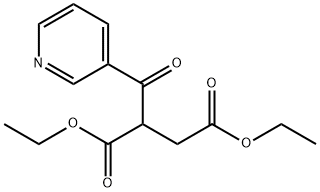
Ethyl -Ethoxycarbonyl--oxo-3-pyridinebutyrate synthesis
- Product Name:Ethyl -Ethoxycarbonyl--oxo-3-pyridinebutyrate
- CAS Number:54109-95-4
- Molecular formula:C14H17NO5
- Molecular Weight:279.29
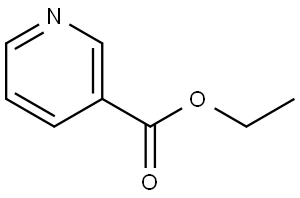
614-18-6
441 suppliers
$5.00/10g
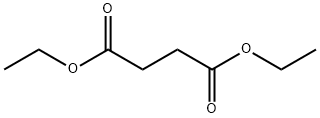
123-25-1
443 suppliers
$5.00/25g

54109-95-4
6 suppliers
$140.00/10mg
Yield:54109-95-4 43%
Reaction Conditions:
with sodium hydride in diethyl ether at 20; for 48 h;
Steps:
A.7.a
Ethyl nicotinate (10 mL, 73 mmol) is dissolved in ether (50 mL). Diethyl succinate (24 mL, 144 mmol) is added, followed by NaH (7.00 g, 60% in oil, 175 mmol) in small spatula loads. The reaction medium is stirred at room temperature in an argon atmosphere for 2 days. It is then pouredcrushed ice. The aqueous phase is collected and treated with 6N HCl in order to bring the pH to 4.5. The product is extracted with chloroform and dried using Na2SO4. After evaporation of the solvents, the crude oil obtained is purified by silica gel column chromatography, with AcOEt/Hexane 1/2 and then 1/1 and 7/3 as eluent. thick orange oil MM(C14H17NO5): 279.29 g mass: 8.87 g Yield: 43% {circle(1)}RMN(1H, 300 MHz, CDCl3): 9, 25(d, 1H, CHarom., J4 = 2, 0 Hz); 8, 81(dd, 1H, CHarom., J 3 = 4, 8 Hz et J4 = 2, 0 Hz); 8, 33-8, 29(M, 1H, CHarom.); 7, 47-7, 43(m, 1H, CHarom.); 4, 82(m, 1H, -CH-); 4, 18-4, 09(m, 4H, 2CH2); 3, 24-2, 99(m, 2H, CH2-COO-); 1, 28-1, 13(m, 6H, 2-CH3).
References:
US2005/113366,2005,A1 Location in patent:Page/Page column 7-8
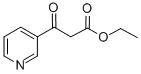
6283-81-4
81 suppliers
$49.00/100mg
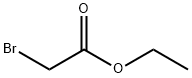
105-36-2
347 suppliers
$5.00/5G

54109-95-4
6 suppliers
$140.00/10mg