|
ChemicalBook Optimization Suppliers |
|
| 化学名: | タクロリムス | | 英語化学名: | Tacrolimus | | 别名: | (2-propenyl)-,(3s-(3r*(e(1s*,3s*,4s*)),4s*,5r*,8s*,9e,12r*,14r*,15s*,16r*,18s*;,19s*,26ar*))-;16-dimethoxy-4,10,12,18-tetramethyl-8-3-(2-(4-hydroxy-3-methoxycyclohexyl)-1;15,19-Epoxy-3H-pyrido[2,1-c][1,4]oxaazacyclotricosine-1,7,20,21(4H,23H)-tetrone, 5,6,8,11,12,13,14,15,16,17,18,19,24,25,26,26a-hexadecahydro-5,19-dihydroxy-3-[2-(4-hydroxy-3-methoxycyclohexyl)-1-methylethenyl]-14,16-dimethoxy-4,10,12,18-tetramethyl-8-(2-propenyl)-, [3S-[3R*[E(1S*,3S*,4S*)],4S*,5R*,8S*,9E,12R*,14R*,15S*,16R*,18S*,19S*,26aR*]]-;L 679934;FK-506:FR-900506;Fujimycin Prozraf;Tacrolimus,micronisedandpharmagrade | | CAS番号: | 104987-11-3 | | 分子式: | C44H69NO12 | | 分子量: | 804.02 | | EINECS: | 1308068-626-2 | | カテゴリ情報: | Inhibitors;API;Chiral Reagents;Immunosuppressant.;Active Pharmaceutical Ingredients;Fujimycin, Prograf;antibiotic;Intermediates & Fine Chemicals;Pharmaceuticals;APIs;104987-11-3 | | Mol File: | 104987-11-3.mol | 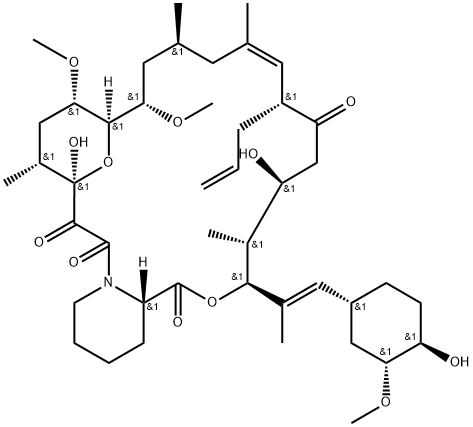 |
| 融点 | 113-115°C | | 沸点 | 871.7±75.0 °C(Predicted) | | 比重(密度) | 1.19±0.1 g/cm3(Predicted) | | 闪点 | 2℃ | | 貯蔵温度 | Sealed in dry,Store in freezer, under -20°C | | 溶解性 | DMSO: >3 mg/mL | | 外見 | solid | | 酸解離定数(Pka) | 9.97±0.70(Predicted) | | 色 | white | | 水溶解度 | Freely soluble in DMSO or ethanol. Poorly soluble in water.Soluble in dimethyl sulfoxide, ethanol, water, acetone, chloroform, ethyl acetate, ether, methanol and dimethyl formamide. | | BCS Class | 2 | | 安定性: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 2 months. | | InChIKey | QJJXYPPXXYFBGM-LFZNUXCKSA-N | | CAS データベース | 104987-11-3(CAS DataBase Reference) |
| | タクロリムス Usage And Synthesis |
| 外観 | 白色〜ほとんど白色, 結晶〜粉末 | | 溶解性 | アセトニトリルに溶ける。 | | 用途 | 強 力 な 免疫抑制剤 で す。
FK506はFK506結合タンパク-12(FKBP12)
と複合体を形成し、さらにカルシニューリン
に結合し、IL-2、インターフェロンに代表さ
れる種々のサイトカインの発現を抑制しま
す。これにより細胞外性 T 細胞の分化増殖を
抑制します。 | | 効能 | 抗アレルギー薬, 免疫抑制薬, カルシニューリン阻害薬 | | 説明 | Tacrolimus, isolated from the microorganism Streptomyces tsukubaensis, is a
macrolide immunosuppressant developed by Fujisawa for organ transplantation. It displays
similar but more potent immunosuppressive activity than cyclosporin. It inhibits both cell
mediated and humoral immune responses. In animal models of organ transplantation,
tacrolimus has been shown to prolong survival of hepatic, renal, cardiac, small intestine,
pancreatic and skin allografts and to reverse cardiac and renal allograft rejection. It has been
used effectively in humans as rescue or primary immunosuppressant therapy in liver or kidney
transplantation. Compared to cyclosporin, tacrolimus causes reduced incidence of infectious
complications and of hypertension and hypercholesterolemia for the allograft recipients. In
common with cyclosporin, tacrolimus binds with high affinity to a family of cytoplasmic
immunosuppressant binding proteins, the immunophilins. This tight complex is proposed as
