| Company Name: |
Energy Chemical
|
| Tel: |
021-58432009 400-005-6266 |
| Email: |
marketing1@energy-chemical.com |
| Products Intro: |
Product Name:Nitrefazole
CAS:21721-92-6
Purity:NULL Package:10g;1g Remarks:NULL
|
| Company Name: |
TargetMol Chemicals Inc.
|
| Tel: |
4008200310 |
| Email: |
marketing@tsbiochem.com |
| Products Intro: |
Product Name:Nitrefazole
CAS:21721-92-6
Purity:98% Package:100 mg;5 mg;50 mg
|
|
| | Nitrefazole Basic information |
| Product Name: | Nitrefazole | | Synonyms: | ALTIMOL;1-(4-NITROPHENYL)-2-METHYL-4-NITROIMIDAZOLE;EMD 15700;2-methyl-4-nitro-1-(4-nitrophenyl)-1h-imidazol;2-methyl-4-nitro-1-(4-nitrophenyl)imidazole;2-methyl-4-nitro-1-(p-nitrophenyl)-imidazol;bt-985;NITREFAZOLE | | CAS: | 21721-92-6 | | MF: | C10H8N4O4 | | MW: | 248.19 | | EINECS: | 244-542-8 | | Product Categories: | | | Mol File: | 21721-92-6.mol | 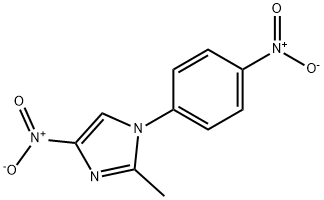 |
| | Nitrefazole Chemical Properties |
| Melting point | 185-187° | | storage temp. | Refrigerator | | solubility | Chloroform (Slightly, Heated), DMSO (Slightly), Methanol (Slightly, Heated) | | form | Solid | | color | Light Yellow to Yellow |
| | Nitrefazole Usage And Synthesis |
| Description | Nitrefazole is an inhibitor of aldehyde dehydrogenase useful in the management of
alcoholism. Cited advantages over disulfiram include longer duration of action,
higher specificity (no inhibition of dopamine β-hydroxylase) and fewer side
effects. It is generally recognized that the efficacy of such agents with respect
to permanent abstinence is low. However, temporary abstinence and/or
controlled drinking may facilitate supportive psychotherapy and/or delay the
onset of severe alcoholic diseases. | | Originator | E. Merck (W. Germany) | | Uses | Nitrefazole is a nitroimidazole derivative with the ability to inhibit aldehyde dehydrogenase, and enzyme crucial to the metabolism of alcohol. This compound is suitable for aldehyde dehydrogenase (ALDH) related research. | | Definition | ChEBI: Nitrefazole is a member of imidazoles. | | Brand name | ALTIMOL | | World Health Organization (WHO) | Nitrefazole, which is used in the treatment of alcoholism, was
introduced in the early 1980s. By 1984 its use had been associated with
hepatotoxic reactions, some of which were fatal. This led to its withdrawal in at
least two countries. WHO has no information to suggest that preparations
containing nitrefazole remain commercially available. |
| | Nitrefazole Preparation Products And Raw materials |
|