|
|
| | 4,4,4-Trifluoro-1-phenyl-1,3-butanedione Basic information |
| Product Name: | 4,4,4-Trifluoro-1-phenyl-1,3-butanedione | | Synonyms: | TRIFLUORO-1-PHENYL-1,3-BUTANEDIONE;1,1,1-Trifluoro-4-phenyl-2,4-butanedione;1-Benzoyl-3,3,3-trifluoro-2-propanone;1-Benzoyl-3,3,3-trifluoroacetone;4,4,4-Trifluoro-1-phenylbutane-1,3-dione ,98%;ω-(Trifluoroacetyl)acetophenone;4,4,4-Trifluoro-1-phenyl-1,3-butanedione,99%;4-Phenyl-1,1,1-trifluorobuta | | CAS: | 326-06-7 | | MF: | C10H7F3O2 | | MW: | 216.16 | | EINECS: | 206-307-8 | | Product Categories: | organofluorine compounds;Miscellaneous | | Mol File: | 326-06-7.mol | 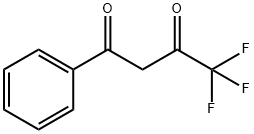 |
| | 4,4,4-Trifluoro-1-phenyl-1,3-butanedione Chemical Properties |
| Melting point | 38-40 °C(lit.) | | Boiling point | 224 °C(lit.) | | density | 1,113 g/cm3 | | Fp | 210 °F | | storage temp. | Sealed in dry,Room Temperature | | solubility | 95% ethanol: soluble25mg/mL, clear, colorless to yellow | | form | Crystalline Low Melting Solid | | pka | pK1: 6.35 (25°C) | | color | White to yellow | | Water Solubility | Sparingly Soluble in water (0.24 g/L) (25°C). | | BRN | 1875083 | | InChI | InChI=1S/C10H7F3O2/c11-10(12,13)9(15)6-8(14)7-4-2-1-3-5-7/h1-5H,6H2 | | InChIKey | VVXLFFIFNVKFBD-UHFFFAOYSA-N | | SMILES | C(C1=CC=CC=C1)(=O)CC(=O)C(F)(F)F | | CAS DataBase Reference | 326-06-7(CAS DataBase Reference) | | EPA Substance Registry System | 1,3-Butanedione, 4,4,4-trifluoro-1-phenyl- (326-06-7) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 24/25 | | WGK Germany | 3 | | TSCA | TSCA listed | | HazardClass | IRRITANT | | HS Code | 29147000 | | Storage Class | 11 - Combustible Solids |
| | 4,4,4-Trifluoro-1-phenyl-1,3-butanedione Usage And Synthesis |
| Chemical Properties | WHITE TO YELLOW CRYSTALLINE LOW MELTING SOLID | | Uses | 4,4,4-Trifluoro-1-phenyl-1,3-butanedione was used in the synthesis of series of NNO ketoimines bearing trifluoromethyl substituents via Schiff base condensation reaction. It was also used in mixed-ligand chelate extraction of trivalent lanthanides and as a ligand in the preparation of ternary lanthanide (Ln) complexes. | | Synthesis | To a three-necked flask fitted with magnetic stirring, a dropping funnel and an inert gas device, 2 mL of anhydrous methanol was added with stirring and 0.230 g (10 ) of clean sodium metal crumbs were added.
mmol) of clean sodium metal crumbs, and after all the sodium was dissolved, a water bath was installed to evaporate the unreacted methanol in the reaction system. After the sodium methanol was completely dried, 5 was added
mL of anhydrous ether, quickly stirred well, and then cooled in an ice bath. 1.2 mL (10 mmol) of ethyl trifluoroacetate was diluted with 5
mL of anhydrous ether was diluted and added to the reaction flask. The ice bath was removed and 1.1 mL (10 mmol) acetophenone was diluted with 5
mL of anhydrous ether was diluted and added dropwise to the reaction system using a dropping funnel. The reaction was stirred at reflux for five hours and at the end of the reaction 10 was added
mL of water was added to dissolve the resulting solid and the mixture was extracted with ether to remove unreacted ketones and esters. The aqueous phase was acidified with 0.5 mL (10
) glacial acetic acid was acidified and then extracted with ether in portions, the organic phases were combined, dried with anhydrous magnesium sulfate and filtered. After the filtrate was evaporated from the ether, a light-yellow crystalline low-melting solid powder 1.691 was precipitated.
g. The light yellow solid powder was recrystallized with ethanol-water to obtain light yellow pure benzoyltrifluoroacetone. |
| | 4,4,4-Trifluoro-1-phenyl-1,3-butanedione Preparation Products And Raw materials |
|