
Acetylsalicylic acid
| Price | $1 |
| Package | 1KG |
| Min. Order: | 1G |
| Supply Ability: | 100KG |
| Update Time: | 2019-07-06 |
Product Details
| Product Name: Acetylsalicylic acid | CAS No.: 50-78-2 |
| Min. Order: 1G | Purity: 98% |
| Supply Ability: 100KG | Release date: 2019/07/06 |
AD68
| Acetylsalicylic acid Basic information |
| What’s acetylsalicylic acid? Chemical property Uses How Aspirin is used in Ischemic Stroke therapy Production method Category Toxicity grading Acute toxicity Flammable hazardous characteristics Handling and storage characteristics Fire extinguishing agent Occupational standard |
| Product Name: | Acetylsalicylic acid |
| Synonyms: | O-ACETYLSALICYLIC ACID;O-ACETOXYBENZOIC ACID;ACETICYL;ACETOXYBENZOIC ACID;ACETYLSALICYLIC ACID;ACETYSALICYLIC ACID;ACETYLSALICYLIC ACID IMPURITY D;ALKENYL SUCCINIC ANHYDRIDES |
| CAS: | 50-78-2 |
| MF: | C9H8O4 |
| MW: | 180.16 |
| EINECS: | 200-064-1 |
| Product Categories: | Pharmaceutical;Aromatics Compounds;Aromatics;Intermediates & Fine Chemicals;Pharmaceuticals;Lipid signaling;Anti-Allergic Agents, Antibiotic;Isotope;LOPROX |
| Mol File: | 50-78-2.mol |
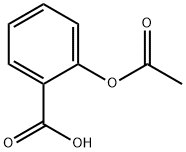 |
|
| Acetylsalicylic acid Chemical Properties |
| Melting point | 134-136 °C(lit.) |
| Boiling point | 272.96°C (rough estimate) |
| density | 1.35 |
| refractive index | 1.4500 (estimate) |
| Fp | 250 °C |
| storage temp. | Store at RT. |
| solubility | H2O: 10 mg/mL at 37 °C |
| form | crystalline |
| color | white |
| pka | 3.5(at 25℃) |
| Water Solubility | 3.3 g/L (20 ºC) |
| Merck | 14,851 |
| BRN | 779271 |
| Stability: | Stable. Keep dry. Incompatible with strong oxidizing agents, strong bases, strong acids, various other compounds such as iodides, iron salts, quinine salts, etc. |
| CAS DataBase Reference | 50-78-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Benzoic acid, 2-(acetyloxy)-(50-78-2) |
| EPA Substance Registry System | Benzoic acid, 2-(acetyloxy)-(50-78-2) |
| Safety Information |
| Hazard Codes | Xn |
| Risk Statements | 22-36/37/38 |
| Safety Statements | 26-36/37/39 |
| RIDADR | UN 1851 |
| WGK Germany | 1 |
| RTECS | VO0700000 |
| TSCA | Yes |
| HazardClass | 6.1 |
| PackingGroup | III |
| HS Code | 29182210 |
| Hazardous Substances Data | 50-78-2(Hazardous Substances Data) |
| Toxicity | LD50 orally in mice, rats: 1.1, 1.5 g/kg (Hart) |
| Acetylsalicylic acid Usage And Synthesis |
| What’s acetylsalicylic acid? | Acetylsalicylic acid, also known as aspirin, is an analgesic-antipyretic medicine made by salicylic acid interacting with acetic anhydride. It is a white crystalline powder, odorless, stable in dry air. It will be slowly hydrolyzed to be salicylic acid and acetic acid in moist air, and aqueous solution has acidic reaction. Slightly soluble in water, soluble in ethanol, ethyl ether, chloroform, sodium hydroxide solution and sodium carbonate solution. Acetylsalicylic acid has antipyretic analgesic, anti-inflammatory and anti-rheumatism effect, that’s why it is often used for fever, headache, muscle pain, neuralgia, rheumatic fever, acute rheumatic arthritis, gout, etc.; also it has antiplatelet aggregation effect, and can be used for prevention of arterial thrombosis, atherosclerosis, transient cerebral ischemia and myocardial infarction; in addition, acetylsalicylic acid also can be used in the treatment of biliary tract roundworm disease and athlete's foot. Pharmacological actions Acetylsalicylic acid is one of the traditional antipyretic analgesics, as well as the role of platelet aggregation. Acetylsalicylic acid in the body has the characteristics of the antithrombotic, can reduce the formation of obstructive blood clots in surrounding arteries, and inhibit release of platelet response and endogenous ADP, 5-HT, etc., therefore to inhibit second phase other than the first phase of platelet aggregation. The mechanism of action of acetylsalicylic acid is to make platelets cyclooxygenase acetylation, thus inhibiting the formation of ring peroxide, and TXA2 formation is also reduced as well. At the mean time make the platelet membrane protein acetylation, and inhibit platelet membrane enzyme, which helps to inhibit platelet function. As the cyclooxygenase is inhibited, it impacts blood vessel wall synthesized to be PGI2, the platelet TXA2 synthetic enzymes also to be inhibited; so it would impact formation of both TXA2 and PGI2 when it is large doses. Suitable for ischemic heart disease, after percutaneous transluminal coronary angioplasty or coronary artery bypass grafting, prevent transient ischemic stroke, myocardial infarction and reduce the incidence of arrhythmia. The above information is edited by the Chemicalbook He Liao Pu. |
| Chemical property | This product is a white crystalline, with melting point 138~140 ℃, insoluble in water, soluble in alcohol, ether, etc. |
