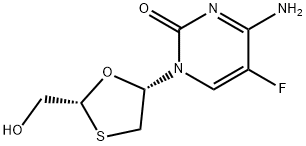
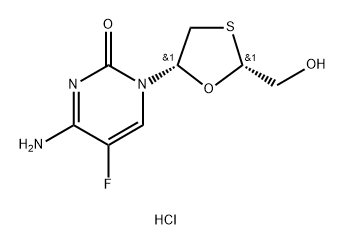
Emtricitabine
- CAS No.
- 143491-57-0
- Chemical Name:
- Emtricitabine
- Synonyms
- FUMITREMORGIN C;EMTRICITABIN;EMtriva;FTC;EMTRITABINE;SM-Q;BW1592;CS-895;BW 1592;BW-1592
- CBNumber:
- CB5266199
- Molecular Formula:
- C8H10FN3O3S
- Molecular Weight:
- 247.25
- MDL Number:
- MFCD00870151
- MOL File:
- 143491-57-0.mol
| Melting point | 136-140°C |
|---|---|
| alpha | D25 -133.60° (c = 0.23 in MeOH) |
| Boiling point | 443.3±55.0 °C(Predicted) |
| Density | 1.82±0.1 g/cm3(Predicted) |
| storage temp. | Keep in dark place,Inert atmosphere,2-8°C |
| solubility | Chloroform (Slightly) |
| form | Solid |
| pka | 13.83±0.10(Predicted) |
| color | White to Pale Yellow |
| λmax | 280nm(H2O)(lit.) |
| Merck | 14,3565 |
| BCS Class | 1 |
| InChIKey | XQSPYNMVSIKCOC-NTSWFWBYSA-N |
| CAS DataBase Reference | 143491-57-0(CAS DataBase Reference) |
| FDA UNII | G70B4ETF4S |
| NCI Drug Dictionary | emtricitabine |
| ATC code | J05AF09 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302-H315-H319-H335 |
| Precautionary statements | P261-P305+P351+P338 |
| RTECS | UW7360500 |
| HS Code | 2934990002 |
Emtricitabine price More Price(51)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 1235106 | Emtricitabine United States Pharmacopeia (USP) Reference Standard | 143491-57-0 | 200MG | $1420 | 2024-03-01 | Buy |
| TCI Chemical | E1007 | Emtricitabine >98.0%(HPLC)(T) | 143491-57-0 | 25mg | $55 | 2024-03-01 | Buy |
| TCI Chemical | E1007 | Emtricitabine >98.0%(HPLC)(T) | 143491-57-0 | 250mg | $308 | 2024-03-01 | Buy |
| Cayman Chemical | 16879 | Emtricitabine ≥98% | 143491-57-0 | 25mg | $34 | 2024-03-01 | Buy |
| Cayman Chemical | 16879 | Emtricitabine ≥98% | 143491-57-0 | 50mg | $62 | 2024-03-01 | Buy |
Emtricitabine Chemical Properties,Uses,Production
Description
Emtricitabine is a nucleoside reverse transcriptaseinhibitor (NRTI) which has been marketed for the prevention and treatment of HIV infection. It also exhibits clinical activity against the hepatitis B virus (HBV) (not yet approved by FDA).Emtricitabine is a synthetic nucleoside analogue of cytidine. It can be phosphorylated by cellular enzymes to form emtricitabine 5'-triphosphate, which competes with the natural substrate deoxycytidine 5'-triphosphate and incorporates into nascent viral DNA, causing early chain termination. Through doing this, it not only lowers the amount of HIV, but also indirectly increases the number of immune system cells (called T cells or CD4+ T-cells).
