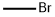
Furaneol synthesis
- Product Name:Furaneol
- CAS Number:3658-77-3
- Molecular formula:C6H8O3
- Molecular Weight:128.13
In another process, a dialkyl ??-methyldiglycolate (formed from an alkyl lactate and an alkyl monochloroacetate) is reacted with dialkyl oxalate in the presence of a sodium alkoxide and dimethylformamide. The reaction product is cyclized, alkylated, hydrolyzed, and decarboxylated.In another process, a dialkyl ??-methyldiglycolate (formed from an alkyl lactate and an alkyl monochloroacetate) is reacted with dialkyl oxalate in the presence of a sodium alkoxide and dimethylformamide. The reaction product is cyclized, alkylated, hydrolyzed, and decarboxylated .
Yield:3658-77-3 81.4%
Reaction Conditions:
with hydrogenchloride;sodium hydroxide in water;
Steps:
1 EXAMPLE 1
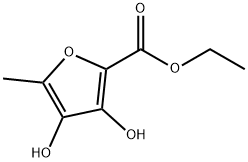
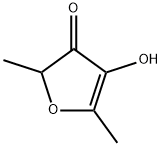
EXAMPLE 1 A solution of 48 g of sodium hydroxide and 37.2 g of ethyl 5-methyl-3,4-dihydroxy-2-furoate of 98.4% purity in 225 ml of water was allowed to stand at room temperature for 40 hours. The mixture was diluted with 200 g of water and 22.8 g of gaseous methyl bromide was added with stirring over a period of one hour at room temperature followed by an additional stirring period of two hours. The pH of the solution was adjusted to 5 by the addition of concentrated hydrochloric acid solution. Extraction with methyl t-butyl ether yielded 21.5 9 of 2,5-dimethyl-4-hydroxy-3[2H] furanone of 95.3% purity (molar yield 81.4%).
References:
US5412121,1995,A
3615-41-6
245 suppliers
$8.00/10g

3658-77-3
460 suppliers
$6.00/100mg
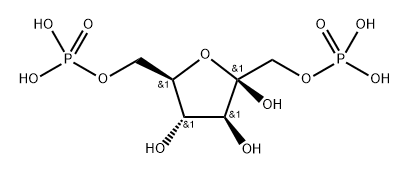
34693-15-7
7 suppliers
inquiry

3658-77-3
460 suppliers
$6.00/100mg
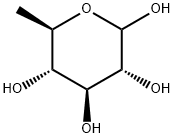
488-79-9
0 suppliers
inquiry

3658-77-3
460 suppliers
$6.00/100mg
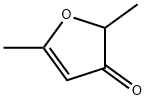
14400-67-0
186 suppliers
$6.00/1g

3658-77-3
460 suppliers
$6.00/100mg