
Ethyl 4-iodo-1-(4-methoxybenzyl)-1H-pyrazole-3-carboxylate synthesis
- Product Name:Ethyl 4-iodo-1-(4-methoxybenzyl)-1H-pyrazole-3-carboxylate
- CAS Number:1355249-29-4
- Molecular formula:C14H15IN2O3
- Molecular Weight:386.18
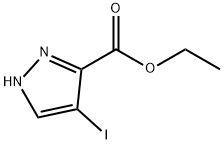
179692-08-1
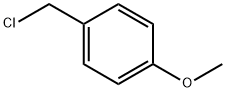
824-94-2

1355249-29-4
To a solution of ethyl 4-iodo-1H-pyrazole-3-carboxylate (38 g, 143 mmol) in acetonitrile (380 mL) was added potassium carbonate (39.5 g, 286 mmol) followed by 1-(chloromethyl)-4-methoxybenzene (24 mL, 177 mmol) at room temperature. The reaction mixture was stirred at 60 °C overnight. After completion of the reaction, the mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to afford ethyl 4-iodo-1-(4-methoxybenzyl)-1H-pyrazole-3-carboxylate (32 g, 58% yield). The compound was characterized by proton nuclear magnetic resonance (1H NMR) and mass spectrometry (MS). 1H NMR (DMSO-d6, 400 MHz) data were as follows: δ 8.13 (s, 1H), 7.23 (d, J = 8.8 Hz, 2H), 6.90 (d, J = 8.8 Hz, 2H), 5.29 (s, 2H), 4.24 (q, J = 6.8 Hz, 2H), 3.71 (s, 3H), 1.26 (t, J = 6.8 Hz, 3H). Mass spectrometry (ESI) showed the molecular ion peak m/z 387 ([M + H]+).

179692-08-1
73 suppliers
$13.00/250mg

824-94-2
445 suppliers
$24.19/5G

1355249-29-4
26 suppliers
$65.00/100mg
Yield:1355249-29-4 58%
Reaction Conditions:
with potassium carbonate in acetonitrile at 60;
Steps:
15.C
Example 15, Step C [00154] To a solution of compound 15c (38 g, 143 mmol) in acetonitrile (380 mL) at r.t. was added K2C03 (39.5 g, 286 mmol) and then PMBCI (24 mL, 177 mmol). The reaction mixture was stirred at 60°C overnight. After cooling to r.t., the mixture was filtered and concentrated under reduced pressure. The residue was purified by chromatography on silica gel to afford compound 15d (32 g, 58%).[00155] This compound was characterized by proton NMR (1HNMR) and mass spectroscopy (MS) in accordance with the procedures described herein. Proton NMR yielded the following results: 1H NMR (DMSO-d6, 400MHz): δ 8.13(s, 1 H), 7.23(d, J = 8.8 Hz, 2H), 6.90(d, J = 8.8 Hz, 2H), 5.29(s, 2H), 4.24 (q, J = 6.8 Hz, 2H), 3.71 (s, 3H); 1.26(t, J = 6.8 Hz, 3H). Mass spectroscopy indicated MS (ESI): m/z 387 (M+H)+.
References:
WO2012/6760,2012,A1 Location in patent:Page/Page column 47-48

179692-08-1
73 suppliers
$13.00/250mg

824-94-2
445 suppliers
$24.19/5G

1355249-29-4
26 suppliers
$65.00/100mg
![1H-Pyrazole-5-carboxylic acid, 4-iodo-1-[(4-methoxyphenyl)methyl]-, ethyl ester](/CAS/20210305/GIF/1355336-09-2.gif)
1355336-09-2
3 suppliers
inquiry

179692-08-1
73 suppliers
$13.00/250mg

824-94-2
445 suppliers
$24.19/5G

1355249-29-4
26 suppliers
$65.00/100mg
![1H-Pyrazole-5-carboxylic acid, 4-iodo-1-[(4-methoxyphenyl)methyl]-, ethyl ester](/CAS/20210305/GIF/1355336-09-2.gif)
1355336-09-2
3 suppliers
inquiry
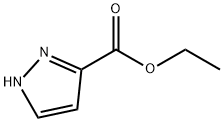
5932-27-4
208 suppliers
$16.18/1G

1355249-29-4
26 suppliers
$65.00/100mg
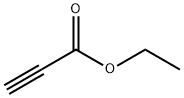
623-47-2
349 suppliers
$10.00/1g

1355249-29-4
26 suppliers
$65.00/100mg