New anti-HIV drugs
Emtricitabine was a novel nucleoside reverse transcriptase inhibitor successfully developed by Gilead Sciences, Inc., belonging to an anti-viral drugs and exhibits antiviral activity on HIV-1, HIV-2 and HBV. Its antiviral activity exhibits that it can specifically anti-HIV-1, HIV-2 and HBV while even with its drug concentration being raised to 100 mmol/L, it still exhibit no activity against HSV-1, HSV-2, HCMV, VZV, corona, yellow fever virus, respiratory syncytial virus, Rota, and influenza virus or rhinovirus. Even at a concentration lower than micromolar, the drug still exhibits potent inhibitor effect against the LAV strain and IIIB 1 of HIV virus and the ROD2 strain and ZY virus strain of HIV-2 with an IC50 value lower being 95 times lower than that of AZT (zidovudine).
It has been increasingly become the focus of the assessment of new drugs that whether new anti-HIV drug will produce cross-resistance to other kinds of antiviral resistant strains such as AZT, etc. The cross-resistance test of emtricitabine against AZT-resistant strains has demonstrated that although IC50 values ??for these resistant strains have been increased slightly, all kinds of virus strains are still relatively sensitive to emtricitabine. Instead, emtricitabine-resistant viral strains will exhibit significant cross-resistance to antiviral 3TC (lamivudine), but are still sensitive to AZT. Studies have shown that the drug can be used together with AZT with synergistic effect.
Emtricitabine exhibited strong antiviral activity in HBV generating cell lines HepG22.2.15 (PSA subspecies). It can dose-dependently reduce the number of extracellular and intracellular HBVDNA with the IC50 value being 10nmol/L; this value was comparable to another kind of HBV replication inhibitor 3TC. Interferon α nl (Wellferon), α 2a (Ro feron), α 2b (Intron A) can all the production of inhibit HBV viral particles and the accumulation of the intracellular replication virus in the chronic producing cells intracellular production. Combination of them associated with emtricitabine can produce a synergistic effect.
The in vivo anti-hepatitis activity of emtricitabine is investigated through the anti-HBV action in a kind of chimeric mouse model as well as the action of anti-woodchuck hepatitis virus (WHV) function in natural infected prairie dogs. In the mouse model, use different doses for oral administration of this drug to mouse of immune deficiency and carrying human tumors as well as producing human HBV, with the antibody capture/PcR experiment, it was found that the blood HBVDNA (Dane particle) was reduced in a dose-dependent manner with even a low dose of 3.5 mg/(kg • d) also being able to produce significant inhibition while the level of intracellular replicated HBVDNA also exhibited a dose-dependent decrease; however, even at the highest dose (89 mg/(kg • d )), the tumor size remained unchanged, suggesting that the drug has selective anti-HBV activity.
The above information is edited by the Chemicalbook of Dai Xiongfeng.
Pharmacological effects
Emtricitabine belongs to chemically synthesized nucleoside cytosine. Its mechanism of anti-HIV-1 is through multi-step in vivo phosphorylation generating active triphosphate which competitively inhibits the HIV-1 reverse transcriptase while competing with natural 5-phosphate cytosine by for penetrating into the viral DNA synthesis process, which ultimately leads to the synthesis of a DNA strand break. Its mechanism of HBV is because that the HBV replication process contains target site of emtricitabine, namely reverse transcription process. It has a low inhibitory effect on mammal DNA polymerases α, β, ε and mitochondrial DNA polymerase γ.
In vitro antiviral activity: in vitro laboratory used and clinical isolate anti-HIV viruses have been assessed in lymphoblastoid cell lines, MAGI-CCR5 cell line, and peripheral blood mononuclear cells. The 50% inhibitory concentration (IC50) value is in the range of 0.0013~0.158 μg/mL. In the combination study with nucleoside reverse transcriptase inhibitors (NRTI), non-nucleoside reverse transcriptase inhibitor (NNRTI) and protease inhibitors (PI), it exhibited a synergistic effect. Most kinds of combined clinical study have not been carried out. In vitro tests have demonstrated that it has antiviral activity against HIV-1 (A, C, D, E, F and G subtypes) (IC50 values in the range of 0.007~0.075 μg/mL) while displaying specific inhibition activity against the HIV-2 type (IC50 values in the range of 0.007~1.5 μg/mL).
