|
ChemicalBook Optimization Suppliers |
|
| 化学名: | 臭化チオトロピウム HYDRATE | | 英語化学名: | Tiotropium bromide hydrate | | 别名: | (1R,2R,4S,5S,7S)-7-[2-HYDROXY-2,2-DI(2-THIENYL)ACETOXY]-9,9-DIMETHYL-3-OXA-9-AZONIATRICYCLO[3.3.1.02,4] NONANE BROMIDE MONOHYDRATE;tiotropium bromide hydrate;TIOTROPIUM BROMIDE MONOHYDRATE;TIOTROPIUM BROMDIE;Tiotropium;(1a,2b,4b,5a,7b)-7-[(hydroxydi-2-thienylacety l)oxy]-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4]nonane bromide hydrate;(1a,2b,4b,5a,7b)-7-[(hydroxydi-2-thienylacetyl)oxy]-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4]nonane bromide;(1α,2β,4β,5α,7β)-7-[(Hydroxydi-2-thienylacety l)oxy]-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4]nonane bromide hydrate | | CAS番号: | 139404-48-1 | | 分子式: | C19H22BrNO4S2.H2O | | 分子量: | 490.43 | | EINECS: | 690-508-4 | | カテゴリ情報: | API;Inhibitors;139404-48-1 | | Mol File: | 139404-48-1.mol | 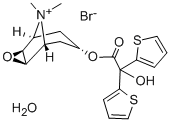 |
| 貯蔵温度 | Store at -20°C | | 溶解性 | Sparingly soluble in water, soluble in methanol, practically insoluble in methylene chloride. | | 外見 | Powder | | InChIKey | MQLXPRBEAHBZTK-SEINRUQRSA-M | | CAS データベース | 139404-48-1(CAS DataBase Reference) |
| | 臭化チオトロピウム HYDRATE Usage And Synthesis |
| 化学的特性 | White or yellowish-white powder or crystals. | | 使用 | Tiotropium Bromide hydrate is a hydrate of tiotropium bromide (Spiriva; Tiova; BA 679BR; tiopropium) that is an anticholinergic and bronchodilator and a muscarinic receptor antagonist. Although tiotropium Bromide (Spiriva; Tiova; BA 679BR; tiopropium) doe | | 定義 | ChEBI: Tiotropium bromide hydrate is a hydrate that is the monohydrate form of tiotropium bromide. Used for maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease. It has a role as a muscarinic antagonist and a bronchodilator agent. | | 臨床応用 | Maintenance treatment of chronic obstructive pulmonary disease | | 薬物相互作用 | Potentially hazardous interactions with other drugs
Avoid administration with other anti-cholinergic
drugs.
Anti-arrhythmics: increased risk of antimuscarinic
side effects with disopyramide. | | 代謝 | Tiotropium is excreted largely unchanged in the urine, although it may undergo some metabolism by non-enzymatic cleavage and by the cytochrome P450 isoenzymes CYP2D6 and CYP3A4. |
|