- Apixaban
-

- $2.00 / 1kg
-
2024-09-20
- CAS:503612-47-3
- Min. Order: 1kg
- Purity: 99.5%
- Supply Ability: 200kgs
- Apixaban
-

- $50.00 / 1kg
-
2024-09-20
- CAS:503612-47-3
- Min. Order: 1kg
- Purity: 99.99%
- Supply Ability: 1000
- Apixaban
-

- $15.00 / 1kg
-
2024-09-20
- CAS:503612-47-3
- Min. Order: 1kg
- Purity: 99.9%
- Supply Ability: 500
|
| Product Name: | Apixaban | | Synonyms: | Apixaban;Apixaban, BMS 562247-01;BMS 562247-01;1-(4-Methoxy-phenyl)-7-oxo-6-[4-(2-oxo-piperidin-1-yl)-phenyl]-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylic acid aMide;1-(4-Methoxyphenyl)-7-oxo-6-[4-(2-oxopiperidin-1-yl)phenyl]-4,
5-dihydropyrazolo[3,4-c]pyridine-3-carboxaMide;Nilotinib and its interMediate;Apixaban ,ELIQUIS;1-(4-Methoxyphenyl)-7-oxo-6-[4-(2-oxopiperidin-1-yl)phenyl]-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide Apixaban | | CAS: | 503612-47-3 | | MF: | C25H25N5O4 | | MW: | 459.5 | | EINECS: | 639-684-6 | | Product Categories: | Inhibitors;Inhibitor;API;503612-47-3 | | Mol File: | 503612-47-3.mol | 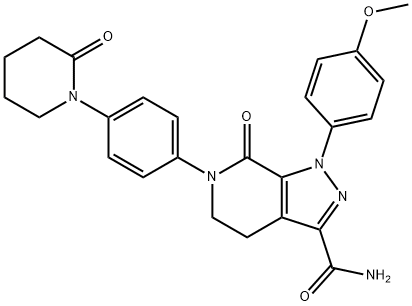 |
| | Apixaban Chemical Properties |
| Melting point | 235-238°C | | Boiling point | 770.5±60.0 °C(Predicted) | | density | 1.42 | | storage temp. | Refrigerator | | solubility | DMSO (Slightly, Heated), Methanol (Slightly) | | pka | 15.01±0.20(Predicted) | | form | Solid | | color | White to Off-White | | InChIKey | QNZCBYKSOIHPEH-UHFFFAOYSA-N | | SMILES | C1(=O)N(C2=CC=C(N3CCCCC3=O)C=C2)CCC2C(C(N)=O)=NN(C3=CC=C(OC)C=C3)C1=2 |
| | Apixaban Usage And Synthesis |
| Indications and Usage | Apixaban is a new form of oral anticoagulant drug developed by Bristol Myers Squibb and Pfizer. It is a new form of oral Xa factor inhibitor, and its commercial name is Eliquis. Apixaban is used to treat adult patients undergoing elective hip or knee replacement surgery to prevent venous thromboembolism (VTE)
| | Mechanisms of Action | Apixaban is an oral selective activated Xa factor inhibitor and can prevent thrombin generation and thrombosis.
| | Clinical Research | Apixaban is the third new oral anticoagulant to go on sale, following dabigatran and rivaroxaban, and it has already been approved in Europe for preventing venous thromboembolism in patients undergoing elective hip or knee replacement surgery. Out of these three oral anticoagulants approved in Europe, compared to the current standard preventative treatment against venous thromboembolism, enoxaparin, rivaroxaban excelled in the record experiment, and apixaban excelled in the advance experiment. Rivaroxaban’s curative effects were slightly superior, but it caused more severe bleeding than apixaban. Researchers attributed these differences to medication time, as rivaroxaban was taken 6-8 hours after surgery in the record experiment, while apixaban was used 18 hours after surgery in the advance experiment. These drugs have better curative effect when used closer to time of surgery, but also have an increased bleeding risk. Clinical research showed that compared to a daily subdermal injection of 40mg enoxaparin, 2 oral 2.5mg dosages of apixaban had better preventative effects against venous thromboembolism following hip or knee replacement surgery and did not increase bleeding risk.
| | Drug Interactions | 1.A double inhibitor of strong CYP3A4 and P-gp increases apixaban’s blood levels: decrease Eliquis dosage to 2.5mg or avoid simultaneous usage.
2.A inductor of strong CYP3A4 and P-gp can decrease apixaban’s blood levels: avoid simultaneous usage.
| | Warnings and Precautions |
- Breastfeeding mothers should stop usage or stop breastfeeding.
- Use during pregnancy is not advised.
- Use while experiencing severe liver damage is not advised.
| | Description | Eliquis (apixaban), a direct inhibitor of factor Xa (FXa), was approved by
the European Commission on May 18, 2011 for prevention of venous thromboembolic
events (VTE) in adult patients who have undergone elective hip or
knee replacement surgery.