the biologically active moiety that interacts with intracellular molecules involved in signal
transduction.It inhibits phosphatase activity of calcineurin, an action that may impair the
generation and/or activation of nuclear transcription factors required for lymphokine
(particularly interleukin-2) gene expression. Tacrolimus has also been reported to have
potential in multiple sclerosis, psoriasis, rheumatoid arthritis and uveitis associated with
Behcet‘s disease. | | 説明 | FK-506 is a potent immunosuppressant in the same molecular class as cyclosporin A and rapamycin . Its mechanism of action involves the formation of a high affinity complex (Ki = 0.2 nM) with FK-506 binding protein 12 (FKBP12). This complex then inhibits the activity of the calcium/calmodulin-dependent protein phosphatase, calcineurin, leading to disruption of T cell activation. The physiological effects of FK-506 also include regulation of nitric oxide neurotoxicity, neurotransmitter release, and regulation of Ca2+ release via the ryanodine and inositol-(1,4,5)-trisphosphate (IP3) receptors. In the latter case, FKBP12 forms a tight complex with both ryanodine and IP3 receptors which can be disrupted by FK-506, thereby rendering the receptors leaky to Ca2+. | | 化学的特性 | White Crystalline Solid | | Originator | Fujisawa (Japan) | | 使用 | Tacrolimus (fujimycin) was discovered as a potent inhibitor of IL2 production in a targeted search for novel immunosuppressants. Tacrolimus acts by blocking T cell proliferation in vitro by inhibiting the generation of several lymphokines, notably the original target IL-2. Tacrolimus inhibits the activity of FK-506 binding protein, Ca2+-dependent phosphatase and calcineurin, and activates NF-κB through phosphorylation and degradation of IκBα. | | 使用 | FK-506 (Tacrolimus) is a macrolide immunosuppressive drug that is mainly used after allogeneic organ transplant to reduce the activity of the patient's immune system | | 使用 | For use after allogenic organ transplant to reduce the activity of the patient's immune system and so the risk of organ rejection. It was first approved by the FDA in 1994 for use in liver transplantation, this has been extended to include kidney, heart, | | 使用 | treatment of Cushing's syndrome | | 使用 | An immunosuppressant that blocks T cell proliferation in vitro by inhibiting the generation of several lymphokines, especially IL-2. Shown to inhibit the activity of FK-506 binding protein, thereby reversing its effects on sarcoplasmic reticulum Ca+2 release. | | 適応症 | Tacrolimus is a macrolide lactone originally derived
from Streptomyces tsukubaensis. Although structurally
unrelated to cyclosporine, tacrolimus has a very similar
mechanism of action; that is, it blocks the production of
proinflammatory cytokines by T lymphocytes by inhibiting
calcineurin.Tacrolimus, however, appears to be
10 to 100 times as potent as an immunosuppressive.
