- Irbesartan
-

- $0.00 / 1Kg/Bag
-
2024-04-22
- CAS:138402-11-6
- Min. Order: 1Kg/Bag
- Purity: EP / USP
- Supply Ability: 20 tons
- Irbesartan
-

- $35.00/ kg
-
2024-03-16
- CAS:138402-11-6
- Min. Order: 1kg
- Purity: 99.8%
- Supply Ability: 200tons/year
- Irbesartan
-

- $0.00 / 1KG
-
2024-03-16
- CAS:138402-11-6
- Min. Order: 100g
- Purity: 98%+
- Supply Ability: 100kg
|
| Product Name: | Irbesartan | | Synonyms: | 2-BUTYL-3-[[2'-(1H-TETRAZOL-5-YL)[1,1'-BIPHENYL]-4-YL]METHYL]-1,3-DIAZASPIRO[4.4]NON-1-EN-4-ONE;IRBESARTAN;BMS-186295;APROVEL;1,3-Diazaspiro[4.4]non-1-en-4-one, 2-butyl-3-[[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-;2-Isopropryl-4-methylthiazole;2-butyl-3-[[4-[2-(2H-1,2,3,4-tetrazol-5-yl)phenyl]phenyl]methyl]-1,3-diazaspiro[4.4]non-1-en-4-one;2-butyl-3-[[4-[2-(2H-tetrazol-5-yl)phenyl]phenyl]methyl]-1,3-diazaspiro[4.4]non-1-en-4-one | | CAS: | 138402-11-6 | | MF: | C25H28N6O | | MW: | 428.53 | | EINECS: | 604-078-2 | | Product Categories: | Aromatics;Heterocycles;Isotopically Labeled Pharmaceutical Reference Standard;LITAREX;Inhibitors;Intermediates & Fine Chemicals;Pharmaceuticals;API's;Hypertension;Active Pharmaceutical Ingredients;Antihypertensive;API | | Mol File: | 138402-11-6.mol | 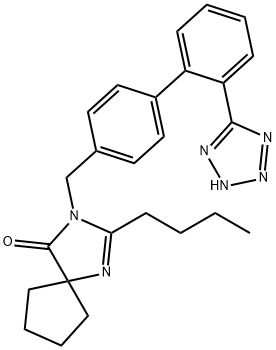 |
| | Irbesartan Chemical Properties |
| | Irbesartan Usage And Synthesis |
| Antihypertensives | Irbesartan is an angiotensin Ⅱreceptor inhibitor, angiotensin Ⅱ receptors are divided into AT1, AT2, irbesartan can inhibit AngⅠtransform into AngⅡby selectively blocking the AT1 receptor of AngⅡ, specifically antagonize angiotensin converting enzyme 1 receptor (AT1), the antagonism of AT1 is 8500 times than that of AT2, it can inhibit vasoconstriction and aldosterone release by selectively blocking the binding AngⅡ with AT1 receptor and result in the antihypertensive effect. This product does not inhibit angiotensin converting enzyme (ACE), renin, and other hormone receptors, neither suppress the blood pressure regulation and the balance of sodium ion channels. Irbesartan can also reduce electrical remodeling of the myocardium, thereby reduce the mortality rate of patients with hypertension, it is the most effective drug for treatment of hypertension and cardiovascular disease.
Angiotensin antagonists (ARB) is applied to the clinical treatment of hypertension and diabetic nephropathy. At present, domestic drugs for treating high blood pressure are divided into angiotensin converting enzyme inhibitors (ACEI), calcium channel blockers, beta blockers and so on according to different working parts. By contrast, ARB has better antihypertensive effect, while Irbesartan is a new type of angiotensin antagonist which has clear antihypertensive effect, and has an important role in inhibiting left ventricular hypertrophy and protecting the kidneys.
