| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:1-Methylphenanthrene
CAS:832-69-9
Purity:50 μg/ML in Toluene Package:1ML
|
|
| | 1-METHYLPHENANTHRENE Basic information |
| Product Name: | 1-METHYLPHENANTHRENE | | Synonyms: | 1-METHYLPHENANTHRENE;1-methyl-phenanthren;PHENANTHRENE,1-METHYL-;1-Methylphenanthrene 100mg [832-69-9];NSC 146583;1-MethylphenanthreneSolution,2,000mg/L,1ml;1-Methylphenanthrene @50 μg/mL in Toluene;1-MethylphenanthreneSolution,5,000mg/L,1ml | | CAS: | 832-69-9 | | MF: | C15H12 | | MW: | 192.26 | | EINECS: | 212-622-1 | | Product Categories: | | | Mol File: | 832-69-9.mol | 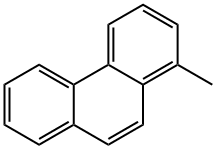 |
| | 1-METHYLPHENANTHRENE Chemical Properties |
| Melting point | 123°C | | Boiling point | 353.25°C (rough estimate) | | density | 1.0561 (estimate) | | refractive index | 1.6031 (estimate) | | storage temp. | Room Temperature | | solubility | Soluble in alcohol (Weast, 1986) | | form | Solid | | color | White powder or solid. | | Water Solubility | 269ug/L(25 ºC) | | Henry's Law Constant | 1.56, 2.33, 3.42, 4.93, and 6.68 x 10-5 atm?m3/mol at 4.1, 11.0, 18.0, 25.0, and 31.0 °C, respectively (Bamford et al.,
1998) | | IARC | 3 (Vol. Sup 7, 92) 2010 | | EPA Substance Registry System | 1-Methylphenanthrene (832-69-9) |
| | 1-METHYLPHENANTHRENE Usage And Synthesis |
| Uses | 1-Methylphenanthrene is a polycyclic aromatic hydrocarbon that has been found in particulate matter from small-scale biomass combustion from old and modern technologies and that has caused acute systemic and lung inflammation in mice after intratracheal aspiration. | | Definition | ChEBI: A member of the class of phenanthrenes that is phenanthrene substituted by a methyl group at position 1. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 45, p. 2009, 1980 DOI: 10.1021/jo01298a054
Tetrahedron Letters, 6, p. 359, 1965 | | General Description | White crystalline powder. | | Air & Water Reactions | Insoluble in water. | | Reactivity Profile | Vigorous reactions, sometimes amounting to explosions, can result from the contact between aromatic hydrocarbons, such as 1-METHYLPHENANTHRENE, and strong oxidizing agents. They can react exothermically with bases and with diazo compounds. Substitution at the benzene nucleus occurs by halogenation (acid catalyst), nitration, sulfonation, and the Friedel-Crafts reaction. 1-METHYLPHENANTHRENE is sensitive to excessive heat and light. | | Fire Hazard | Flash point data for 1-METHYLPHENANTHRENE are not available. 1-METHYLPHENANTHRENE is probably combustible. | | Source | Detected in 8 diesel fuels at concentrations ranging from 0.10 to 210 mg/L with a mean
value of 44.33 mg/L (Westerholm and Li, 1994). Identified in a South Louisiana crude oil at a
concentration of 111 ppm (Pancirov and Brown, 1975). Schauer et al. (1999) reported 1-
methylphenanthrene in diesel fuel at a concentration of 28 μg/g and in a diesel-powered mediumduty
truck exhaust at an emission rate of 17.0 μg/km.
California Phase II reformulated gasoline contained 1-methylphenathrene at a concentration of
3.91 g/kg. Gas-phase tailpipe emission rates from gasoline-powered automobiles with and without
catalytic converters were approximately 1.63 and 122 μg/km, respectively (Schauer et al., 2002).
Schauer et al. (2001) measured organic compound emission rates for volatile organic
compounds, gas-phase semi-volatile organic compounds, and particle-phase organic compounds
from the residential (fireplace) combustion of pine, oak, and eucalyptus. The respective gas-phase
and particle-phase emission rates of 1-methylphenanthrene were 2.22 and 0.579 mg/kg of pine
burned and 1.04 and 0.050 mg/kg of oak burned. The gas-phase emission rate was 0.720 mg/kg of
eucalyptus burned. |
| | 1-METHYLPHENANTHRENE Preparation Products And Raw materials |
|