Oral tacrolimus (FK506) is used for prevention of organ
rejection in recipients of renal and hepatic transplants. | | 定義 | ChEBI: Tacrolimus is a macrolide containing a 23-membered lactone ring, originally isolated from the fermentation broth of a Japanese soil sample that contained the bacteria Streptomyces tsukubaensis. | | 適応症 | Tacrolimus (Prograf) is a second-generation immunosuppressive
agent that has been approved for use in
liver transplantation. Its efficacy for other transplantations
is being evaluated. It has properties similar to
those of cyclosporine except that weight for weight it is
10 to 100 times more potent. It is a macrolide antibiotic
that selectively inhibits transcription of a specific set of
lymphokine genes in T lymphocytes (e.g., IL-2, IL-4, and
interferon-�) and binds to cytoplasmic proteins in lymphocytes.
Although the binding proteins (cytophilins)
for cyclosporine and tacrolimus are different, they share
similar functions in that the cytophilins are important
for the intracellular folding of proteins. It is speculated
that these proteins are important in regulating gene expression
in T lymphocytes and that both drugs somehow
interfere in this process.
Absorption of tacrolimus from the gastrointestinal
(GI) tract is variable. It is extensively metabolized in
the liver and excreted in the urine.As with cyclosporine,
nephrotoxicity is its principal side effect. | | Manufacturing Process | The novel 17-allyl-1,14-dihydroxy-12-[2-(4-hydroxy-3-methoxycyclohexyl)-1-
methylvinyl]-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-
azatrcyclo[22.3.1.04,9]octacos-18-ene-2,3,10,16-tetraone (FR-900506),
substance can be produced by culturing a FR-900506 substance(s)-producing
strain belonging to the genus Streptomyces (e.g. Streptomyces tsukubaensis
No. 9993, FERM BP-927) in a nutrient medium.
Fermentation
A culture medium (160 ml) containing glycerin (1%), corn starch (1%),
glucose (0.5%), cottonseed meal (1%), dried yeast (0.5%), corn steep liquor
(0.5%) and calcium carbonate (0.2%) (adjusted to pH 6.5) was poured into
each of ten 500 ml-Erlenmeyer flasks and sterilized at 120°C for 30 min. A
loopful of slant culture of Streptomyces tsukubaensis No. 9993 was inoculated
to each of the medium and cultured at 30°C for 4 days on a rotary shaker.
The resultant culture was inoculated to a medium containing soluble starch
(5%), peanut powder (0.5%), dried yeast (0.5%), gluten meal (0.5%),
calcium carbonate (0.1%) and Adekanol (deforming agent, Trade Mark, maker
Asasi Denka Co.) (0.1%) (150 liters) in a 200-liter jar-fermentor, which had
been sterilized at 120°C for 20 min in advance, and cultured at 30C for 4 days
under aeration of 150 liters/minutes and agitation of 250 rpm.
Isolation and Purification
The cultured broth thus obtained was filtered with an aid of diatomaseous
earth (5 kg). The mycelial cake was extracted with acetone (50 liters),
yielding 50 liters of the extract. The acetone extract from mycelium and the
filtrate (135 L) were combined and passed through a column of a non-ionic
adsorption resin "Diaion HP-20" (Trade Mark, maker Mitsubishi Chemical
Industries Ltd.) (10 L). After washing with water (30 L) and 50 % aqueous
acetone (30 L), elution was carried out with 75 aqueous acetone. The eluate
(30 liters) was evaporated under reduced pressure to give residual water (2
L). This residue was extracted with ethyl acetate (2 L) three times. The ethyl
acetate extract was concentrated under reduced pressure to give an oily
residue. The oily residue was mixed with twice weight of acidic silica gel
(special silica gel grade 12, maker Fuji Devison Co.), and this mixture was
slurried in ethyl acetate. After evaporating the solvent, the resultant dry
powder was subjected to column chromatography of the same acidic silica gel
(800 ml) which was packed with n-hexane. The column was developed with nhexane
(3 L), a mixture of n-hexane and ethyl acetate (4:1 v/v, 3 L) and
ethyl acetate (3 L). The fractions containing the object compound were
collected and concentrated under reduced pressure to give an oily residue.
