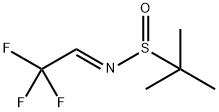
(S,E)-2-Methyl-N-(2,2,2-trifluoroethylidene)propane-2-sulfinaMide synthesis
- Product Name:(S,E)-2-Methyl-N-(2,2,2-trifluoroethylidene)propane-2-sulfinaMide
- CAS Number:929642-48-8
- Molecular formula:C6H10F3NOS
- Molecular Weight:201.21
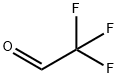
75-90-1
43 suppliers
inquiry
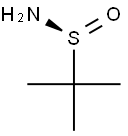
343338-28-3
378 suppliers
$4.00/1g

929642-48-8
27 suppliers
$90.00/250mg
Yield:929642-48-8 88%
Reaction Conditions:
in toluene at 20; for 48 h;
Steps:
(S,E)-N-(2,2,2-Trifluoroethylidene)-tert-butylsulfinamide (5k)
The preparation of 2,2,2-trifluoroacetaldehyde was carried out as describedin the dissertation of Gerhard Greier [34]: Fluoral hydrate (75 %,55.9 mmol) was placed under argon atmosphere in a closed vessel, which islinked by a Claisen condenser to a vessel with molecular sieves (3 ?), cooled to -78 °C.Concentrated sulfuric acid (10 mL) was added dropwise to the fluoral hydrate (6 mL) viadropping funnel and the solution was heated to 70 °C. The developing2,2,2-trifluoroacetaldehyde was condensed in the cooled flask, where its mass was determined(2.08 g, 21.2 mmol).To the condensed 2,2,2-trifluoroacetaldehyde, an equimolar amount of (S)-tert-butylsulfinamide (S)-1 (2.57 g, 21.2 mmol) was added in one portion. The mixture was dilutedwith dry toluene (20 mL) and stirred for 48 h at rt. The suspension was filtered under argonatmosphere through a frit and the solution of imine 5k was stored under argon atmosphere forsubsequent reactions.Imine 5k is formed as intermediate without isolation, as first described by Truong, Menardand Dion [35].
References:
Wünsch, Matthias;Schr?der, David;Fr?hr, Tanja;Teichmann, Lisa;Hedwig, Sebastian;Janson, Nils;Belu, Clara;Simon, Jasmin;Heidemeyer, Shari;Holtkamp, Philipp;Rudlof, Jens;Klemme, Lennard;Hinzmann, Alessa;Neumann, Beate;Stammler, Hans-Georg;Sewald, Norbert [Beilstein Journal of Organic Chemistry,2017,vol. 13,p. 2428 - 2441] Location in patent:supporting information
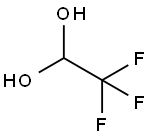
421-53-4
139 suppliers
$15.00/1g

343338-28-3
378 suppliers
$4.00/1g

929642-48-8
27 suppliers
$90.00/250mg