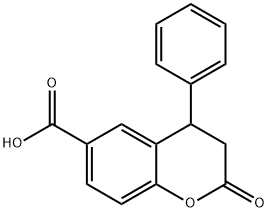
6-CARBOXYL-4-PHENYL-3,4-DIHYDROCOUMARIN synthesis
- Product Name:6-CARBOXYL-4-PHENYL-3,4-DIHYDROCOUMARIN
- CAS Number:356782-33-7
- Molecular formula:C16H12O4
- Molecular Weight:268.26
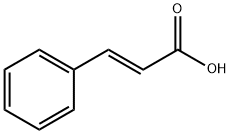
140-10-3
643 suppliers
$5.00/10g
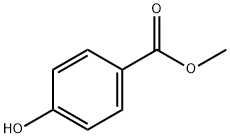
99-76-3
813 suppliers
$5.00/25g

356782-33-7
17 suppliers
$109.90/5mg
Yield:356782-33-7 78%
Reaction Conditions:
with sulfuric acid;acetic acid in water at 100; for 18 h;
Steps:
1 1.
1. (R,S)-4-Phenyl-2-chromanone-6-carboxylic acid (Formula 2a) A mixture of cinnamic acid (100 g, 0.68 mol), methyl 4-hydroxybenzoate (108 g, 0.71 mol) and acetic acid (80 ml) is heated to 100° C. 80 ml of 96% sulfuric acid are then added to the resulting clear solution, while stirring. After 2 hours, crystal formation begins. Stirring is continued at the same temperature for 16 hours, the mixture is cooled to ambient temperature and diluted with 500 ml water. The precipitated crystal mass is filtered off, washed with diethyl ether and dried in vacuo. Crude yield: 142 g (78% of theory), pale beige crystals. Mp 246° C. 1H-NMR (DMSO-d6): 3.18 (d, 2H, J=6.6 Hz, CH2), 4.62 (t, 1H, J=6.6 Hz, CH), 7.14-7.43 (m, 6H), 7.62 (s, 1 H), 7.90 (d, 1 H, J=8.6 Hz). 13C-NMR (DMSO-d6): 35.93, 39.26,117.20,126.81, 127.13,127.65,127.70,129.24, 129.95, 130.25, 140.91, 154.80, 166.66,167.30
References:
US2003/212292,2003,A1 Location in patent:Page/Page column 5
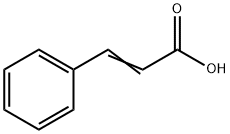
621-82-9
377 suppliers
$29.12/100G
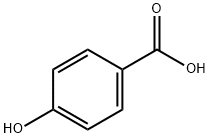
99-96-7
823 suppliers
$5.00/10g

356782-33-7
17 suppliers
$109.90/5mg