According to foreign reports, it can quickly absorb by oral and the bioavailability is 60-80%, not affected by food. Plasma tmax is 1-1.5 hours, plasma protein combined rate is 90%, elimination half-life is 11-15 hours, reaching steady state in three days. By aldehyde oxidation acidification or glucose metabolism, vitro studies have shown that mainly oxidation by cytochrome enzymes of P450 and 2C9. This product and its metabolites excrete by biliary tract and kidney.
Irbesartan (Emberd) produced by Sanofi-aventis hangzhou minsheng pharmaceutical co., LTD won the approval of the SFDA used in the treatment of hypertension in type 2 diabetic nephropathy on March 8, 2007, and becomes China's first effective antihypertensive drug. | | Telmisartan | Telmisartan is a new type of blood pressure drug, is a kind of specific angiotensin Ⅱ receptor (AT Ⅰ) antagonist, used in the treatment of essential hypertension. To instead of high affinity of angiotensin receptor Ⅱwith AT Ⅰ receptor subtypes (known angiotensin Ⅱ loci). Telmisartan has no effects in the AT Ⅰ receptor agonist sites, selectively combined with ATⅠ receptor and the combined effect is durable. Has no affinity with other receptor (including AT2 and other characteristics of less AT receptors). The other receptor function remains to be seen, excessive receptor stimulation effect due to telmisartan angiotensin Ⅱ level is also not known. Telmisartan is not inhibit human plasma renin, and also don't block the ion channel. Don't inhibit angiotensin converting enzymeⅡ, the enzyme can degrade and enhance adverse reactions caused by the excitation peptide inhibition. 80 mg of telmisartan in the human body is almost completely inhibit angiotensin Ⅱ causing increased blood pressure. Inhibition effect is last for a full of 24 hours and can still be detectable in 48 hours. Antihypertensive effect is obvious in 3 hours after the first dose. At 4 weeks after treatment began to gain maximum antihypertensive effect, and can be maintained in the long-term treatment. If the treatment was interrupted suddenly, the blood pressure returns to the treatment level in just a few days, rather than a resilient high blood pressure. In direct comparison of two kinds of high blood pressure drugs in clinical trials, treatment group was significantly lower than that of patients with dry cough angiotensin converting enzyme inhibitors in treatment group.
The above information is edited by the Chemicalbook of Duan Yalan. | | Usage and Dosage | Oral: recommended starting dose of 0.15 g, 1 time a day. It can be increased to 0.3 g according to the condition, 1 time a day. Can be used alone, also can be shared with other anti-hypertensive drugs.
Severe hypertension and not satisfied with drop in blood pressure after the drug increment, can add with small dose of diuretic (such as thiazide) or other antihypertensive drugs. | | Drug interaction | It has no obvious interactions with hydrochlorothiazide, digoxin, warfarin, nitrate benzene, pyridine.
It should be paid attention to when shared with diuretics due to insufficient blood volume and low sodium which can cause low blood pressure (hypotension). When shared with potassium diuretics (such as ammonia, benzene with organism), should avoid potassium increasing.
Do not affect each other pharmacokinetics when shared with digitalis drugs such as digoxin, beta blockers such as atenolol, calcium antagonists such as benzene, pyridine nitrate. | | Overdose | After an overdose of this product can occur hypotension, tachycardia or bradycardia, vomiting, gastric lavage and support therapy should be adopted. | | Side effects | Common adverse reactions: headache, dizziness, palpitation, etc., I have a cough, general degree is slight, the majority of patients continue to drugs are tolerated.
Rare urticaria and angioneurotic oedema.
Literature on this product is: the incidence of adverse reactions to more than 1% of indigestion, stomach burning, diarrhea, skeletal muscle pain, fatigue, and upper respiratory tract infection, but with the blank control group no significant difference.
