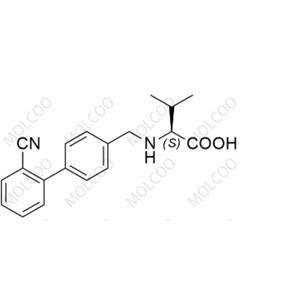
Product Details
| Product Name: Valsartan Impurity | Min. Order: 10mg |
| Purity: 99%+ HPLC | Supply Ability: 1000 |
| Release date: 2025/07/31 |
Valsartan Impurity
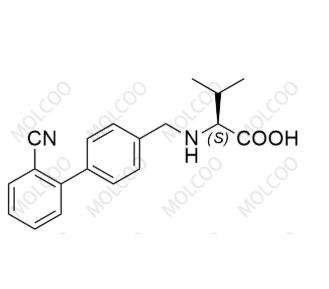
Product Information
Advantages
Applications
Background Description
Research Status
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!
Company Profile Introduction
-
You may like
Recommended supplier
| Product name | Price | Suppliers | Update time | |
|---|---|---|---|---|
| $0.00/1kg |
VIP2Y
|
HANGZHOU HAILAN CHEMICAL CO.,LTD.
|
2025-09-02 | |
| $/ |
VIP5Y
|
RongNa Biotechnology Co.,Ltd
|
2025-04-29 | |
| $5.00/1KG |
VIP5Y
|
Hebei Chuanghai Biotechnology Co,.LTD
|
2024-08-20 | |
| $0.00/10mg |
VIP2Y
|
ShenZhen H&D Pharmaceutical Technology Co., LTD
|
2024-06-14 | |
| $36.00/1kg |
VIP4Y
|
Hebei Dangtong Import and export Co LTD
|
2023-09-19 | |
| $0.00/1kg |
VIP3Y
|
Hangzhou ICH Biofarm Co., Ltd
|
2023-07-12 | |
| $30.00/1Kg |
Xinxiang Hongqi District Houyuan Trading Co.,Ltd
|
2022-10-06 | ||
| $0.00/1kg |
henan kanbei chemical co.,ltd
|
2022-09-22 | ||
| $0.00/1kg |
VIP4Y
|
Henan Aochuang Chemical Co.,Ltd.
|
2022-09-20 | |
| $0.00/25Kg/Drum |
Guangzhou Dihao Medical Technology Co., Ltd
|
2021-09-22 |
INQUIRY












 China
China