The discovery of apixaban was the culmination of a succession of novel and innovative medicinal
chemistry discoveries starting with the identification of nonpeptide
leads, rational drug design using computer-aided and X-ray
crystallographic information, and the building of drug-like properties
through the systematic replacement of basic groups with neutral
moieties. Apixaban arose from modifications to razaxaban by constraining a pyrazole amide to form a
bicyclic pyrazolo-pyridinone scaffold. Optimization of the P1 group
resulted in the identification of the nonbasic methoxy phenyl group,
while a P4 piperidinone improved the balance of potency and
pharmacokinetics with low Vdss. The synthesis of apixaban begins with
the generation of a hydrazone of 4-methoxyaniline which is then used in
a 3+2 cycloaddition with a dihydropiperidinone to form a bicyclic
pyrazolo-pyridinone scaffold. The distal piperidinone group is installed
using an Ullmann coupling reaction followed by aminolysis of an ethyl
ester on the pyrazole ring to complete the synthesis of apixaban. | | Description | Apixaban is an orally bioavailable, selective inhibitor of both free and prothrombinase-bound factor Xa (Kis = 0.8 nM). In vivo, apixaban has antithrombotic effects in a rabbit model of venous thrombosis. Apixaban (357.5 mg) also prevents thrombus formation without inducing adverse bleeding events in a porcine model of aortic heterotopic valve replacement. Formulations containing apixaban have been used to prevent blood clot formation in patients with atrial fibrillation. | | Originator | Bristol Myers Squibb Company (United States) | | Uses | Apixaban is a highly selective, reversible inhibitor of Factor Xa with Ki of 0.08 nM and 0.17 nM in human and rabbit, respectively. | | Definition | ChEBI: A pyrazolopyridine that is 7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide substituted at position 1 by a 4-methoxyphenyl group and at position 6 by a 4-(2-oxopiperidin-1-yl)phenyl group. It is used for the
prevention and treatment of thromboembolic diseases. | | Brand name | Eliquis | | Pharmacokinetics |
The maximum plasma concentration (Cmax) of apixaban occurs 3–4 h after oral administration. The absorption of apixaban appears to occur primarily in the small intestine and decreases progressively throughout the gastrointestinal tract. Compared with oral administration, the bioavailability of 2.5 mg of apixaban solution was approximately 60% and 84% lower when released in the distal small bowel and ascending colon, respectively. For oral doses up to 10 mg, the absolute bioavailability of apixaban is~50%, resulting from the incomplete absorption and first-pass metabolism in the gut and liver[1].
| | Clinical Use | Apixaban is an oral anticoagulant with highly selective inhibition
of factor Xa. It was approved by the European Medicines
Agency (EMA) for the treatment of venous thromboembolic events
and first marketed in Germany under the brand name Eliquis in
June 2011. Apixaban was co-developed by Bristol-Myers Squibb
and Pfizer and represents the first approved drug for this indication
since warfarin over 50 years ago. | | Side effects | Possible side effects of Apixaban are: bleeding gums, nosebleeds, heavy vaginal bleeding
, red, pink, or brown urine; red or black, tarry stools; coughing or spitting up blood or a substance that looks like coffee grounds; swelling or joint pain, headache, rash, chest pain or tightness in the chest, swelling of the face or tongue, trouble breathing, wheezing. Feeling dizzy or fainting.Apixaban prevents your blood from clotting properly, so if you get a cut or injury, it may take longer than usual for the bleeding to stop. This medication may also cause you to bruise or bleed more easily. | | Synthesis | Although several convenient
preparations of apixaban (BMS-562247) have been reported, the most likely process-scale route is described in the scheme. The
starting material 4-iodoaniline (14) was acylated with 5-bromovaleryl
chloride (15) and triethylamine followed by cyclization under
basic conditions to give lactam 16 in 49% yield. Intermediate 16