Pharmacokinetics
Pharmacokinetic assessment was performed in healthy volunteers and HIV-infected individuals. The pharmacokinetics of the two groups is similar with each other.
Absorption: oral administration gives rapid absorption with the drug being widely distributed. After administration of 1 to 2 hours, the plasma concentration reaches peak. 20 cases of HIV infected patients were subject to multiple fold doses of drugs through oral administration (mean ± SD) the plasma concentration peak of emtricitabine (Cmax) is 1.8 ± 0.7μg/mL. At 24 hours, the plasma drug concentration-time area under the curve (AUC) is 10.0 ± 3.1 (h • μg)/mL. At 24h hours after administration, the average steady-state plasma concentration is 0.09μg/mL. The average bioavailability is 93%. At multiple folds of doses, the pharmacokinetics is in proportion with the dose (25~200mg).
Distribution: In vitro binding rate of emtricitabine to human plasma proteins is < 4%; when the concentration exceeds the range of 0.02~200 μg/mL, it exists in its free state. At the time of peak concentration, the ratio of plasma drug concentration and blood drug concentration is 1.0, and the ratio of the seminal fluid concentration and plasma drug concentration is 4.0.
Metabolism: In vitro studies have shown that emtricitabine is not an inhibitor of human CYP450 enzymes. Taking 14C-labeled emtricitabine with the prototype reaching the urine (86%) and it (14%). 13% of the dose is converted into three metabolites. The biotransformation substances include the oxidized sulfur portion for generating 3'-sulfoxide diastereomers (9%), and binding to glucuronide for formation of 2'-oxy-glucuronide (4%). Other metabolites have not been determined.
Excretion: The half life of the emtricitabine plasma concentration is about 10 hours. The renal clearance rate of emtricitabine is higher than the creatinine clearance rate, suggesting that it is excreted through glomerular filtration and tubular secretion, and there may be substance competing with it for the kidneys.
Indications
1. Emtricitabine is combined with other antiviral drugs for treating adult HIV-1 infection. Patients should be those who haven’t been subject to treatment by reverse transcriptase inhibitors or those who have received reverse transcriptase inhibitors with virus having been suppressed.
2. It can be used for the treatment of chronic hepatitis B.
Side effects
In the clinical applications, for patients who are subject to emtricitabine and other antiviral drugs, the most common adverse reactions include headache, diarrhea, nausea and rash with the extent from being mild to moderate to severe. About 1% of patients discontinued medication due to the above reasons. The frequency of all kinds of adverse reactions is comparable with the control group quite but with the skin pigmentation being slightly higher in the emtricitabine group. Skin pigmentation usually significantly appears in the palm/foot, being generally mild and not accompanied by other symptoms. The clinical significance and the mechanism are not clear.
Precautions
This product is mainly excreted by the kidneys, so patients with renal insufficiency should reduce the administered amount upon taking this drug.
Pregnant women, when taking emtricitabine, may get an adverse effect on the fetus and newborn. This product can also be secreted through breast milk which may also affect the infant. Therefore, it is generally not recommended for pregnant and lactating women to use this product unless in situation of considering save the mother's life.
It has not been established of the children's safety basis. Therefore, it is not recommended for children. Elderly should be cautious when choosing the dose. They can apply appropriate reduction on the dose according to the actual conditions of liver, kidney, heart function decline, associated diseases and the effects of other kinds of drugs.
The impact of excessive drugs is unknown, if there is drug overdose, monitoring should be carried out. Apply maintenance dose treatment when necessary.
Uses
It is white, white-like powder or crystalline powder Application: treatment of chronic hepatitis B; It has a strong antiviral activity with the safety and tolerance being good.