The oily residue was dissolved in a mixture of n-hexane and ethyl acetate
(1:1 v/v, 30 ml) and subjected to column chromatography of silica gel (maker
Merck Co., Ltd. 230-400 mesh) (500 ml) packed with the same solvents
system. Elution was carried out with a mixture of n-hexane and ethyl acetate (1:1 v/v, 2 liters and 1:2 v/v, 1.5 L) and ethyl acetate (1.5 L). Fractions
containing the first object compound were collected and concentrated under
reduced pressure to give crude FR-900506 substance (3 g) in the form of
yellowish powder.
This powder of the FR-900506 substance could be transformed into a form of
white crystals by recrystallization thereof from acetonitrile. Melting point:
127°-129°C. | | brand name | Prograf (Astellas); Protopic (Astellas). | | Therapeutic Function | Immunosuppressive | | 一般的な説明 | Immunosuppressant that blocks T-cell proliferation in vitro by inhibiting the generation of several lymphokines, especially IL-2. Shown to inhibit the activity of FK-506 Binding Protein, thereby reversing its effects on sarcoplamic reticulum Ca2+ release. Shown to inhibit the Ca2+-dependent phosphatase, Calcineurin (Cat. No. 539565), as well as Na+-K+-ATPase in nephron segments. Also shown to inhibit aldosterone-induced synthesis of Giα-3 protein. | | Biochem/physiol Actions | Product does not compete with ATP. | | 臨床応用 | A topical formulation (Protopic) has recently been approved
for treatment of moderate to severe atopic dermatitis
in children and adults who have not responded
to other therapies. Levels of systemic absorption are
low even when applied to a relatively large body surface
area. | | 副作用 | Local irritant reactions (burning, stinging, erythema)
are a common side effect, but these usually resolve
within the first few days of treatment. The major
benefit of topical tacrolimus over topical corticosteroids
is that tacrolimus does not cause atrophy, striae, or
telangiectasia, even with chronic use. | | Veterinary Drugs and Treatments | Tacrolimus has recently been studied at the University of Tennessee
College of Veterinary Medicine where investigators found it equally
effective as cyclosporine and effective for cyclosporine-resistant
cases of KCS. It exerts its effects through a mechanism similar to
that of cyclosporine, however exact mechanisms of action in causing
tear production are still being determined. | | Veterinary Drugs and Treatments | Tacrolimus ointment may be of benefit in veterinary patients in the adjunctive treatment of atopic dermatitis, discoid lupus erythematosus,
pemphigus erythematosus or foliaceous, pinnal vascular disease, alopecia areata, vitiligo and for perianal fistulas (terminal phase or
maintenance treatment after cyclosporine therapy). Unlike topical corticosteroids, tacrolimus or pimecrolimus do not have atrophogenic
or metabolic effects associated with long-term or large area treatment.
Tacrolimus acts similarly as cyclosporine, namely inhibiting T-lymphocyte activation primarily by inhibiting the phosphatase activity
of calcineurin. It also inhibits the release of inflammatory cytokines and mediators from mast cells and basophils. | | in vitro | tacrolimus (fk506) has been reported to inhibit the interleukin 2(il-2) production and the response of mixed lymphocyte culture. in addition, tacrolimus (fk506) added to the cell cultures has been revealed to inhibit murine or human mixed lymphocyte reactivity and the generation of cytotoxic t cells. furermor, tacrolimus (fk506) has also been reported to dose-dependently suppress the production of various cytokines including il-2, il-3, il-4, and γ-interferon, in response to different stimulis. besides, tacrolimus has shown its efficacy in the prevention of allograft rejection in animal transplant models. tacrolimus has been found to be significantly efficient in experimental hepatic allografts, and has hepatotrophic properties [1]. | | target | Immunology & Inflammation related | | 薬物相互作用 | Potentially hazardous interactions with other drugs
Ciclosporin: may increase the half-life of ciclosporin
and exacerbate any toxic effects. The two should not
be prescribed concomitantly. Care should be taken
when converting from ciclosporin to tacrolimus.