Greater than 1% but less than control group in the incidence of abdominal pain, anxiety, nervousness, nausea, vomiting, chest pain, pharyngitis, skin rashes, tachycardia, etc. Low blood pressure and incidence of orthostatic hypotension is about 0.4%. | | Chemical property | Crystallization from 96% ethanol, melting point is 180-181 ℃. | | Uses | Antihypertensive drugs. Angiotensin Ⅱ-l (Ⅱ 1 A) receptor antagonist. Used to treat high blood pressure. | | Production Method | Methods 1:
1-(fluorene methoxy carbonyl amino) ring e carboxylic acid (I) benzylamine reaction with 4-(2-phenyl cyano) amidation, product (Ⅲ) and water release N to produce compound (Ⅳ). (Ⅳ) and Triethyl orthobutyrate condensation, cyclization to compound (V), then azide to formation tetrazolium with sodium reaction to obtain Telmisartan.
Methods 2:
1-(fluorene methoxyl carbonyl amino) ring e carboxylic acid (I) condensation with compound(Ⅳ), the product remove N protection based to compound (Ⅷ), reacts with Triethyl orthobutyrate, obtain the product. | | Description | Avapro was launched in Germany, the UK and the US for hypertension. It
can be prepared in six steps starting with cyclopentanone or in three steps from 1-
aminocyclopentanecarboxylic acid ethyl ester and pentanimidic ethyl ester. Avapro is
an angiotensin Ⅱ receptor antagonist that is non-competitive and selective for AT,
subtypes and has no AT2 activity at postsynaptic receptors compared to presynatpic. It
has no affinity for various non angiotensin Ⅱ receptor types in binding, no interaction
with calcium channels or antiports, and no affinity for α1- and α2--adrenoreceptors,
serotonergic receptors, muscarinic m1 and m2 or other receptors. It is as potent as
saralasin but with no agonist activity and is 10 times more potent than DuP753 in rats.
It is similar in efficacy to enalapril (in those with severe hypertension) and atenolol,
while more effective than losartan for mild to moderate hypertension. | | Chemical Properties | Crystalline Solid | | Originator | Sanofi (France) | | Uses | antidepressant | | Uses | An angiotensin II type 1 (AII1)-receptor antagonist | | Uses | For the treatment of hypertension, as well as diabetic nephropathy with an elevated serum creatinine and proteinuria (>300 mg/day) in patients with type 2 diabetes and hypertension. Irbesartan is also used as a second line agent in the treatment of conges | | Definition | ChEBI: A biphenylyltetrazole that is an angiotensin II receptor antagonist used mainly for the treatment of hypertension. | | Manufacturing Process | 1. Synthesis of 2-n-Butyl-4-spirocyclopentane-2-imidazolin-5-one
Method 1:
The ethyl ester of 1-aminocyclopentanecarboxylic acid is prepared according
to Adkins and Billica (J. Amer. Chem. Soc., 1948, 70, 3121).
Ethyl valerimidate hydrochloride is prepared according to Mac Elvain (J. Amer.
Chem. Soc., 1942, 64, 1825-1827) and then freed from its hydrochloride by
reaction with potassium carbonate and extraction with CH2Cl2.
The ethyl ester of 1-aminocyclopentanecarboxylic acid (1.57 g) and ethyl
valerimidate (1.56 g) are dissolved in 12 ml of xylene containing 6 drops of
acetic acid. After refluxing for 6.5 h, the reaction medium is concentrated
under vacuum, the residue is chromatographed on silica gel using a
chloroform/methanol/acetic acid mixture (94/4/2; v/v/v) as the eluent. The
fraction containing the expected product is evaporated several times in the
presence of xylene and then benzene in order to remove the acetic acid. 1.91
g of 2-n-butyl-4-spirocyclopentane-2-imidazolin-5-one are obtained in the
form of a thick oil.
Method 2:
1.97 g of sodium cyanide are dissolved in 3.9 ml of water in a round�bottomed flask and a solution containing 2.33 g of ammonium chloride in 5.9
ml of water and 3.5 ml of 20% aqueous ammonia is added; finally, 3 g of
cyclopentanone in 3.8 ml of methanol are added to the flask. After stirring for
1.5 h, the mixture is heated at 60°C for 45 min, heating is then stopped,
stirring is continued for 45 min and the mixture is then cooled to 25°C. It is
extracted several times with methylene chloride.
