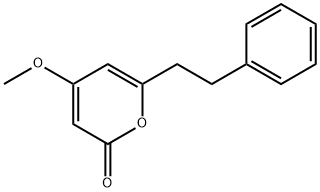
4-HYDROXY-6-PHENETHYL-PYRAN-2-ONE synthesis
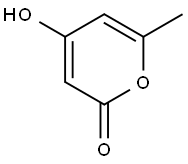
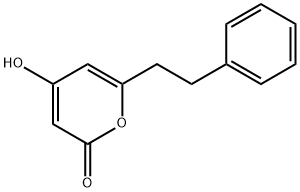
- Product Name:4-HYDROXY-6-PHENETHYL-PYRAN-2-ONE
- CAS Number:33253-32-6
- Molecular formula:C13H12O3
- Molecular Weight:216.23
Yield:33253-32-6 40%
Reaction Conditions:
Stage #1: 4-hydroxy-6-methyl-2-pyronwith 1,1,1,3,3,3-hexamethyl-disilazane at 80; for 1 h;Inert atmosphere;
Stage #2: with n-butyllithium in tetrahydrofuran;hexane at -78; for 1.25 h;Inert atmosphere;
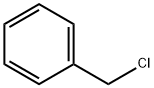
Stage #3: benzyl bromide in tetrahydrofuran;hexane at -78 - 20; for 16 h;Inert atmosphere;
Steps:
General Procedure 3: Lithiation/alkylations of 4-hydroxy-6-methyl-2-pyrone
General procedure: 4-Hydroxy-6-methyl-2-pyrone 3a (1 mmol, 1 eq.) was heated to 80 °C under nitrogen in HMDS (3 mL) for 1 hour. The solution was allowed to cool and the HMDS removed under vacuum. THF (3 mL) was then added and the solution cooled to -78 °C at which point n-BuLi (2.5 M in hexanes, 1.25 mmol, 1.25 eq.) was added carefully over 15 minutes, and the solution stirred for 1 hour. The alkyl halide (1.7-2.3 mmol, 1.7-2.3 eq.) was then added over 10 minutes and the solution allowed to warm gradually to 20 °C and stirred for 16 hours. The reaction was then quenched with 6 M aq. HCl until the pH ~ 2 and the solvent removed in vacuo. The residue was taken up in ethyl acetate (5 mL), washed twice in brine, dried over MgSO4, filtered and concentrated in vacuo to afford a crude brown residue which was purified by flash column chromatography (3 % MeOH in dichloromethane).
References:
Burns, Michael J.;Ronson, Thomas O.;Taylor, Richard J. K.;Fairlamb, Ian J. S. [Beilstein Journal of Organic Chemistry,2014,vol. 10,p. 1159 - 1165] Location in patent:supporting information
100-39-0
434 suppliers
$10.00/10g

33253-32-6
12 suppliers
inquiry
3155-51-9
19 suppliers
$185.00/1mg

33253-32-6
12 suppliers
inquiry
2021-28-5
267 suppliers
$33.00/25g

33253-32-6
12 suppliers
inquiry