Tacrolimus levels increased by: amlodipine,
atazanavir, basiliximab, boceprevir, bromocriptine,
chloramphenicol, cimetidine, cortisone, danazol,
dapsone, diltiazem, ergotamine, ethinyloestradiol,
felodipine, fosamprenavir, gestodene, grapefruit
juice, imidazole and triazole antifungals, lidocaine,
lansoprazole, possibly levofloxacin, macrolides,
midazolam, nicardipine, nifedipine, norethisterone,
omeprazole, pantoprazole, posaconazole, ranolazine;
ritonavir, saquinavir, Chinese herbal remedies
containing extracts of Schisandra sphenanthera,
tamoxifen, theophylline, verapamil and voriconazole.
Tacrolimus levels decreased by: carbamazepine,
caspofungin, fosphenytoin, isoniazid, phenobarbital,
phenytoin (fosphenytoin and phenytoin levels
possibly increased), primidone, rifampicin, possibly
rifabutin and St John’s wort.
Increased nephrotoxicity with: aminoglycosides,
amphotericin, NSAIDs, sulfamethoxazole,
trimethoprim and vancomycin.
Increased risk of hyperkalaemia with: potassium�sparing-diuretics and potassium salts.
Anticoagulants: possibly increases concentration of
dabigatran - avoid.
Antipsychotics: avoid with droperidol, increased risk
of ventricular arrhythmias.
Antivirals: increased risk of nephrotoxicity with
acyclovir, ganciclovir, valaciclovir and valganciclovir;
concentration affected by efavirenz; concentration of
both drugs increased with telaprevir; concomitant
use with dasabuvir and ombitasvir/paritaprevir/
ritonavir is not recommended unless the benefits
outweigh the risks, if used concomitantly, tacrolimus
should not be administered on the day dasabuvir
and ombitasvir/paritaprevir/ritonavir are initiated.
Beginning the day after dasabuvir and ombitasvir/
paritaprevir/ritonavir are initiated; reinitiate
tacrolimus at a reduced dose based on tacrolimus
levels. The recommended tacrolimus dosing is 0.5
mg every 7 days, monitor levels at initiation and
throughout treatment.
Clotrimazole: more than doubles the bioavailability
of tacrolimus (US-based researchers report that
concomitant clotrimazole substantially increases
the relative oral bioavailability of tacrolimus in renal
transplant recipients. Inpharma. 2005 Dec 10; 1517:
15).
Cytotoxics: concentration of afatinib possibly
increased - separate dose by 6-12 hours; use
crizotinib with caution; concentration increased by
imatinib. | | 代謝 | Tacrolimus is extensively bound to erythrocytes in the
blood, and variations in red cell binding account for much
of the variability in pharmacokinetics. It is extensively
metabolised in the liver, mainly by cytochrome P450
isoenzyme CYP3A4, and excreted, primarily in bile,
almost entirely as metabolites. Considerable metabolism
also occurs in the intestinal wall.
There are several metabolites identified. Only one of
these has been shown in vitro to have immunosuppressive
activity similar to that of tacrolimus. The other
metabolites have only weak or no immunosuppressive
activity. In systemic circulation only one of the inactive
metabolites is present at low concentrations. Therefore,
metabolites do not contribute to pharmacological activity
of tacrolimus. | | 貯蔵 | -20°C | | 参考文献 | 1) Yu et al. (2006), Effects of cyclosporine A, FK506 and rapamycin on calcineurin phosphatase activity in mouse brain; IUBMB Life, 58 429 |
|