was then reacted with phosphorus pentachloride to provide the
a,a-dichlorinated lactam 17 in 87% yield.30 This dichloride was reacted
with excess morpholine to affect an alkylation¨Celimination
sequence to afford enaminolactam 18 in 86% yield. N-Arylation
of this iodide with valerolactam 19 using a copper (I) catalyst resulted
in a 77% yield of the desired p-bispiperidone 20. Interestingly,
sequential exposure of 20 to a nitrile imine generated from
the treatment of ethyl 2-chloro-2-(2,4-methoxyphenyl)-hydrazono)
acetate 21 with base resulted in a [3+2] dipolarcycloadditon
reaction. Upon acidification with 4 N HCl, pyrazole 22 was furnished
in 67% over two steps. Conversion of the ester within 22
to the corresponding amide was achieved via a mixture of formamide
and sodium methoxide to give apixaban (III) in 71% yield. It is
important to note that intermediate 21 was prepared from commercially
available 4-methoxyaniline (23) by sequential diazotization
and condensation with ethyl 2-chloroacetoacetate (24). 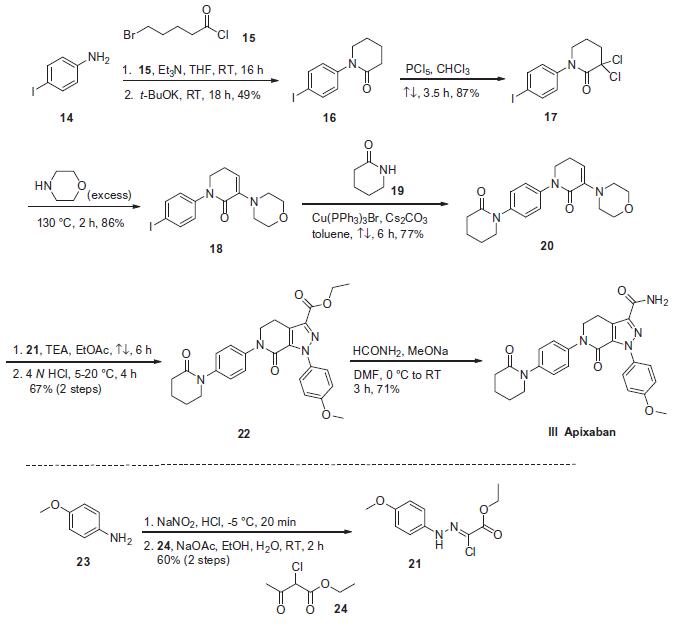
| | in vitro | apixabanhas exhibited a high degree of potency, selectivity, and efficacy on factor xa with ki of 0.08 nm and 0.17 nm for human factor xa and rabbit factor xa, respectively [1]. apixaban prolonged the clotting times of normal human plasma with the concentrations (ec2x) of 3.6, 0.37, 7.4 and 0.4 μm, which are required respectively to double the prothrombin time (pt), modified prothrombin time (mpt), activated partial thromboplastin time (aptt) and heptest. besides, apixaban showed the highest potency in human and rabbit plasma, but less potency in rat and dog plasma in both the pt and aptt assays [3]. | | in vivo | apixaban exihibited the excellent pharmacokinetics with very low clearance (cl: 0.02 l kg-1h-1), and low volume of distribution (vdss: 0.2 l/kg) in the dog. besides, apixaban also showed a moderate half-life with t1/2 of 5.8 hours and good oral bioavailability (f: 58%) [1]. in the arteriovenous-shunt thrombosis (avst), venous thrombosis (vt) and electrically mediated carotid arterial thrombosis (ecat) rabbit models, apixaban produced antithrombotic effects with ec50 of 270 nm, 110 nm and 70 nm in a dose-dependent manner[3]. apixaban significantly inhibited factor xa activity with an ic50 of 0.22 μm in rabbit ex vivo[4]. in chimpanzee, apixaban also showed small volume of distribution (vdss: 0.17 l kg-1), low systemic clearance (cl: 0.018 l kg-1h-1), and good oral bioavailability (f: 59%) [5]. | | Drug interactions | Potentially hazardous interactions with other drugs
Analgesics: increased risk of haemorrhage with IV
diclofenac and ketorolac - avoid.
Antibacterials: avoid with clarithromycin and
telithromycin; concentration possibly reduced by
rifampicin - avoid if treating DVT/PE.
Anticoagulants: increased risk of haemorrhage with
other anticoagulants - avoid.
Antidepressants: concentration possibly reduced by
St John’s wort - avoid if treating DVT/PE.
Antiepileptics: concentration possibly reduced
by carbamazepine, fosphenytoin, phenobarbital,
phenytoin and primidone - avoid if treating DVT/
PE with carbamazepine.
Antifungals: concentration increased by ketoconazole
- avoid; avoid with itraconazole, posaconazole and
voriconazole.
Antivirals: avoid with atazanavir, boceprevir,
darunavir, fosamprenavir, indinavir, lopinavir,
ritonavir, saquinavir, telaprevir and tipranavir.
Cobicistat: avoid concomitant use. | | Metabolism | Apixaban is metabolised in the liver mainly via the P450
cytochromes CYP3A4 and CYP3A5.
Apixaban has multiple routes of elimination. Of the
administered apixaban dose in humans, approximately
25% was recovered as metabolites, with the majority
recovered in faeces. Renal excretion of apixaban accounts
for approximately 27% of total clearance. There are also
additional contributions from biliary and direct intestinal
excretion. | | References |
https://en.wikipedia.org/wiki/Apixaban
https://www.drugbank.ca/drugs/DB06605
[1] Wonkyung Byon. “Apixaban: A Clinical Pharmacokinetic and Pharmacodynamic Review.” Clinical Pharmacokinetics 58 10 (2019): 1265–1279.
|
| | Apixaban Preparation Products And Raw materials |
|