-124 is a nonaminoglycoside that has been reported to selectively induce ribosomes to read through premature nonsense stop signals on mRNA, thus allowing the production of full-length, functional proteins. In a mouse model of cystic fibrosis caused by nonsense mutations, PTC-124 treatment (60 mg/kg s.c. injection or 0.3-0.9 mg/ml orally) has been shown to restore cystic fibrosis transmembrane conductance regulator (CFTR) protein expression and function. The target activity of PTC-124 was initially evaluated by firefly luciferase reporter cell-based nonsense codon assay (IC50 = 7 nM); however, subsequent assessments using a Renilla reniformis luciferase reporter have failed to produce nonsense codon suppression activity. Thus, while PTC-124 is in clinical testing in patients with nonsense mutations within the CFTR or dystrophin genes, controversy surrounds its exact mechanism of action.[Cayman Chemical] | | 使用 | PTC-124 is a nonaminoglycoside that has been reported to induce ribosomes to read through premature nonsense stop signals on mRNA, allowing the production of full-length functional proteins, | | 定義 | ChEBI: 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]benzoic acid is a ring assembly and an oxadiazole. | | 合成 | The sequence to construct ataluren, which was described by the
authors at PTC Therapeutics, commenced with commercially available
methyl 3-cyanobenzoate (38). This ester was exposed to
hydroxylamine in aqueous tert-butanol and warmed gently until
the reaction was deemed complete. Then this mixture was treated
with 2-fluorobenzoyl chloride dropwise and subsequently triethylamine
dropwise. To minimize exotherm and undesired side products,
careful control of the addition of reagents was achieved
through slow dropwise addition of these liquid reagents. Upon
complete consumption of starting materials and formation of amidooxime
39, the aqueous reaction mixture was then heated to
85 ?? to facilitate 1,2,4-oxadiazole formation, resulting in the tricyclic
ester 40 in excellent yield across the three steps. Finally,
saponification of ester 40 through the use of sodium hydroxide followed
by acidic quench gave ataluren (V) in 96% over the two-step
sequence. 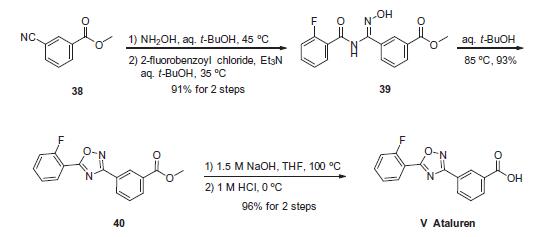
| | target | CFTR |
|