- Docetaxel Side Chain
-

- $0.00 / 1g
-
2026-04-30
- CAS:196404-55-4
- Min. Order: 1g
- Purity: 98%min HPLC
- Supply Ability: 10000G
|
| | (4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylic acid Basic information |
| Product Name: | (4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylic acid | | Synonyms: | (4R,5R)-3-(2,2-Dimethylpropanoyl)-2-(4-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylic acid;(4S,5R)-3-tert-butoxycarbony-2-(4-anisyl)--phenyl-5-oxazolidinecarboxylic acid;(4S,5R)-2-(4-methoxyphenyl)-4-phenyl-3,5-Oxazolidinedicarboxylic acid 3-(1,1-dimethylethyl)ester;side chain of docetaxel;(4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylic acid;(4S,5R)-2-(4-Methoxyphenyl)-4-phenyl-3,5-oxazolidinedicarboxylic acid 3-tert-butyl ester;(4S,5R)-3-tert-Butoxycarbony-2-(4-anisyl)-4-phenyl-5-oxazolidinecarboxylic acid;(4S,5R)-2-(4-Methoxy-phenyl)-3-N-Boc-4-phenyl-3,5-oxazolidine carboxylic acid | | CAS: | 196404-55-4 | | MF: | C22H25NO6 | | MW: | 399.44 | | EINECS: | 1592732-453-0 | | Product Categories: | API | | Mol File: | 196404-55-4.mol | 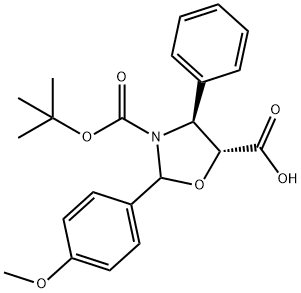 |
| | (4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylic acid Chemical Properties |
| Melting point | 134-138℃ | | Boiling point | 573.8±50.0 °C(Predicted) | | density | 1.238 | | storage temp. | 2-8°C | | pka | 2.99±0.60(Predicted) | | Appearance | White to off-white Solid | | InChI | InChI=1S/C22H25NO6/c1-22(2,3)29-21(26)23-17(14-8-6-5-7-9-14)18(20(24)25)28-19(23)15-10-12-16(27-4)13-11-15/h5-13,17-19H,1-4H3,(H,24,25)/t17-,18+,19/m0/s1 | | InChIKey | MSVWUXLRSKRKFZ-PAMZHZACSA-N | | SMILES | O1[C@@H](C(O)=O)[C@H](C2=CC=CC=C2)N(C(OC(C)(C)C)=O)C1C1=CC=C(OC)C=C1 |
| Safety Statements | 24/25 | | HS Code | 29349990 |
| | (4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylic acid Usage And Synthesis |
| Chemical Properties | White solid | | Uses | (4S,5R)-3-tert-Butoxycarbony-2-(4-anisyl)-4-phenyl-5-oxazolidine carboxylic acid is an intermediate/impurity for docetaxel hydrate (D494420). It can also be used to synthesize quinoline-docetaxel analogues which exhibit anticancer activity. | | Synthesis | The (4S,5R)-3-(tert-butoxycarbonyl)-2-(4-methoxyphenyl)-4-phenyloxazolidine-5-carboxylic acid precursor obtained in the previous step (4.83 g, 9.88 mmol) was dissolved in 10 mL of methanol and 1.0 g of palladium hydroxide was added as a catalyst. Hydrogen was introduced into the reaction system at room temperature (pressure maintained at 20 psi) and the reaction was carried out for 1 h, during which the progress of the reaction was monitored by thin layer chromatography (TLC). Upon completion of the reaction, the reaction mixture was filtered to remove the catalyst, and the filtrate was concentrated under reduced pressure and purified by column chromatography (eluent ratio of petroleum ether/ethyl acetate = 5:1) to afford (4S,5R)-3-(tert-butoxycarbonyl)-2-(4-methoxyphenyl)-4-phenyl-oxazolidine-5-carboxylic acid as a white solid (2.68 g, 67.9% yield). | | References | [1] Patent: US2016/340365, 2016, A1. Location in patent: Paragraph 0140; 0141
[2] Patent: US2016/297784, 2016, A1. Location in patent: Paragraph 0149; 0164; 0165
[3] Bioorganic and Medicinal Chemistry, 2014, vol. 22, # 1, p. 194 - 203 |
| | (4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylic acid Preparation Products And Raw materials |
| Raw materials | tert-butyl (1S,2R)-2-((benzyloxy)carbonyl)-2-hydroxy-1-phenylethylcarbamate-->2-Propanesulfinamide, 2-methyl-N-(phenylmethylene)-, [S(R)]--->(SR,2R,3S)-2-(O-Boc)-3-(tert-butylsulfinyl)-3-phenylisoserine benzyl ester-->3,5-Oxazolidinedicarboxylic acid, 2-(4-methoxyphenyl)-4-phenyl-, 3-(1,1-dimethylethyl) 5-(phenylmethyl) ester, (4S,5R)--->(4S,5R)-3-tert-butoxycarbony-2-(4-anisy)-4-phenyl-5-oxazolidinecarboxylate-->Methyl (2R,3S)-3-(tert-butoxycarbonylamino)-2-hydroxy-3-phenylpropionate-->Docetaxel Impurity 46-->Benzaldehyde-->Di-tert-butyl dicarbonate-->Methanol |
|