The 1-aminocyclopentanenitrile obtained is dissolved in 300 ml of acetone,
and a solution of 2.25 g of oxalic acid dihydrate in 200 ml of acetone is
added, with stirring. The precipitate of 1-aminocyclopentanenitrile formed is
filtered off.
5.1 g of the oxalate obtained in the previous step are treated with 7.65 ml of
concentrated sulfuric acid (d = 1.84) over 45 min, with stirring. The evolution
of a gas is observed and the temperature rises to 100°C. The mixture is
cooled to about 35°C and poured into a mixture of ice and concentrated
aqueous ammonia (10 g/2.8 ml). The suspension formed is extracted with
chloroform containing 5% of methanol. The 1-aminocyclopentanecarboxamide
was obtained.
3 g of the compound prepared in the previous step are placed in 70 ml of
anhydrous THF and 3.3 ml of triethylamine, and 3 ml of valeryl chloride in 10
ml of anhydrous THF are added, with stirring. A white suspension is formed.
The intermediate which is formed, but not isolated, is 1-(N�valeryl)aminocyclopentanecarboxamide. 6 g of potassium hydroxide pellets, 7
ml of water and 16 ml of methanol are added. The mixture is refluxed for 2.5
h and 9 g of ammonium chloride are then added. After stirring for 15 min, the
mixture is concentrated under vacuum. The residue of the 2-n-butyl-4-
spirocyclopentane-2-imidazolin-5-one obtained is taken up in water and
extracted with ethyl acetate.
2. Synthesis of 2-n-butyl-4-spirocyclopentane-1-[(2'-(tetrazol-5-yl)biphenyl-4-
yl)-methyl]-2-imidazolin-5-one
A mixture containing 250 mg of sodium hydride (as an 80% dispersion in
mineral oil) and 5 ml of DMF is prepared under a nitrogen atmosphere and a
solution containing 0.97 g of 2-n-butyl-4-spirocyclopentane-2-imidazolin-5-
one in 10 ml of DMF is added dropwise. The mixture is stirred for 30 min at
20°C and a solution of 1.5 g of 4-bromomethyl-2'-cyanobiphenyl in 10 ml of
DMF is then added. After stirring for 1 h at 20°C, the DMF is evaporated off
under reduced pressure, the residue is then taken up with ethyl acetate,filtered and evaporated. The residue of 1-[(2'-cyanobiphenyl-4-yl)methyl]-2-n�butyl-4-spirocyclopentane-2-imidazolin-5-one is purefied by chromatography
1.56 g of the previous product, 2.6 g of tributyltin azide and 30 ml of xylene
are refluxed for 66 h. The xylene is then evaporated off and the residue is
dissolved in 20 ml of CH2Cl2 and 5 ml of THF with the addition of 0.8 ml of 10
N sodium hydroxide solution and, after stirring for 30 min, 2.5 g of trityl
chloride, and the mixture is stirred for 26 h. After evaporation of the solvents,
the residue is taken up in ethyl acetate in ethyl acetate, washed with water
and then with a 3% solution of potassium bisulfate and water. It is dried and
evaporated. The residue is chromatographed on alumina using a hexane/ethyl
acetate mixture (9/1; v/v) as the eluent to give 1.97 g of the 2-n-butyl-4-
spirocyclopentane-1-[(2'-(triphenylmethyltetrazol-5-yl)biphenyl-4-yl)methyl]-
2-imidazolin-5-one. Melting point 150-152°C.
