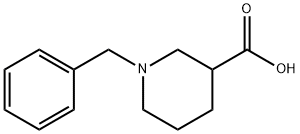
1-BENZYLPIPERIDINE-3-CARBOXYLIC ACID synthesis
- Product Name:1-BENZYLPIPERIDINE-3-CARBOXYLIC ACID
- CAS Number:141943-04-6
- Molecular formula:C13H17NO2
- Molecular Weight:219.28
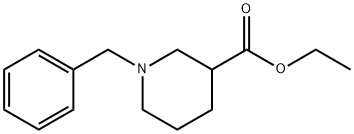
72551-53-2

141943-04-6
The general procedure for the synthesis of 1-benzylpiperidine-3-carboxylic acid from ethyl 1-benzylpiperidine-3-carboxylate was as follows: a 4N aqueous sodium hydroxide solution (15 ml) was slowly added to a mixture of tetrahydrofuran (30 ml) and 1,4-dioxane (30 ml) containing ethyl 1-benzyl-3-piperidinecarboxylate (7.00 g, 28.3 mmol). The reaction mixture was stirred at room temperature for 4 hours. Subsequently, 4N aqueous sodium hydroxide solution (15 ml) was added again and stirring was continued at room temperature overnight. After completion of the reaction, 2N hydrochloric acid (15 ml) was slowly added to neutralize the reaction solution under ice bath cooling conditions. The neutralized mixture was azeotropically dehydrated using toluene. The obtained residue was suspended in ethanol and after filtration, the filtrate was concentrated to finally obtain 1-benzyl-3-piperidine carboxylic acid (6.3 g, 100% yield).

72551-53-2
86 suppliers
$8.00/5g

141943-04-6
54 suppliers
$60.00/250mg
Yield:141943-04-6 100%
Reaction Conditions:
with sodium hydroxide in tetrahydrofuran;1,4-dioxane;water at 20;
Steps:
138.b Example 138; Synthesis of N-(1-benzylpiperidin-3-yl)-1H-indazole-5-carboxamide; (b) Synthesis of 1-benzyl-3-piperidinecarboxylic acid
A 4N-aqueous sodium hydroxide solution (15 ml) was added to a solution of ethyl 1-benzyl-3-piperidinecarboxylate (7.00 g, 28.3 mmol) in a mixture of tetrahydrofuran (30 ml) and 1,4-dioxane (30 ml), and the resulting mixture was stirred at room temperature for 4 hours. After a 4N-aqueous sodium hydroxide solution (15 ml) was added again, the resulting mixture was stirred overnight at room temperature. After completion of the reaction, the reaction solution was neutralized by the addition of 2N-hydrochloric acid (15 ml) under ice-cooling and the resulting mixture was subjected to azeotropic concentration with toluene. The residue was suspended in ethanol, followed by filtration, and the filtrate was concentrated to obtain 1-benzyl-3-piperidinecarboxylic acid (6.3 g, 100%).
References:
EP1403255,2004,A1 Location in patent:Page 66

141943-03-5
0 suppliers
inquiry

141943-04-6
54 suppliers
$60.00/250mg
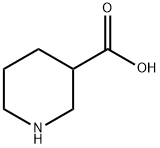
498-95-3
376 suppliers
$5.00/5g
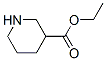
71962-74-8
50 suppliers
inquiry

141943-04-6
54 suppliers
$60.00/250mg
100-44-7
659 suppliers
$13.50/250G

498-95-3
376 suppliers
$5.00/5g

141943-04-6
54 suppliers
$60.00/250mg

100-44-7
659 suppliers
$13.50/250G

141943-04-6
54 suppliers
$60.00/250mg