|
|
| | 1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester Basic information |
| Product Name: | 1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester | | Synonyms: | 5-AMINO-1-METHYL-1H-BENZIMIDAZOLE-2-BUTANOIC ACID ETHYL ESTER;Ethyl-4-(5-aMino-1-Methy-1H-benzo[d] iMidazol-2-yl)butanoate;1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester;4-(5-Amino-1-methyl-1H-benzoimidazol-2-yl)-butyric acidethylester;5-amino-1-methyl-2-Benzimidazolebutyric acid ethyl ester;5-AMino-1-Methyl-;4-[5-AMino-1-MethylbenziMidazol-2-yl]butanoic acid ethyl ester;1-Methyl-5-aMino-1H-benziMidazole-2-butanoic acid ethyl este | | CAS: | 3543-73-5 | | MF: | C14H19N3O2 | | MW: | 261.32 | | EINECS: | 680-673-0 | | Product Categories: | Aromatics;Heterocycles;Intermediates | | Mol File: | 3543-73-5.mol | 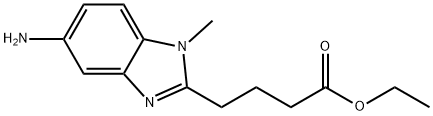 |
| | 1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester Chemical Properties |
| Melting point | 130-135°C | | Boiling point | 471.6±25.0 °C(Predicted) | | density | 1.21±0.1 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2–8 °C | | solubility | Chloroform (Slightly), Ethyl Acetate (Slightly) | | form | Solid | | pka | 6.73±0.10(Predicted) | | color | Off-White to Pale Beige | | InChI | InChI=1S/C14H19N3O2/c1-3-19-14(18)6-4-5-13-16-11-9-10(15)7-8-12(11)17(13)2/h7-9H,3-6,15H2,1-2H3 | | InChIKey | JUMGOLYNZBZPKE-UHFFFAOYSA-N | | SMILES | C1(CCCC(OCC)=O)N(C)C2=CC=C(N)C=C2N=1 |
| | 1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester Usage And Synthesis |
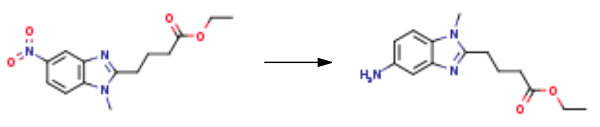
| Chemical Properties | Brown Solid | | Uses | 1-Methyl-5-amino-1H-benzimidazole-2-butanoic Acid Ethyl Ester is a Bendamustine impurity. | | Synthesis |
In the hydrogenation reaction kettle by adding 50 g 1 - methyl -5 - nitro - 1H - benzimidazole -2 - butyric acid ethyl ester, 2.5 g 5% palladium/carbon, 1.3 L methanol, 475 ml ethyl acetate, hydrogen pressure control in the 0.1 - 0.2 mpa, reaction solution is 25 °C reaction 15 hours, TLC monitoring endpoint of the reaction to the reaction is complete. The fluid in a 35 °C decompression filter collects the filtrate; the filtrate is concentrated under reduced pressure to dry after adding 75 ml ethyl acetate, 70 °C stirring for 0.5 hours. After that, the mixture was to 5 °C stirring for 0.5 hours, for 5 °C standing crystallization for 2 hours, filtering, washing the filter cake with acetic acid ethyl ester, filtration cake at 50 °C decompression drying for 5 hours to afford 1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester 35 g, yield 77.8%.
|
| | 1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester Preparation Products And Raw materials |
|