1.96 g of the product prepared in the previous step are dissolved in 10 ml of
methanol and 10 ml of THF. After the reaction medium has been cooled to
5°C, 1.5 ml of 4 N hydrochloric acid are added and the mixture is stirred for 3
h at 20°C and 1 h at 30°C. After evaporation of the solvents, the residue is
taken up in water and the pH is brought to 12 by the addition of 10 N sodium
hydroxide solution. The aqueous phase is extracted with ether, toluene and
ether again. The aqueous phase is acidified to pH 2 by the addition of 1 N
hydrochloric acid and then extracted with ethyl acetate and the extract is
evaporated. The aqueous phase is acidified to pH 2 by the addition of 1 N
hydrochloric acid and then extracted with ethyl acetate and the extract is
dried and evaporated. The white solid obtained is dried at 50°C under 0.05
mm of mercury to give 840 mg of the 2-n-butyl-4-spirocyclopentane-1-[(2'-
(tetrazol-5-yl)biphenyl-4-yl)methyl]-2-imidazolin-5-one. Melting point 180-
181°C. | | Brand name | Avapro
(Sanofi Aventis);Aprovel/Avapro. | | Therapeutic Function | Antihypertensive | | General Description | Irbesartan, 2-butyl-3-[[29-(1H-tetrazol-5-yl)[1,19-biphenyl]-4-yl]methyl]1,3-diazaspiro[4,4]non-1en-4-one (Avapro), like losartan, possesses the acidic tetrazolesystem, which most likely plays a role, similar to the acidicgroups of angiotensin II, in binding to the angiotensin II receptor.In addition, the biphenyl system that serves to separatethe tetrazole from the aliphatic nitrogen is still present.A major difference in this agent is that it does not possessthe acidic side chain. Even so, irbesartan has good affinityfor the angiotensin II receptor because of hydrogen bondingwith the carbonyl moiety of the amide system. Also, thisparticular agent does not require metabolic activation ascandesartan does. | | Biochem/physiol Actions | Irbesartan is an angiotensin II type 1 (AT1) receptor antagonist with antihypertensive activity. It also elicits selective peroxisome proliferator-activated receptor γ (PPARγ)-modulating activity and possesses anti-inflammatory properties. Irbesartan shows protective cardiovascular effects and provides protection against chronic glomerulonephritis. | | Clinical Use | Angiotensin-II receptor antagonist:
Hypertension
Diabetic nephropathy | | Veterinary Drugs and Treatments | Although experience in veterinary medicine is minimal irbesartan
may be useful in treating canine hypertension associated with renal
insufficiency. It may be effective in treating heart failure when dogs
are unable to tolerate ACE inhibitors, but documentation for this
use is lacking. One study, using very high irbesartan dosages (60
mg/kg PO twice daily) in dogs with subacute mitral regurgitation,
demonstrated no improvement in left ventricular function or prevention
of left ventricular remodeling (Perry, Wei et al. 2002). | | Drug interactions | Potentially hazardous interactions with other drugs
Anaesthetics: enhanced hypotensive effect
.
Analgesics: antagonism of hypotensive effect and
increased risk of renal impairment with NSAIDs;
hyperkalaemia with ketorolac and other NSAIDs.
Antihypertensives: increased risk of hyperkalaemia
hypotension and renal impairment with ACE-Is and
aliskiren.
Ciclosporin: increased risk of hyperkalaemia and
nephrotoxicity.
Diuretics: enhanced hypotensive effect;
hyperkalaemia with potassium-sparing diuretics.
ESAs: increased risk of hyperkalaemia; antagonism
of hypotensive effect.
Lithium: reduced excretion (possibility of enhanced
lithium toxicity).
Potassium salts: increased risk of hyperkalaemia.
Tacrolimus: increased risk of hyperkalaemia and
nephrotoxicity. | | Metabolism | Following oral or intravenous administration of 14C
irbesartan, 80-85% of the circulating plasma radioactivity
is attributable to unchanged irbesartan. Irbesartan is
metabolised by the liver via glucuronide conjugation and
oxidation. The major circulating metabolite is irbesartan
glucuronide (approximately 6%). In vitro studies indicate
that irbesartan is primarily oxidised by the cytochrome P450
enzyme CYP2C9; isoenzyme CYP3A4 has negligible effect.
Irbesartan and its metabolites are eliminated by both
biliary and renal pathways. After either oral or IV
administration of [14C]-irbesartan, about 20% of the
radioactivity is recovered in the urine, and the remainder
in the faeces. | | storage | Store at +4°C |
| | Irbesartan Preparation Products And Raw materials |
|