Description
Emtricitabine is a synthetic nucleoside inhibitor of HIV-1 reverse transcriptase. Its mechanism of action entails the phosphorylation of the oxathiolane carbinol function by cellular enzymes to form the corresponding 5′-triphosphate, which competes with the endogenous 2′-deoxycytidine 5′-triphosphate substrate. The in vitro activity of emtricitabine ranges from IC50 of 0.00013 to 0.64 mM against lymphoblastoid cell lines, MAGI-CCR5 cell lines, and peripheral blood mononuclear cells. Emtricitabine is prepared in about 16 steps from L-gulose, from which the O–C–S carbon stereochemistry is derived in the formation of substituted key 4-O-Acetyl-1,3- oxathiolane intermediate. This intermediate is coupled with N-benzoyl-O-trimethylsilyl- 5-fluoro-cytosine in the presence of trimethylsilyl triflate. The resulting anomeric mixture is separated by silica chromatography and subjected to two deprotection steps. Emtricitabine resistant isolates (M184V/I) were cross-resistant to lamivudine (desfluoro version of emtricitabine) and zalcitabine but remained sensitive to abacavir, didanosine, stavudine, tenofovir, zidovudine, and non-nucleoside reverse transcriptase inhibitors (delavirdine, efavirenz, and nevirapine). In a 48-week clinical trial using emtricitabine with didanosine and efavirin versus stavudine, didanosine and efavirenz produced 81 versus 68% responder rate respectively. In one study, 37.5% of treatment na?¨ve patients that were not achieving successful viral levels were found to exhibit a reduced potency toward emtricitabine. This resistance was due to M184V/I mutation in the HIV reverse transcriptase gene. Recommended dosing of emtricitabine is 200 mg (capsule) once per day. It is 93% bio-available and has a plasma half-life about 10 h, with 86% renal clearance. Side effects associated with emtricitabine treatment were generally mild to moderate and included headache, diarrhea, nausea, and rash. A generally mild and asymptomatic hyperpigmentation of the palms and/or soles was also observed.
Chemical Properties
White to Off-White Crystalline Solid
Originator
Emory University (US)
Uses
anti-Alzheimer’s treatment
Uses
Labeled Emtricitabine, intended for use as an internal standard for the quantification of Emtricitabine by GC- or LC-mass spectrometry.
Uses
A reverse transcriptase inhibitor. A nucleoside analog structurally related too lamivudine.It is an effective antiviral agent against HIV, HBV, and other viruses replicating in a similar manner. A nucleoside analog structurally related to Lamivudine (L172500).
Definition
ChEBI: An organofluorine compound that is 5-fluorocytosine substituted at the 1 position by a 2-(hydroxymethyl)-1,3-oxathiolan-5-yl group (2R,5S configuration). It is used in combination therapy for the treatment of HIV-1 infect on.
brand name
Emtriva (Gilead Sciences) [Note—Emtricitabine has appeared in the literature with the trivial names, (-)-FTC and FTC-(-).].
Acquired resistance
Resistance is associated with a substitution in the HIV-1 reverse transcriptase gene at codon 184 (M184V/I). Emtricitabineresistant isolates are cross-resistant to lamivudine. HIV-1 isolates with the K65R substitution in the reverse transcriptase coding region have reduced susceptibility.
General Description
Emtricitabine is an orally active NRTI whose pharmacokineticsare favorable for once-daily administration.
Pharmaceutical Applications
A synthetic nucleoside analog of cytosine, formulated for oral use.
Pharmacokinetics
Oral absorption: capsules 93%
Cmax 200 mg oral once daily: 1.8 ± 0.7 mg/L
Plasma half-life: c. 10 h
Volume of distribution: 1.4 ± 0.3 L/kg
Plasma protein binding: <4%
Absorption and distribution
It is rapidly and extensively absorbed. There is moderate CNS penetration. The estimated semen:plasma ratio is approximately 4. There are presently no data on levels in breast milk.
Metabolism and excretion
It does not inhibit human cytochrome P450 enzymes. About 80% is excreted in the urine, the rest in feces. Renal clearance is greater than the estimated creatinine clearance, suggesting elimination by both glomerular filtration and active tubular secretion. There may be competition for elimination with other compounds that are renally excreted. Exposure is significantly increased in renal insufficiency, but dose reductions are not generally recommended. It is unlikely that a dose adjustment would be required in the presence of hepatic impairment.
