|
|
| | (R)-1-N-Boc-2-(hydroxymethyl)piperazine Basic information |
| Product Name: | (R)-1-N-Boc-2-(hydroxymethyl)piperazine | | Synonyms: | (R)-1-Boc-2-Hydroxymethyl-piperazine;1-Piperazinecarboxylic acid, 2-(hydroxymethyl)-, 1,1-dimethylethyl ester, (2R)-;tert-butyl (R)-2-(hydroxymethyl)piperazine-1-carboxylate;(R)-1-N-BOC-2-HYDROXYMETHYLPIPERAZINE HCl;2-Methyl-2-propanyl(2R)-2-(hydroxymethyl)-1-piperazinecarboxylate;(R)-tert-Butyl 2-(hydroxymethyl)piperazine-1-carboxylate;(R)-1-Boc-2-Hydroxymethyl-piperazine ,98%;(S)-1-Boc-2-Hydroxymethyl-piperazine ,98% | | CAS: | 169448-87-7 | | MF: | C10H20N2O3 | | MW: | 216.28 | | EINECS: | | | Product Categories: | Piperaizine | | Mol File: | 169448-87-7.mol | 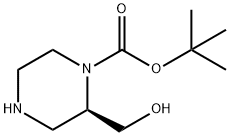 |
| | (R)-1-N-Boc-2-(hydroxymethyl)piperazine Chemical Properties |
| Boiling point | 324.0±17.0 °C(Predicted) | | density | 1.085±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C(protect from light) | | pka | 14.97±0.10(Predicted) |
| | (R)-1-N-Boc-2-(hydroxymethyl)piperazine Usage And Synthesis |
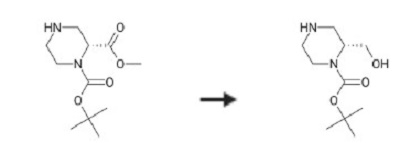
| Uses | (R)-tert-Butyl 2-(hydroxymethyl)piperazine-1-carboxylate is used in preparation of novel substituted quinoline-8-carbonitrile derivatives with androgen receptor degradation activity and uses thereof. | | Synthesis | Lithium aluminum hydride (1M in THF, 26 ml) was added to a solution of L-TERT- butyl 2-methyl (2R)-piperazine-1, 2-dicarboxylate (2. 55 g) in THF (70 ml) at-40°C, and then the reaction was warmed to room temperature. After stirring for 1 h, this reaction system was cooled to 0°C and quenched by sequential addition of water (1 ml), sodium hydroxide (2N, 1 ml), and then water (2 ml). The resulting slurry was filtered and concentrated in vacuo to give (R)-1-N-Boc-2-(hydroxymethyl)piperazine.
 |
| | (R)-1-N-Boc-2-(hydroxymethyl)piperazine Preparation Products And Raw materials |
|