- Ganirelix Acetate
-

- $30.00 / 1KG
-
2023-07-19
- CAS:129311-55-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 20 Tons
- Ganirelix Acetate
-

- $0.00 / 1g
-
2023-01-13
- CAS:129311-55-3
- Min. Order: 1g
- Purity: 98%min
- Supply Ability: 1000g
- Ganirelix USP/EP/BP
-
- $1.10 / 1g
-
2021-06-02
- CAS:129311-55-3
- Min. Order: 1g
- Purity: 99.9%
- Supply Ability: 100 Tons Min
|
| | Ganirelix Basic information |
| Product Name: | Ganirelix | | Synonyms: | N-Acetyl-3-(2-naphthalenyl)-D-alanyl-4-chloro-D-phenylalanyl-3-(3-pyridinyl)-D-alanyl-L-seryl-L-tyrosyl-N6-[bis(ethylaMino)Methylene]-D-lysyl-L-leucyl-N6-[bis(ethylaMino)Methylene]-L-lysyl-L-prolyl-;Ganirelix acetate salt;N-Acetyl-3-(2-naphthalenyl)-D-alanyl-4-chloro-D-phenylalanyl-3-(3-pyridinyl)-D-alanyl-L-seryl-L-tyrosyl-N6-[bis(ethylamino)methylene]-D-lysyl-L-leucyl-N6-[bis(ethylamino)methylene]-L-lysyl-L-prolyl-D-alaninamide diacetate;183552-38-7 (free base);GANIRELIX (ACETATE);183552-38-7 (FREE BASE);Ganirelix USP/EP/BP;Ganirelix Diacetate salt | | CAS: | 129311-55-3 | | MF: | C80H113ClN18O13 | | MW: | 1570.31902 | | EINECS: | | | Product Categories: | Chiral Reagents;Intermediates & Fine Chemicals;Pharmaceuticals | | Mol File: | 129311-55-3.mol | 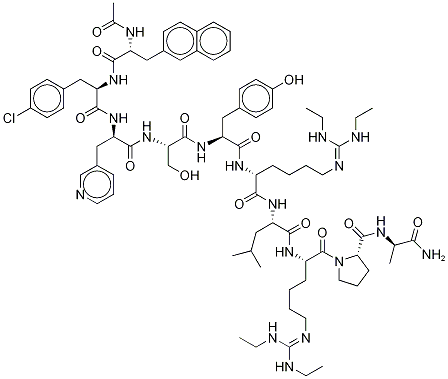 |
| | Ganirelix Chemical Properties |
| storage temp. | -20°C | | solubility | DMF: 30 mg/ml; DMSO: 30 mg/ml; Ethanol: 0.25 mg/ml; PBS (pH 7.2): 10 mg/ml | | pka | 4.2 (3-pyridinylalanine); 9.8 (tyrosine) | | form | lyophilized powder | | color | white | | InChIKey | GJNXBNATEDXMAK-PFLSVRRQSA-N |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | WGK Germany | 3 |
| | Ganirelix Usage And Synthesis |
| Description | Ganirelix acetate was introduced in Germany as prefilled syringes for subcutaneous
injections that inhibit premature luteinizing hormone surges in women undergoing
controlled ovarian hyperstimulation. This decapeptide analog of luteinizing hormonereleasing
hormone (LH-RH) is the second third-generation LH-RH antagonist to be
launched after citrorelix (Asta Medica). This highly bioavailable compound immediately
blocks the endogenous release by the pituitary gland of luteinizing hormone (LH) and
follicle-stimulating hormone (FSH), the hormone that induces ovulation. After
discontinuation of the treatment, the pituitary-gonadal function is rapidly recovered due to
its short-half life. As a consequence, ganirelix at daily doses of 0.25 mg S.C. efficiently
prevented LH surges during clinical trials in infertile women under controlled ovarian
hyperstimulation with recombinant FSH before in vitro fertilization or similar reproductive
techniques. Unlike first and second-generation gonadotropin-releasing hormone
antagonists, ganirelix has minimal histamine-releasing effects thus avoiding the formation of edema of the face and extremities. Ganirelix is very resistant to hydrolysis and, in contrast to the already established cetrorelix, has good water solubility. | | Originator | Roche Bioscience (US) | | Uses | Decapeptide LH-RH antagonist. Used in treatment of infertility. | | Manufacturing Process | The abbreviations for common aminoacids are those recommended by IUPAC�IUB Comission on Biochemical Nomenclature. Other abbreviations useful in
describing the replacements of aminoacids in the natural LH-RH peptide are
following:
Nal(2) - 3-(2-naphthyl)alanyl; p-Cl-Phe - 3-(p-chlorophenyl)alanyl; Pal(3) - 3-
(3-pyridyl)alanyl; ; hArg(Et)2 - NG,NG - bis(ethtyl)homoarginyl; Boc - t�butyloxycarbonyl.
