|
|
| | TALIPEXOLE Basic information |
| Product Name: | TALIPEXOLE | | Synonyms: | TALIPEXOLE;5,6,7,8-Tetrahydro-6-(2-propenyl)-4H-thiazolo[4,5-d]azepin-2-amine;Domin;6-Allyl-2-amino-5,6,7,8-tetrahydro-4H-thiazolo[4,5-d]azepine;6-Allyl-5,6,7,8-tetrahydro-4H-thiazolo[4,5-d]azepin-2-amine;Talipexol;Talipexol [spanish];Talipexolum | | CAS: | 101626-70-4 | | MF: | C10H15N3S | | MW: | 209.31 | | EINECS: | | | Product Categories: | | | Mol File: | 101626-70-4.mol | 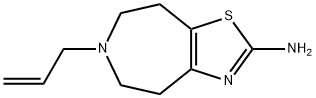 |
| | TALIPEXOLE Chemical Properties |
| Boiling point | 364.6±37.0 °C(Predicted) | | density | 1.167±0.06 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | Soluble in DMSO | | form | Powder | | pka | 7.22±0.20(Predicted) |
| | TALIPEXOLE Usage And Synthesis |
| Description | Domin was launched in Japan for Parkinson's disease. The compound is
readily accessible in two steps from I-allylhexahydro-4H-azepin4-one. Its
antiparkinson activity resides in the fact that talipexole is a selective agonist for
presynaptic dopamine D2 receptors with no D1 receptor activity. It is a clonidine-like α2-adrenoreptor agonist and has fewer GI side effects than bromocriptine the most
widely used dopamine angonist. Also it is 70 times more potent than bromocriptine
and has a more rapid onset of action. In addition to its anti-tremor activity there was
a sedative effect but no emesis. The latter probably a result of talipexole's 5-HT3
receptor antagonistic activity. Its metabolites have no biological activity, there was
no development of tolerence and its effect could be enhanced by L-dopa. | | Originator | Boehringer Ingelheim (Germany) | | Definition | ChEBI: 6-prop-2-enyl-4,5,7,8-tetrahydrothiazolo[4,5-d]azepin-2-amine is an azepine. | | Brand name | Domin |
| | TALIPEXOLE Preparation Products And Raw materials |
|