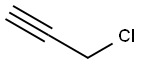
3-Chloropropyne: Physical Properties and Dissociative Adsorption Mechanism on Si(111)-(7 × 7) Surface
Aug 29,2025
3-Chloropropyne is generally a colorless fluid that may appear slightly yellow. It's often found as a clear liquid with a distinct, sharp odor reminiscent of astringent chemicals. At room temperature, 3-Chloropropyne exists in a liquid state. It is a volatile compound prone to evaporation when exposed to air due to its low boiling point. While 3-Chloropropyne displays a degree of solubility in both polar and non-polar solvents, it is particularly notable for its compatibility with non-polar environments. This dual behavior underscores the versatility and importance of understanding solubility in the practical manipulation of chemical compounds.

Dissociative Adsorption of 3-Chloropropyne on Si(111)-(7 × 7)
Functionalization of semiconductor surfaces with covalent attachment of organic molecules has been attracting much attention recently because of its fundamental scientific significance as well as potential applications in chemical and biological sensors and molecular electronics. The Si(111)-(7 × 7) surface reconstruction is perhaps best known as the most complex and scientifically important semiconductor surface, which can serve as a model template for understanding the interaction between organic molecules and surface silicon atoms. Its structure, can be described by the dimer–adatom–stacking-fault (DAS) model. 3-Chloropropyne (Cl—CH2—C≡CH) is an interesting multifunctional organochloride, consisting of the ethyne group (—C≡CH), and the chlorine (Cl) atom. The propargyl radical C3H3 is a prototypical example of a three-center conjugated hydrocarbon, and is thought to be a precursor to benzene and other aromatic hydrocarbons. The photodissociation of propargyl halides attracted considerable attention and has been studied extensively since it can produce propargyl radicals involving C–Br or C–Cl bond fission. Less attention was paid to the study of 3-chloropropyne binding onto the semiconductor surfaces, and it may provide new insight into the reaction mechanism of 3-chloropropyne at surfaces.[1]
It may selectively bind on the Si(111)-(7 × 7) surface through a typically [2 + 2] cycloaddition of the ethyne group with the adjacent adatom–rest atom pair, maintaining its chlorine atom in the addition product. Another possible binding mode is to retain the ethyne group through the cleavage of C–Cl bond. Thus, 3-chloropropyne was chosen as a model to demonstrate the selective attachment of multifunctional molecules on the Si(111)-(7 × 7) surface. The Si(111) samples (20 mm × 8 mm × 0.38 mm) were cut from p-type boron-doped silicon wafers (purity 99.999%, thickness 0.38 mm, resistivity 1–30 Ω cm, Goodfellow). A Ta-sheet resistive heater (thickness 0.025 mm, Goodfellow) was sandwiched between two Si(111) crystals held together using Ta clips, and in turn spot-welded to Ta posts at the bottom of a Dewar-type liquid nitrogen (N2) cooled sample holder. The adsorption of 3-chloropropyne on the Si(111)-(7 × 7) surface was investigated using X-ray photoelectron spectroscopy (XPS), high resolution electron energy loss spectroscopy (HREELS), and density functional theory (DFT) calculations. The experimental and theoretical results clearly suggest that 3-chloropropyne adsorbs on the Si(111)-(7 × 7) surface at room temperature through the cleavage of the C–Cl bond. The covalently bonded organic molecule with the intact —C≡CH bond would serve as a precursor for further photochemical modification and funtionalization.
Vibrational studies were carried out to further support the binding mechanism of 3-chloropropyne on the Si(111)-(7 × 7) surface.The vibratonal frenquencies and their assignments for physisobed and chemisorbed molecules are summarized. Vibrational signatures at 500, 700, 865, 1015, 1260, 1415, 2140, 2990, and 3310 cm–1 can be clearly identified in the spectra of physisorbed molecules. The physisorbed and chemisorbed adsorption of 3-chloropropyne on the Si(111)-(7 × 7) surface was studied by XPS, HREELS and DFT. The experimental and theoretical results showed that 3-chloropropyne dissociatively binds to silicon surface by forming the Si—C linkage with the methyl group and the Si—Cl bond through the cleavage the C—Cl bond. The preserved ethynyl group (—C≡CH) in the binding configuration may be employed as an intermediate for further attachment of other functional molecules.
References
[1]Zhang, Yongping et al. “Dissociative adsorption of 3-chloropropyne on Si(111)-(7 × 7): binding and structure.” Langmuir : the ACS journal of surfaces and colloids vol. 29,6 (2013): 1868-74. doi:10.1021/la304018a
- Related articles
- Related Qustion
Supplementation with pyridoxal 5'-phosphate monohydrate can synthesize neurotransmitters such as dopamine and serotonin, maintaining a healthy nervous system.....
Nov 4,2025Biochemical Engineering2',5'-Dihydroxyacetophenone lowers uric acid, inhibits melanin, induces multiple myeloma cell apoptosis via key pathways.....
Sep 1,2025API3-Chloropropyne
624-65-7You may like
- 3-Chloropropyne
-

- $10.00 / 1KG
- 2025-11-27
- CAS:624-65-7
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10 mt
- propargyl chloride solution
-
- $6.00 / 1KG
- 2025-09-25
- CAS:624-65-7
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- 3-Chloropropyne
-
- $0.00 / 25kg
- 2025-09-23
- CAS:624-65-7
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1000kgs