Clinical Use
Treatment of HIV infection (in combination with other antiretroviral drugs)
Side effects
At least 10% of patients suffer headache, diarrhea, nausea, fatigue, dizziness, depression, insomnia, abnormal dreams, rash, abdominal pain, asthenia, increased cough and rhinitis. Skin hyperpigmentation is common (≥10%) in pediatric patients. Emtricitabine competes with lamivudine for the enzymes involved in intracellular phosphorylation; their co- administration is contraindicated.
Synthesis
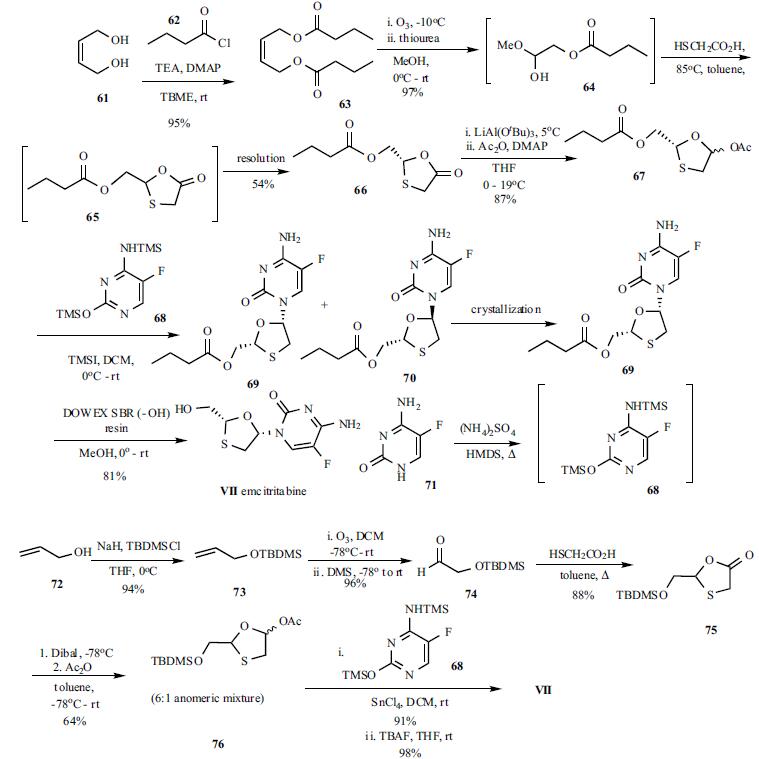
Emcitritabine (VII) was discovered by researchers at Emory University and licensed to Triangle Pharmaceuticals, which started the development work before being acquired by Gilead. Because emcitritabine (VII) belongs to an important structural class of nucleosides with marketed drugs, such as 3TC, several processes for the manufacture of this class of oxathiolane nucleosides have appeared in patents and scientific literature. However, only the synthesis described in the latest patent filed for the manufacture of emcitritabine (VII) and one other efficient synthesis from the Liotta group will be described . The synthesis started with diacylation with butyryl chloride (62) of the 2-butene-1,4-diol (61) in methyl t-butylether at 0??C to room temperature in the presence of triethylamine to give diacylated product 63 in 95% yield. Ozonolysis followed by reduction with thiourea provided a mixture of hemiacetal 64 mixed with acetals, dimers and trimers in 97% yield, which was used in the next step directly. The hemiacetal mixture was reacted with thioacetic acid in toluene at 85??C for 3 hr to give the crude keto oxathiolane mixture, which was purified by vacuum distillation in a 2-in Pope Scientific wiped film still to remove impurities and collect about 92% pure 66 in 54% yield. Also mentioned in the patent is the potential use of enzymatic resolution of the isomers as reported previously. This keto oxathiolane 66 was reduced at 5??C with lithium aluminum t-butoxide, which was prepared in situ via reaction of LAH and t-butanol, and the resulting lactol was trapped with acetic anhydride in the presence of DMAP in the same reaction vessel to give, after workup, 87% yield of the key intermediate acetate 67. The bis-silyl protected 5- fluorocytosine 68, prepared in situ by reacting 5- fluorocytosine (71) with HMDS, was reacted with acetate 67 in the presence of trimethylsilyliodide at 0??C to room temperature to give a 1:1 mixture of alpha and beta-anomers 69 and 70. Pure 69 could be isolated by recrystallization from toluene. Cleavage of the butyryl group with a strongly basic DOWEX SBR resin in methanol at room temperature gave emcitritabine (VII) in 81% yield. An alternate concise synthesis reported by Liotta et al is worth mentioning. This synthetic route accessed the key thioxalane acetate 76 as the TBDMS ether in four steps from allyl alcohol 72. The key step to the preparation of the final compound was the coupling of the bis-silyl 5-fluorocytosine (68) with acetate 76 with tin tetrachloride in a stereoselective manner, after cleavage of the silyl groups and recrystallization, to give pure cis isomer emcitritabine (VII) in excellent yield.
