| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
18210857532; 18210857532 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:AsoxiMe Chloride
CAS:34433-31-3
Package:10Mg,250Mg,25Mg,50Mg
|
ASOXIME CHLORIDE manufacturers
- Asoxime dichloride
-
- $118.00 / 10mg
-
2026-04-20
- CAS:34433-31-3
- Min. Order:
- Purity: 99.75%
- Supply Ability: 10g
- Asoxime dichloride
-
- $118.00 / 10mg
-
2026-04-20
- CAS:34433-31-3
- Min. Order:
- Purity: 99.75%
- Supply Ability: 10g
|
| | ASOXIME CHLORIDE Basic information |
| Product Name: | ASOXIME CHLORIDE | | Synonyms: | HI 6 Chloride;1-[[[4-(Aminocarbonyl)pyridinio]methoxy]methyl]-2-[(hydroxyimino)methyl]-pyridinium chloride;4-Carbamoyl-1-[[[2-[(hydroxyimino)methyl]pyridinium-1-yl]methoxy]methyl]pyridinium dichloride;Transant;(((4-(iminocarbonyl)pyridinio)methoxy)methyl)-2-((hydroxyimino)methyl)pyridi;4’-carbamoyl-2-formyl-1,1’-(oxydimethylene)di-pyridinium-dichloride-2-oxime;hi6;hi-6 | | CAS: | 34433-31-3 | | MF: | C14H16Cl2N4O3 | | MW: | 359.21 | | EINECS: | | | Product Categories: | Activators;Intermediates & Fine Chemicals;Pharmaceuticals;Amines;Heterocycles | | Mol File: | 34433-31-3.mol | 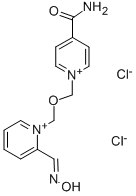 |
| | ASOXIME CHLORIDE Chemical Properties |
| Melting point | 145-147°C | | storage temp. | -20°C | | solubility | H2O: soluble15mg/mL (clear solution) | | form | powder | | color | white to beige | | Water Solubility | H2O: 15mg/mL (clear solution) | | Stability: | 1-10% Aqueous solution | | InChI | 1S/C14H14N4O3.2ClH/c15-14(19)12-4-7-17(8-5-12)10-21-11-18-6-2-1-3-13(18)9-16-20;;/h1-9H,10-11H2,(H-,15,19);2*1H | | InChIKey | QELSIJXWEROXOE-UHFFFAOYSA-N | | SMILES | [Cl-].[Cl-].NC(=O)c1cc[n+](COC[n+]2ccccc2\C=N\O)cc1 |
| WGK Germany | 3 | | Storage Class | 11 - Combustible Solids |
| | ASOXIME CHLORIDE Usage And Synthesis |
| Chemical Properties | Off-White Solid | | Uses | Cholinesterase reactivator. A potential antidote for organophosphate poisoning | | General Description | HI-6 functions as an antidote for treating intoxication caused by nerve agents. It acts as a potent drug in radiation protection and suppresses oxidative stress. HI-6 prevents the toxicity of the antineoplastic drug irinotecan. | | Biological Activity | asoxime, also known as hi-6, is an asymmetric bis-pyridinium aldoxime and reactivates ache via nucleophilic attack of oximate anions on organophosphate-ache conjugates.ache, one of the primary targets of organophosphates, is inactivated by organophosphates via reacting covalently with the active center serine, which blocks the hydrolyzation of acetylcholine at peripheral and central synapses. additionally, the accumulation of acetylcholine leads to an over-stimulation of cholinergic receptors, which disrupts a variety of biological functions. | | Biochem/physiol Actions | HI-6 is an efficient oxime cholinesterase reactivator that is used as an antidote for organophosphates (Ops) exposure. | | in vitro | hi-6 showed a bacteriostatic effect on francisella tularensis (f. tularensis) and inhibited the growth of f. tularensis when treated with 100 mm hi-6 [1]. | | in vivo | female balb/c mice, infected by f. tularensis, were administrated subcutaneously with 100 μl hi-6 for ten days. the lower dose of hi-6 yielded in different symptoms and mortality. compared to mice injected only with tularemia, groups of mice exposed to the lower dose of hi-6 showed a decline in mortality, which can be explained via higher efficacy of bacteriostatic effect while the toxic effect was decreased [1]. female wistar rats were treated intramuscularly with hi-6 at a dose of 15.6 or 156 mg/kg for one day. the level of thiobarbituric acid reactive substances was increased, which was triggered by hi-62 [2]. | | IC 50 | 8.9 μm: blocks ache in human; 126 μm: inhibits acetylcholinesterase (ache) in bovine; 160 μm: blocks muscarinic acetylcholine receptor m1, m2, m3 and m4 in mouse. | | references | [1]. pohanka, m., pavli, o., pikula, j., treml, f., & kuca, k. modulation of tularemia disease progress by the bisquaternary pyridinium oxime hi-6. acta veterinaria brno. 2010; 79(3): 443-448.
[2]. pohanka, m., sobotka, j., svobodova, h., & stetina, r. sulfur mustard induced oxidative stress and its alteration using asoxime (hi-6). interdisciplinary toxicology. 2013; 6(4). |
| | ASOXIME CHLORIDE Preparation Products And Raw materials |
|