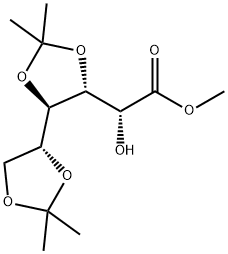
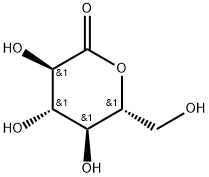
(R)-Methyl 2-hydroxy-2-((4R,4'R,5R)-2,2,2',2'-tetraMethyl-4,4'-bi(1,3-dioxolan)-5-yl) acetate synthesis
- Product Name:(R)-Methyl 2-hydroxy-2-((4R,4'R,5R)-2,2,2',2'-tetraMethyl-4,4'-bi(1,3-dioxolan)-5-yl) acetate
- CAS Number:114743-85-0
- Molecular formula:C13H22O7
- Molecular Weight:290.31
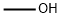
67-56-1
789 suppliers
$7.29/5ml-f
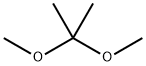
77-76-9
397 suppliers
$9.00/5ml
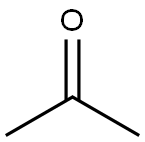
67-64-1
0 suppliers
$17.30/10ml
90-80-2
577 suppliers
$5.00/25g

114743-85-0
16 suppliers
inquiry
Yield: 79%
Reaction Conditions:
with toluene-4-sulfonic acid at 20;
Steps:
3
Example 3: Synthesis of 3-epicasuarine; Experimental; Methyl 3,4:5,6-di-O-isopropylidene-D-qluconate 2; Compound 2 was prepared according to a literature procedure (Regeling ef al. (1987) Reel. Trav. Chim. Pays-Bas (106) :461). D-glucono-1,5-lactone 1 (10.0 g, 56 mmol)was suspended in 2,2-dimethoxypropane (20 ml), MeOH (2 ml) and acetone (6 ml). After addition of para-toluenesulphonic acid (150 mg), the mixture was stirred at room temperature overnight under a nitrogen atmosphere. Then, solid NaHC03 (1.0 g) was added to the clear yellow solution and the resulting mixture was filtered over celite. After evaporation of the solvents In vacuo, the residue was redissolved in DCM (100 ml) and washed with water (100 ml). The organic layer was subsequently dried on Na2S04. Column chromatography (EtOAc/cyclohexane 1/3 v/v) afforded the product as a clear, colourless oil (12.80 g, 79%). Rf 0.71 (EtOAc/cyclohexane 1/1 v/v); [a]D -2.0 (c, 0.72 in CHCI3) [Lit. -1.7 (c, 1.18 in CHCI3)5]; 5H (400 MHz, CDCI3) 1.38, 1.40, 1.42, 1.47 (12H, 4xs, C(CH3)2), 3.08 (1H, d, OH, J 9.1 Hz), 3.88 (3H, s, OCH3), 4.03 (1H, dd, H-6', J 3.4 Hz, 8.2 Hz), 4.10 (1H, m, H-4), 4.14 (1H, m, H-5), 4.19 (1H, dd, H-6, J 5.8 Hz, 8.2 Hz), 4.26 (1H, dd, H-3, J 1.6 Hz, 7.6 Hz), 4.38 (1H, dd, H-2, J 1.6 Hz, 9.2 Hz); 5c (100 MHz, CDCI3) 25.7, 27.0, 27.1, 27.6 (C(CH3)2), 53.1 (OCH3), 68.3 (C-6), 69.9 (C-2), 76.9 (C-4), 77.5 (C-5), 81.3 (C-3), 110.3, 110.5 (C(CH3)2), 173.4 (C=0); MS m/z (ESI +ve) 310.95 (M+Na+).; The opening stages of the synthesis of 3-epicasuarine are outlined in Scheme 1. The first step was the ring opening and concomitant acetonide protection of D-glucono-8-lactone 1 according to a known procedure1. Thus, treatment of 1 with 2,2-dimethoxypropane in a mixture of acetone and methanol in the presence of a catalytic amount of para-toluenesulphonic acid afforded ester 2 in 79% yield. Then, the required nitrogen centre was introduced by nucleophilic displacement of triflate ester 3 which was formed by reaction of 2 with trifluoromethanesulphonic anhydride in the presence of pyridine. The substitution reaction with sodium azide in DMF to yield azide 4 proceeded with near quantitative yield.
References:
MNL PHARMA LIMITED WO2006/8493, 2006, A1 Location in patent:Page/Page column 27-28; 34-35
67-56-1
789 suppliers
$7.29/5ml-f

77-76-9
397 suppliers
$9.00/5ml

90-80-2
577 suppliers
$5.00/25g

114743-85-0
16 suppliers
inquiry

77-76-9
397 suppliers
$9.00/5ml

90-80-2
577 suppliers
$5.00/25g

114743-85-0
16 suppliers
inquiry

134639-65-9
6 suppliers
inquiry
67-56-1
789 suppliers
$7.29/5ml-f

77-76-9
397 suppliers
$9.00/5ml

90-80-2
577 suppliers
$5.00/25g

114743-85-0
16 suppliers
inquiry

134639-65-9
6 suppliers
inquiry