| Uses | 1.Acetylsalicylic acid is the raw material for rodenticide intermediates 4-hydroxycoumarin. 2.Used to make outdoor structural members and equipment parts exposed in highlights, such as the automobile body, agricultural machinery parts, meters and electric lamps, road marking, etc. 3.It is the earliest applied, the most popular and the most common antipyretic analgesics anti-rheumatism medicine, has aspects of pharmacological effects as antipyretic-analgesic and anti-inflammatory, anti-platelet aggregation and works quickly and effectively. Overdosage can be easily diagnosed and treated, with rare allergic reactions. Often used to cold fever, headache, neuralgia, joint ache, muscle pain, rheumatic fever, acute wet sex arthritis, rheumatoid arthritis and toothache, etc. Listed in National Essential Medicine List. Acetylsalicylic acid also works as an intermediate of other medicines. |
| How Aspirin is used in Ischemic Stroke therapy | In the therapy of Ischemic Stroke, 50 to 325 mg/day started between 24 and 48 hours after completion of alteplase has also been shown to reduce long-term death and disability. Aspirin, clopidogrel, and extended-release dipyridamole plus aspirin are all considered first-line antiplatelet agents. The combination of aspirin and clopidogrel can only be recommended in patients with ischemic stroke and a recent history of myocardial infarction or coronary stent placement and then only with ultra-low-dose aspirin to minimize bleeding risk. |
| Production method | Acetylation of salicylic acid: add acetic anhydride (feeding ratio is 0.7889 times of the total salicylic acid) in reaction vessel, and then add two thirds of salicylic acid, stir it and temperature rises. React 40-60min in 81-82℃. Cool it to 81-82 ℃ and keep the temperature for 2h. When free salicylic acid is qualified, cool it to 13 ℃, precipitation crystallization, rejection filter, dry it in 65-70℃ air flow, then we get acetylsalicylic acid. |
| Category | Toxic substance |
| Toxicity grading | High toxic |
| Acute toxicity | Oral-rat LD50: 200 mg/kg; Oral-mice LD50: 250 mg/kg |
| Flammable hazardous characteristics | Flammable in fire; irritant gas would be decomposed out when heated. |
| Handling and storage characteristics | Warehouse needs to be ventiIative and dry with low temperature; separate it from oxidant and food additives. |
| Fire extinguishing agent | Water fog, foam, carbon dioxide, sandy soil. |
| Occupational standard | TWA 5 mg/m3 |
| Chemical Properties | White Solid |
| Uses | Analgesic; antipyretic; anti-inflammatory; antithrombotic |
| Uses | antifungal |
| Uses | Axepim Cephalosporin antibiotic |
| Definition | ChEBI: A member of the class of benzoic acids that is salicylic acid in which the hydrogen that is attached to the phenolic hydroxy group has been replaced by an acetoxy group. A non-steroidal anti-inflammatory drug with moA cyclooxygenase inhibitor activity. |
| General Description | Odorless white crystals or crystalline powder with a slightly bitter taste. |
| Air & Water Reactions | Slowly hydrolyzes in moist air. Has been involved in dust cloud explosions. Water insoluble. Solution in water is acid to methyl red indicator. |
| Reactivity Profile | The active ingredient in common aspirin. Incompatible with oxidizers and strong acids. Also incompatible with strong bases. May react with water or nucleophiles (e.g. amines and hydroxy groups). May also react with acetanilide, amidopyrine, phenazone, hexamine, iron salts, phenobarbitone sodium, quinine salts, potassium and sodium iodides, alkali hydroxides, carbonates, stearates and paracetanol. |
| Fire Hazard | Acetylsalicylic acid is combustible. |
Company Profile Introduction
Established in 2014,Career Henan Chemical Co. is a manufacturerspecializing in the sale of fine chemicals.
Mainly deals in the sales of:
Pharmaceutical intermediates
OLED intermediates:
Pharmaceutical intermediates;
OLED intermediates;
You may like
Recommended supplier
| Product name | Price | Suppliers | Update time | |
|---|---|---|---|---|
| $46.00/500mg |
VIP4Y
|
TargetMol Chemicals Inc.
|
2026-04-20 | |
| $46.00/500mg |
VIP6Y
|
TargetMol Chemicals Inc.
|
2026-04-20 | |
| $0.00/1kg |
VIP5Y
|
WUHAN FORTUNA CHEMICAL CO., LTD
|
2025-07-23 | |
| $/ |
VIP6Y
|
RongNa Biotechnology Co.,Ltd
|
2025-04-29 | |
| $46.00/500mg |
VIP2Y
|
TargetMol Chemicals Inc.
|
2025-04-27 | |
| $40.00/1KG |
Shandong Xuhuang New material CoLTD
|
2025-02-21 | ||
| $3.50/1000kg |
Yujiang Chemical (Shandong) Co.,Ltd.
|
2024-10-25 | ||
| $1.00/1KG |
VIP8Y
|
Hebei Chuanghai Biotechnology Co., Ltd
|
2024-10-25 | |
| $2.50/200kg |
VIP2Y
|
Aurora Industry Co., Ltd.
|
2024-09-02 | |
| $1.00/1KG |
VIP5Y
|
Hebei Chuanghai Biotechnology Co,.LTD
|
2024-08-20 |
- Since: 2014-12-17
- Address: Zhengzhou High tech Zone, Henan Province, China
INQUIRY
杨俊青
sales@coreychem.com
sales@coreychem.com





 China
China