Ipriflavone synthesis
- Product Name:Ipriflavone
- CAS Number:35212-22-7
- Molecular formula:C18H16O3
- Molecular Weight:280.32
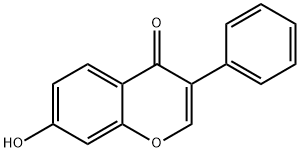
13057-72-2
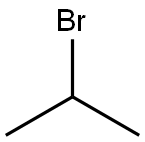
75-26-3

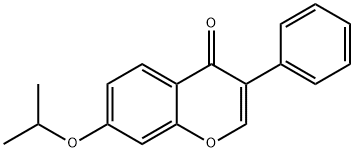
35212-22-7
General procedure for the synthesis of epoxyflavone from 7-hydroxy-3-phenyl-4H-benzopyran-4-one and 2-bromopropane: To a 30 ml DMF solution of 2.50 g of 7-hydroxy-3-phenyl-4H-benzopyran-4-one (10.5 mmol) was added 1.80 g of anhydrous K2CO3 (13.0 mmol) and 3.0 ml of 2-bromopropane ( 32.0 mmol). The reaction mixture was stirred at 80 °C for 4 h. After completion of the reaction, the mixture was poured into ice water. The precipitate was collected by filtration and washed with a small amount of water and dried to give the crude product. The crude product was purified by Sephadex LH-20 column chromatography (eluent: chloroform:methanol = 7:3), and the fraction containing the target product was collected, concentrated, and recrystallized from acetone to give 1.05 g of epsilon-flavonoid crystals with a yield of 42% (w/w) and a melting point of 110-111 °C. The product structure was determined by 1H NMR. The structure of the product was confirmed by 1H NMR, 13C NMR and mass spectrometry (MS).1H NMR (DMSO-d6) δ: 1.33 (-CH3), 1.34 (-CH3), 4.86 (m, 1H, >CH2-), 7.05 (dd, 1H, J = 8.86, 2.20 Hz, 6-H), 7.16 (d, 1H, J = 2.50 Hz, 8-H), 7.38 (t, 1H, J = 6.78 Hz, 4'-H), 7.44 (t, 2H, J = 7.76, 1.6 Hz, 3',5'-H), 7.59 (d, 2H, J = 8.09, 1.6 Hz, 2',6'-H), 8.22 (d, 1H, J = 8.86 Hz, 5-H), 8.46 (s, 1H 128.1 (C-3',5'), 128.9 (C-2',6'), 132.0 (C-4'), 154.0 (C-2), 157.5 (C-9), 162.0 (C-7), 174.1 (C-4). , 279.5 (M-H)-. Elemental analysis (C18H16O3) calculated values: C, 77.12; H, 5.75. measured values: C, 76.50; H, 5.69.

13057-72-2
164 suppliers
$62.79/1g

75-26-3
471 suppliers
$10.00/10g
15892-23-6
39 suppliers
inquiry

35212-22-7
384 suppliers
$27.00/1g
Yield:35212-22-7 96.6%
Reaction Conditions:
with potassium carbonate in N-methyl-acetamide;ethanol;water;
Steps:
1.D Preparation of Ipriflavone (I)
EXAMPLE 1D Preparation of Ipriflavone (I) 7-Hydroxyisoflavone (35 kg), dimethylformamide (35 kg), potassium carbonate (35 kg) and isopropyl bromide (25 kg) were fed to a reactor. The reaction mass was heated to 70° C. for 6 h and added, at said temperature, with sec-butyl alcohol (90 kg) and water (300 kg). Then it was cooled. The solid was filtered and washed with water. The wet solid was dissolved in ethanol (200 kg) at boiling. The solution was cooled and the solid precipitate was filtered, washed with ethanol and dried at 70° C. 39.7 kg of 7-isopropoxy-isoflavone was obtained. Stoichiometric yield =96.6%. Chromatographic purity: (HPLC) ≥99.9%.
References:
US5973169,1999,A

13057-72-2
164 suppliers
$62.79/1g

75-26-3
471 suppliers
$10.00/10g

35212-22-7
384 suppliers
$27.00/1g
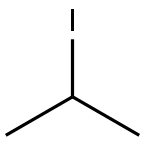
75-30-9
277 suppliers
$15.00/1g

13057-72-2
164 suppliers
$62.79/1g

35212-22-7
384 suppliers
$27.00/1g

1092975-69-3
0 suppliers
inquiry

35212-22-7
384 suppliers
$27.00/1g
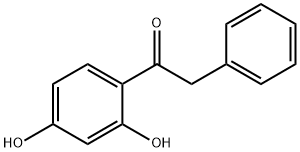
3669-41-8
112 suppliers
$36.89/1g

35212-22-7
384 suppliers
$27.00/1g