Drug interactions
Potentially hazardous interactions with other drugs Antivirals: avoid concomitant use with lamivudine. Orlistat: absorption of emtricitabine possibly reduced.
Metabolism
There is limited metabolism of emtricitabine. The biotransformation of emtricitabine includes oxidation of the thiol moiety to form the 3'-sulphoxide diastereomers (approximately 9% of dose) and conjugation with glucuronic acid to form 2'-O-glucuronide (approximately 4% of dose). Emtricitabine is primarily excreted by the kidneys with complete recovery of the dose achieved in urine (approximately 86%) and faeces (approximately 14%). 13% of the emtricitabine dose was recovered in urine as three metabolites
Emtricitabine Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Sigma Audley | +86-18336680971 +86-18126314766 | nova@sh-teruiop.com | China | 525 | 58 |
| Shanghai Affida new material science and technology center | +undefined15081010295 | 2691956269@qq.com | China | 359 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618740459177 | sarah@tnjone.com | China | 893 | 58 |
| Frapp's ChemicalNFTZ Co., Ltd. | +86 (576) 8169-6106 | sales@frappschem.com | China | 885 | 50 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 | sales01@cooperate-pharm.com | CHINA | 1811 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | fandachem@gmail.com | China | 9348 | 55 |
| Shanghai Yingrui Biopharma Co., Ltd. | +86-21-33585366 - 03@ | sales03@shyrchem.com | CHINA | 738 | 60 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
Related articles
- Emtricitabine---A potent, synthetic nucleoside reverse transcriptase inhibitor.
- Emtricitabine ( FTC) is the negative enantiomer of a thio analog of dideoxycytidine with a fluorine in the 5 position. Emtrici....
- Apr 18,2022
View Lastest Price from Emtricitabine manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-25 | Emtricitabine
143491-57-0
|
US $0.00 / kg | 1kg | 99% | 1000kg | Shaanxi TNJONE Pharmaceutical Co., Ltd | |
 |
2024-04-22 | Emtricitabine
143491-57-0
|
US $0.00 / KG | 2KG | 99% up, DMF, FDA (zero defect) | 20 tons | Sinoway Industrial co., ltd. | |
 |
2024-03-19 | Emtricitabine
143491-57-0
|
US $45.00-35.00 / kg | 1kg | 99.8% | 200tons/year | Sigma Audley |
-

- Emtricitabine
143491-57-0
- US $0.00 / kg
- 99%
- Shaanxi TNJONE Pharmaceutical Co., Ltd
-

- Emtricitabine
143491-57-0
- US $0.00 / KG
- 99% up, DMF, FDA (zero defect)
- Sinoway Industrial co., ltd.
-

- Emtricitabine
143491-57-0
- US $45.00-35.00 / kg
- 99.8%
- Sigma Audley
143491-57-0(Emtricitabine)Related Search:
1of4