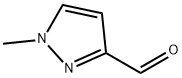
1-METHYL-1H-PYRAZOLE-3-CARBALDEHYDE synthesis
- Product Name:1-METHYL-1H-PYRAZOLE-3-CARBALDEHYDE
- CAS Number:27258-32-8
- Molecular formula:C5H6N2O
- Molecular Weight:110.11
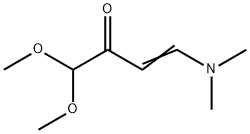
67751-23-9
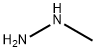
60-34-4

27258-32-8
Methylhydrazine (24 cm3, 19.5 g, 0.423 mol) was slowly added dropwise to 225 cm3 of aqueous solution containing 17.0 g NaOH (0.423 mol) under ice bath cooling conditions. Subsequently, 4-(dimethylamino)-1,1-dimethoxy-3-buten-2-one (73.3 g, 0.423 mol) was added slowly dropwise at room temperature and the reaction mixture was stirred overnight. After completion of the reaction, it was cooled to room temperature, 36% HCl (28.2 g, 0.278 mol) was added and stirring was continued for 1 hour. The reaction mixture was extracted with dichloromethane (4 x 100 cm3), the organic phases were combined and dried over anhydrous sodium sulfate. The solvent was removed by evaporation under reduced pressure and the residue was dissolved in 420 cm3 1M HCl aqueous solution and stirred at room temperature for 3 hours. Subsequently, the pH was adjusted to 10 with 10% K?CO? aqueous solution and extracted with dichloromethane (3 x 200 cm3). The organic layer was dried over anhydrous sodium sulfate and the solvent was evaporated under reduced pressure. The residue was purified by vacuum distillation to give 1-methyl-1H-pyrazole-3-carbaldehyde (10a). Yield 32.8 g (70% yield); colorless liquid; boiling point: 70-72 °C (3 mbar); mass spectrum (ESI): m/z = 111 ([M + H]?) ; 1H NMR (500 MHz, CDCl?): δ = 9.84 (s, 1H), 7.36 (d, J = 1.4 Hz, 1H), 6.68 (d, J = 1.4 Hz, 1H), 3.91 (s, 3H) ppm; 13C NMR (126 MHz, CDCl?): δ = 185.8, 150.9, 131.8, 105.8, 39.2 ppm.
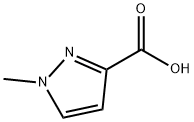
25016-20-0
211 suppliers
$9.00/100mg

27258-32-8
139 suppliers
$22.00/1g
Yield: 63%
Reaction Conditions:
Stage #1:1-methylpyrazole-3-carboxylic acid with thionyl chloride;N,N-dimethyl-formamide for 2 h;Heating / reflux;
Stage #2: with hydrogen;palladium 10% on activated carbon in toluene for 8 h;Heating / reflux;
Steps:
3
A suspension of the acid (90 g, 0.71 mol) and DMF (1 drop) in thionyl chloride (250 mL) was stirred at reflux under nitrogen for 2 h. The solvent was evaporated from the reaction mixture, the residue azeotroped with toluene (3X200 mL), diluted into toluene (250 mL), added to a suspension OF PD-C (10 wt%, 9.3 g) in toluene (500 mL), and the mixture stirred at reflux for 8 h with a gentle flow of hydrogen gas through the suspension. After cooling to room temperature, the suspension was filtered through celite, washed with toluene, and concentrated in vacuo. The residue was fractionally distilled under vacuum to provide the title compound (50 g, 63%) as a low melting white solid (bp = 92 °C @ 8 mmHg)
References:
MERCK & CO., INC. WO2004/58702, 2004, A2 Location in patent:Page 31; 24

67751-23-9
100 suppliers
$25.00/1g

60-34-4
0 suppliers
$51.79/25g

27258-32-8
139 suppliers
$22.00/1g
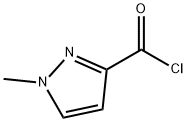
84547-60-4
38 suppliers
$90.00/100mg

27258-32-8
139 suppliers
$22.00/1g
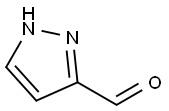
3920-50-1
253 suppliers
$9.00/1g

74-88-4
357 suppliers
$15.00/10g

27258-32-8
139 suppliers
$22.00/1g

67751-23-9
100 suppliers
$25.00/1g

60-34-4
0 suppliers
$51.79/25g

27258-32-8
139 suppliers
$22.00/1g
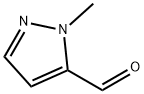
27258-33-9
188 suppliers
$20.00/1g