- Dimethyl sulfoxide
-

- $50.00 / 1kg
-
2024-09-20
- CAS:67-68-5
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 500ton
- Dimethyl sulfoxide
-

- $6.00 / 1kg
-
2024-09-20
- CAS:67-68-5
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 2000KG/Month
- Dimethyl sulfoxide
-

- $0.00 / 1KG
-
2024-09-20
- CAS:67-68-5
- Min. Order: 1KG
- Purity: >99% GC
- Supply Ability: 1000KG
|
| Product Name: | Dimethyl sulfoxide | | Synonyms: | DIMETHYL SULFOXIDE;Methyl sulfoxide, for analysis ACS, 99.9+%;Methyl sulfoxide, water <50 ppm, extra dry over mol. sieve, 99.7+%;Methyl sulfoxide, water <50 ppm, extra dry, 99.7+%;METHYL SULFOXIDE Distilled in quartz solvent;METHYL SULFOXIDE, REAGENT (ACS);syntexan,sulfinylbis-methane,dimethylsulfoxide,sclerosol,kemsol;dimethyl sulfoxide, hplc grade | | CAS: | 67-68-5 | | MF: | C2H6OS | | MW: | 78.13 | | EINECS: | 200-664-3 | | Product Categories: | Dimethyl Sulfoxide (DMSO);Anhydrous Solvents;Solvent by Application;Solvent by Type;Solvents;Plastic Bottles;Analytical Reagents;Analytical Reagents for General Use;Analytical/Chromatography;C-D;Puriss p.a. ACS;Solvent Bottles;Solvent Packaging Options;BRDU Cell Proliferation Assay Reagents;Cell Culture;Cell Freezing Media and Reagents;Chemical ER Chaperones;Endoplasmic Reticulum Stress;General Stem Cell Biology;Hybridoma Platform;Hybridoma Reagents;Life Science Reagents for Cell Culture;Reagents and Supplements;Biochemicals;Enzymes;-Glucuronidase;-Glucuronidase Drug Analysis Bundle;Inhibitors;Life Science Reagents for RNAi;Anhydrous;ACS and Reagent Grade Solvents;ACS Grade;ACS Grade Solvents;Amber Glass Bottles;Composite Drums;Drums Product Line;Semi-Bulk Solvents;VerSA-Flow Products;CHROMASOLV Plus;Chromatography Reagents &;HPLC &;HPLC Plus Grade Solvents (CHROMASOLV);HPLC/UHPLC Solvents (CHROMASOLV);NOWPak Products;UHPLC Solvents (CHROMASOLV);Solvents for Spectrophotometry;Sulfur Compounds (for Synthesis);Synthetic Organic Chemistry;Serum-free Media;Stem Cell Biology;Cell Biology;Cell Signaling and Neuroscience;Core Bioreagents;Cryopreservation;General Reagents;Research Essentials;Apoptosis and Cell Cycle;Life Science Reagents for Nucleic Acid Purification;Life Science Reagents for Protein Expression and Purification;Other Apoptosis Related Reagents;Analytical Chemistry;Solvents for HPLC & Spectrophotometry;Organics;INORGANIC & ORGANIC CHEMICALS;Pharmaceutical Intermediates;Biostabilization Agents;PCR Optimization;Stabilization of Biomolecules;Biochemicals and Reagents;Life Science Reagents for PCR;Molecular Biology;PCR Reaction Components;PCR/Amplification;Plant Biotechnology;Plant Growth Regulators;Plant Tissue Culture;Primary Human Cell Culture;Specialty Media Systems;Hepatocytes;Biotech;Biotech Solvents;Life Science Reagents for Northern and Southern Blotting;Products;Returnable Containers;Sure/Seal Bottles;DMSO - Dimethyl sulfoxide;Molecular Biology Reagents;Analytical and Industrial Enzymes;and Substrates;Universal Solvent;67-68-5;BDO;bc0001;TOP2 | | Mol File: | 67-68-5.mol | 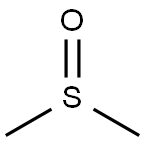 |
| | Dimethyl sulfoxide Chemical Properties |
| Melting point | 18.4 °C | | Boiling point | 189 °C(lit.) | | density | 1.100 g/mL at 20 °C | | vapor density | 2.7 (vs air) | | vapor pressure | 0.42 mm Hg ( 20 °C) | | FEMA | 3875 | METHYLSULFINYLMETHANE | | refractive index | n20/D 1.479(lit.) | | Fp | 192 °F | | storage temp. | Store at +5°C to +30°C. | | solubility | H2O: miscible (completely) | | pka | 35(at 25℃) | | form | liquid (temperature dependent) | | color | clear colorless | | Relative polarity | 0.444 | | Odor | Mild garlic odor | | explosive limit | 1.8-63.0%(V) | | Odor Type | alliaceous | | Water Solubility | Soluble in water, methanol, acetone, ether, benzene, chloroform. | | FreezingPoint | 18.4℃ | | Sensitive | Hygroscopic | | λmax | λ: 285 nm Amax: ≤0.20
