|
|
| | Methylprednisolone sodium succinate Basic information |
| Product Name: | Methylprednisolone sodium succinate | | Synonyms: | 6ALPHA-METHYLPREDNISOLONE 21-HEMISUCCINATE SODIUM SALT;6-ALPHA-METHYLPREDNISOLONE SODIUM SUCCINATE;6α-methyl-11β,17α,21-trihydroxy-1,4-pregnadiene-3,20-dione 21-hemisuccinate;Methylprednisolone sodium succinate (JAN/USP);6a-Methylprednisolone 21-sodium hemisuccinate;6a-Methylprednisolone 21-sodium succinate;6a-Methylprednisolone sodium hemisuccinate;6a-Methylprednisolone sodium succinate | | CAS: | 2375-03-3 | | MF: | C26H33O8.Na | | MW: | 496.53 | | EINECS: | 219-156-8 | | Product Categories: | | | Mol File: | 2375-03-3.mol | 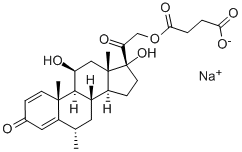 |
| | Methylprednisolone sodium succinate Chemical Properties |
| Melting point | >186°C (dec.) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | lyophilized powder | | color | White to Off-White | | InChIKey | FQISKWAFAHGMGT-JXAFMBTDNA-M | | SMILES | [C@]12(C[C@@H](C3=CC(=O)C=C[C@]3(C)[C@@]1([H])[C@H](C[C@]1(C)[C@@](CC[C@@]21[H])(O)C(=O)COC(=O)CCC([O-])=O)O)C)[H].[Na+] |&1:0,2,9,11,13,15,17,20,r| | | CAS DataBase Reference | 2375-03-3(CAS DataBase Reference) |
| Hazard Codes | T | | Risk Statements | 61-40 | | Safety Statements | 53-22-36/37/39-45 | | WGK Germany | 3 | | RTECS | TU4154060 | | Toxicity | LD50 oral in rat: > 5gm/kg |
| | Methylprednisolone sodium succinate Usage And Synthesis |
| Description |
Methylprednisolone Sodium Succinate is the sodium succinate salt of a synthetic glucocorticoid receptor agonist with immunosuppressive and anti-inflammatory effects. It is converted into active prednisolone in the body, which activates glucocorticoid receptor-mediated gene expression. This includes inducing the synthesis of the anti-inflammatory protein IkappaB-alpha and inhibiting the synthesis of nuclear factor kappa B (NF-kappaB). As a result, proinflammatory cytokine production, such as IL-1, IL-2, and IL-6, is down-regulated, and cytotoxic T-lymphocyte activation is inhibited. Therefore, an overall reduction in chronic inflammation and autoimmune reactions may be achieved.
| | Uses | 6α-Methylprednisolone 21-Hemisuccinate is a glucocorticosteroid used in anti-inflammatory medication in the treatment of spinal cord injuries. | | Uses | 6α-methylprednisolone 21-hemisuccinate sodium salt has been used to investigate glucocorticoid-induced osteonecrosis under hypoxic conditions in the MC3T3-E1 osteoblast cell line using a cell cytotoxicity assay, flow cytometry and western blotting. | | Definition | ChEBI: Methylprednisolone sodium succinate is a corticosteroid hormone. | | Brand name | A-Methapred (Abbott);
A-Methapred (Hospira); Solu-Medrol (Pharmacia & Upjohn). | | Biochem/physiol Actions | Glucocorticoid with slightly longer half-life than that of prednisolone. | | Side effects | Methylprednisolone Sodium Succinate is commonly given by injection and common side effects include: injection site pain/redness/swelling, nausea, vomiting, heartburn, headache, dizziness, difficulty sleeping, appetite changes, increased sweating, acne, increased blood sugar, sore throat, fever, chills, cough, white spots in the mouth. Serious side effects manifest as abnormal weight gain, menstrual disorders, bone/joint pain, easy bruising/bleeding, mental/mood changes, fast/slow/irregular heartbeat, stomach/intestinal bleeding symptoms, seizures, liver problems, and symptoms of severe allergic reactions. Contact your doctor if any of these serious adverse symptoms occur. |
| | Methylprednisolone sodium succinate Preparation Products And Raw materials |
|