Ganirelix (N-Ac-Nal(2)-D-pCl-Phe-D-Pal(3)-Ser-Tyr-D-hArg(Et)2-Leu-hArg(Et)2-
Pro-Ala-NH2) was prepared using the following side chain protection protocol:
salt protection for L- and D-hArg(Et)2 (as the chloride) and t-butyl protection
for serine.
Amino acids were added to the Nα-Boc-D-Ala-O-Resin (1.0 mmol of resin was
replaced in the reaction vessel of 5.0 L Vega 296 automated solid phase
peptide synthesizer; in the following sequence:
Acetic anhydride
An acetylation (capping of the resin) was done after Ala, Pro and Leu with
N,N'-diisopropyl carbodiimide - 1-hydroxybenztriazole (HBt). Excess HBt (2
equiv.) was used for the coupling of the basic amino acids, hArg(Et)2 and
Pal(3).
The following protocols were used to remove the Nα-protecting group following
each addition.
Program A: The resin was first washed with CH2Cl2 1 times/1 min, TFA-CH2Cl2
(40/60) 1 times/1 min, TFA-CH2Cl2 (40/60) 1 times/30 min, CH2Cl2 2 5
times/1 min, Et3N-CH2Cl2 (5/95) 3 times/1 min, CH2Cl2 4 times/1 min.
Program B: The resin was first washed with CH2Cl2 1 times/1 min, 4-4.5 N
HCl in CH2Cl2/i-PrOH (1/1) 1 times/1 min, 4-4.5 N HCl in CH2Cl2/i-PrOH (1/1)
1 times/30 min, CH2Cl2 3 times/1 min, DMF 1 times/1 min, Et3N-CH2Cl2
(5/95) 3 times/1 min, DMF 1 times/1 min, CH2Cl2 4 times/1 min.
After each deprotecting and washing step, following protocol A or B, the next
amino acid in sequence was added and the resin washed with CH2Cl2 3
times/1 min, MeOH 4 times/1 min, DMF 2 times/1 min and CH2Cl2 4 times/1
min.
Program A was used for the removal of the protecting groups on Ala, Pro, L�hArg(Et)2, Leu and D-Nal(2).
Program B was used for the removal of the protecting groups on D-hArg(Et)2,
Tyr, Ser, D-Pal(3) and p-Cl-Phe.
The crude peptide was first dissolved in 2 M acetic acid and converted to its acetate salt by passage through a column of AG3-X4A resin (Bio-Rad). The
acetate was subjected to chromatography on a silica gel column (CH2Cl2/i�PrOH/MeOH/H2O/HOAc solvent); the acetate fractions dissolved in H2O and
loaded onto a reversed-phase column (Vydec C-18, 15-20 μ), and purified
using acetonitrile/TEAP (pH 3). Fractions of the desired purity were combined
and diluted with water and reloaded on a reversed-phase HPLC column, then
washed with 1% acetic acid in water. The peptide was stripped with a mixture
of MeOH/CH3CN/HOAc/H2O (44/50/1/5). The residue was dissolved in acetic
acid and precipitated over ether, filtered, washed with ether and dried under
vacuum. Amino acid analyses were performed on a Beckman 119CL amino
acid analyzer. Samples for amino acid analyses were hydrolyzed with 6 N HCl
at 110°C for 20 hrs. Analytical HPLC was performed on a Spectra Physics
8800 chromatograph. Synthesis of ganirelix was confirmed by the presence of
a main peak at rt 18 min; no other peak over 1% was noted at rt 16 min. | | Brand name | Orgalutran | | Therapeutic Function | LHRH antagonist | | Biochem/physiol Actions | Ganirelix is a decapeptide GnRH antagonist. Ganirelix acts by blocking the action of GnRH upon the pituitary, thus rapidly suppressing the production and action of LH and FSH. |
| | Ganirelix Preparation Products And Raw materials |
|