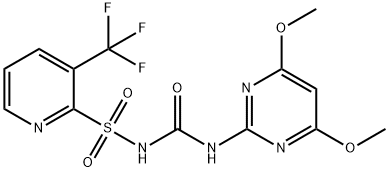
Flazasulfuron synthesis
- Product Name:Flazasulfuron
- CAS Number:104040-78-0
- Molecular formula:C13H12F3N5O5S
- Molecular Weight:407.33
89392-03-0
214 suppliers
$6.00/10g
104040-76-8
21 suppliers
inquiry

104040-78-0
157 suppliers
$155.00/50mg
Yield:-
Reaction Conditions:
with potassium carbonate for 1 h;Reflux;
Steps:
13.5
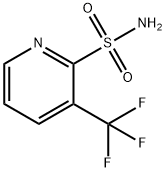
To 1/3 of the reaction solution obtained in the above step (2), 32.3 g of 3-trifluoromethylpyridine 2-sulfonamide and 20.7 g of potassium carbonate as fine powder were introduced, followed by heating and refluxing for one hour. To the reaction mixture, water was introduced, and the organic layer was removed by liquid separation. To this organic layer, water was added, followed by stirring, and the mixture was left to stand still, whereupon the organic layer was again removed by liquid separation. The obtained aqueous layers were put together, and concentrated hydrochloric acid was dropwise added to acidify the aqueous layer thereby to precipitate crystals. The precipitated crystals were collected by filtration and dried to obtain 51.2 g of 1-(4,6-dimethoxypyrimidin-2-yl)-3-[(3-trifluoromethylpyridin-2-yl)sulfonyl]urea. The crude yield thereof was 84% based on 2-amino-4,6-dimethoxypyrimidine. 1 H-NMR (400 MHz, CDCl3 ) δ ppm, 8.78 (d, J=4.4 Hz, 1H), 8.24 (d, J=8Hz, 1H), 7.66 (dd, J=8.0, 4.5 Hz, 1H), 5.81 (s, 1H), 3.99 (s, 1H), LC/MS m/z (FAB+) = 408
References:
ISHIHARA SANGYO KAISHA, LTD. EP2264023, 2010, A1 Location in patent:Page/Page column 20