- BOC-4-ABZ-OH
-

- $1.00 / 1KG
-
2019-12-20
- CAS:66493-39-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: Customized
|
| | BOC-4-ABZ-OH Basic information |
| Product Name: | BOC-4-ABZ-OH | | Synonyms: | N-ALPHA-T-BUTOXYCARBONYL-4-AMINO-BENZOIC ACID;N-ALPHA-T-BUTOXYCARBONYL-P-AMINOBENZOIC ACID;N-BOC-4-AMINOBENZOIC ACID;Boc-4-aminobenzoic acid98+%;RARECHEM EM WB 0012;N-Boc-4-aminobenzoic;Boc-4-Abz-OH(Boc-4-aMinobenzoic Acid);Boc-PABA-OH | | CAS: | 66493-39-8 | | MF: | C12H15NO4 | | MW: | 237.25 | | EINECS: | 676-774-4 | | Product Categories: | Benzene series;Amino Acids | | Mol File: | 66493-39-8.mol | 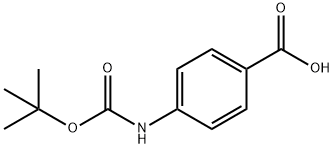 |
| | BOC-4-ABZ-OH Chemical Properties |
| Melting point | ~200 °C (dec.) | | Boiling point | 336.2±25.0 °C(Predicted) | | density | 1.242±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,2-8°C | | solubility | soluble in Methanol | | pka | 4.33±0.10(Predicted) | | form | Powder | | color | Beige | | BRN | 2115614 | | Major Application | peptide synthesis | | InChI | 1S/C12H15NO4/c1-12(2,3)17-11(16)13-9-6-4-8(5-7-9)10(14)15/h4-7H,1-3H3,(H,13,16)(H,14,15) | | InChIKey | ZJDBQMWMDZEONW-UHFFFAOYSA-N | | SMILES | CC(C)(C)OC(=O)Nc1ccc(cc1)C(O)=O |
| Risk Statements | 36/37/38 | | Safety Statements | 22-24/25 | | WGK Germany | 3 | | HazardClass | IRRITANT | | HS Code | 29242990 | | Storage Class | 11 - Combustible Solids |
| | BOC-4-ABZ-OH Usage And Synthesis |
| Chemical Properties | Light brown powder | | Uses | 4-(N-tert-Butoxycarbonyl)aminobenzoic Acid is the tert-Butyloxycarbonyl group protected derivative of 4-Aminobenzoic Acid (A591500), an widely distributed B complex factor in nature. | | reaction suitability | reaction type: Boc solid-phase peptide synthesis | | Synthesis | General procedure for the synthesis of 4-(N-tert-butoxycarbonylamino)benzoic acid from di-tert-butyl dicarbonate and p-aminobenzoic acid: sodium hydroxide (1.46 g, 36.5 mmol) was added to a mixture of dioxane (70 mL) and water (35 mL) of 4-aminobenzoic acid (5.00 g, 36.5 mmol), followed by di-tert-butyl dicarbonate (11.9 g, 54.8 mmol). The reaction mixture was stirred at room temperature for 24 hours. Upon completion of the reaction, the solvent was removed by rotary evaporation and aqueous 3N hydrochloric acid was added slowly and dropwise to the residue to adjust the pH to 3. The precipitate precipitated was collected by filtration, washed with water and dried to afford the target product 4-(N-tert-butoxycarbonylamino)benzoic acid (8.28 g, 96% yield) as a white solid. | | References | [1] Journal of Medicinal Chemistry, 2001, vol. 44, # 3, p. 441 - 452
[2] Journal of Catalysis, 2016, vol. 340, p. 344 - 353
[3] Tetrahedron Letters, 2003, vol. 44, # 37, p. 7103 - 7106
[4] Synthesis, 2005, # 7, p. 1061 - 1068
[5] Chemistry - A European Journal, 2003, vol. 9, # 2, p. 557 - 560 |
| | BOC-4-ABZ-OH Preparation Products And Raw materials |
|