|
|
| | 4-Methoxy-3-(trifluoromethyl)aniline Basic information |
| Product Name: | 4-Methoxy-3-(trifluoromethyl)aniline | | Synonyms: | 3-TRIFLUOROMETHYL-P-ANISIDINE;5-AMINO-2-METHOXYBENZOTRIFLUORIDE;4-METHOXY-3-(TRIFLUOROMETHYL)ANILINE;TIMTEC-BB SBB002596;3-AMINO-6-METHOXYBENZOTRIFLUORIDE;5-amino-2-methoxybenzotrifluoride,97%;3-Amino-6-methoxybenzotriflyoride;3-Amino-6-methoxybenzotrifloride | | CAS: | 393-15-7 | | MF: | C8H8F3NO | | MW: | 191.15 | | EINECS: | 670-719-8 | | Product Categories: | | | Mol File: | 393-15-7.mol | 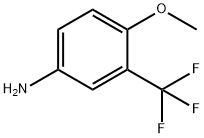 |
| | 4-Methoxy-3-(trifluoromethyl)aniline Chemical Properties |
| Melting point | 28 °C | | Boiling point | 112 °C | | density | 1.280±0.06 g/cm3(Predicted) | | Fp | 112°C/18mm | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | form | solid | | pka | 3.98±0.10(Predicted) | | Appearance | Yellow to brown Solid | | BRN | 2723972 | | InChI | InChI=1S/C8H8F3NO/c1-13-7-3-2-5(12)4-6(7)8(9,10)11/h2-4H,12H2,1H3 | | InChIKey | CQJCPOVTPNWVBW-UHFFFAOYSA-N | | SMILES | C1(N)=CC=C(OC)C(C(F)(F)F)=C1 | | CAS DataBase Reference | 393-15-7(CAS DataBase Reference) |
| Hazard Codes | Xi,T,Xn | | Risk Statements | 20/21/22-36/37/38-52-22 | | Safety Statements | 26-36/37/39 | | RIDADR | UN2811 | | WGK Germany | WGK 3 | | Hazard Note | Irritant | | HazardClass | TOXIC | | HazardClass | 6.1 | | HS Code | 2922290090 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Acute Tox. 4 Oral
Aquatic Chronic 3 |
| Provider | Language |
|
ALFA
| English |
| | 4-Methoxy-3-(trifluoromethyl)aniline Usage And Synthesis |
| Uses | 4-Methoxy-3-(trifluoromethyl)aniline is a fluorinated aniline derivative used as a pharmaceutical intermediate for the synthesis of antibacterial and antiparasitic quinoxaline-2,3-diamine derivatives. | | Synthesis | The general procedure for the synthesis of 4-methoxy-3-trifluoromethylaniline using 2-methoxy-5-nitrobenzotrifluoride as starting material was as follows: 2-methoxy-5-nitrobenzotrifluoride (5.08 g, 23.0 mmol) was dissolved in methanol (200 mL), and 10% Pd-C catalyst (50% water, 250 mg) was added. The reaction mixture was stirred continuously for 3.5 h at room temperature and under atmospheric pressure hydrogen atmosphere. Upon completion of the reaction, the catalyst was removed by diatomaceous earth filtration and the filtrate was concentrated to dryness under reduced pressure to afford 4-methoxy-3-(trifluoromethyl)aniline (4.67 g, 100% yield). Results of mass spectrometry analysis: m/z = 192 ([M+H]+). | | References | [1] Patent: EP1403255, 2004, A1. Location in patent: Page 121
[2] Patent: US2005/209260, 2005, A1. Location in patent: Page/Page column 143-144
[3] Patent: US2007/49609, 2007, A1. Location in patent: Page/Page column 122
[4] Synthesis, 2006, # 19, p. 3316 - 3340
[5] Patent: FR745293, 1932, |
| | 4-Methoxy-3-(trifluoromethyl)aniline Preparation Products And Raw materials |
|