λ: 295 nm Amax: ≤0.20 | | JECFA Number | 507 | | Merck | 14,3259 | | BRN | 506008 | | Dielectric constant | 41.9(55℃) | | Stability: | Stable. Incompatible with a very wide range of materials, including acid chlorides, strong acids, strong oxidizing agents, strong reducing agents, phosphorus halides, moisture, copper wool + trichloroacetic acid. Reacts violently with a number of materials - consult a full data sheet before use. Hygroscopic. | | InChIKey | IAZDPXIOMUYVGZ-UHFFFAOYSA-N | | LogP | -1.35 | | CAS DataBase Reference | 67-68-5(CAS DataBase Reference) | | NIST Chemistry Reference | Dimethyl sulfoxide(67-68-5) | | EPA Substance Registry System | Dimethyl sulfoxide (67-68-5) |
| | Dimethyl sulfoxide Usage And Synthesis |
| Overview | Dimethyl sulfoxide (abbreviated DMSO) is a sulfur-containing organic compound; molecule formula: (CH3) 2SO; It exhibits as colorless, odorless, hygroscopic and flammable transparent liquid at room temperature. It has both high polarity as well as high-boiling point. It also has aprotic and water-miscible characteristics. It has low toxicity, good thermal stability, and is not miscible with paraffin. It is soluble in water, ethanol, propanol, ether, benzene and chloroform and many other kinds of organic substance and is called as the "universal solvent." It is a common organic solvent which has the strongest dissolving ability. It can dissolve most the organic compounds including carbohydrates, polymers, peptides, as well as many inorganic salts and gases. It can dissolve certain amount solute whose weight is equals to 50-60% of itself (other common solvents usually only dissolve 10-20%), so it is very important in the sample management as well as high-speed screening of drugs. Under certain conditions, contact between dimethyl sulfoxide and chloride can even lead to explosive reaction.
dimethyl sulfoxide is widely used as solvents and reagents, particularly as the processing reagent and spinning solvent at the reaction of acrylonitrile polymerization used for polyurethane synthesis and the spinning solvent. It can also be used as the synthetic solvent for polyamide, polyimide and polysulfone resin as well as the extraction solvents for aromatic hydrocarbon and butadiene extraction solvents and solvents for synthesizing chlorofluoroaniline. In addition, dimethyl sulfoxide can also be directly used as the raw material or carrier of some drug in the pharmaceutical industry. dimethyl sulfoxide itself has various effects such as anti-inflammatory, analgesic, diuretic, and sedative, and thus also being known as "panacea" which is usually as the active ingredient of analgesic medication being added to the drug. It also has special properties of easy penetration through the skin, resulting in personnel feeling of oyster-like flavor. The dimethyl sulfoxide solution of sodium cyanide can cause cyanide poisoning through skin contact while dimethyl sulfoxide itself has a relative low toxicity. dimethyl sulfoxide is widely applied as the extraction agent in a lot of chemical and pharmaceutical companies. However, because of its higher boiling point, it causes very high operating temperatures and further causes the coking of materials, thus affecting the recycling of dimethyl sulfoxide and further cleaning of the equipments as well as resulting in increased energy consumption. Therefore, the recycling of dimethyl sulfoxide has become a bottleneck as its further wide application as extraction agent.
The above information is edited by the Chemicalbook of Dai Xiongfeng.
| | Method of purification and water removal | dimethyl sulfoxide can be mixed with water and can be placed for long term storage and drying through molecular sieve. Further apply it to vacuum distillation and collect the fraction of 76 °C/1600Pa (12mmHg). Upon distillation, keep the temperatures at below 90 °C; otherwise it will result in disproportionation reaction which generates dimethyl sulfone and dimethyl sulfide. It may also be dried through calcium oxide, calcium hydroxide, barium oxide and barium sulfates and then distilled under reduced pressure. It may also be purified through partially crystallization. It can lead to explosion when mixing the dimethyl sulfoxide with certain substance such as sodium hydride, periodic acid or magnesium perchlorate, and thus should be paid attention.
| | Identification and detection methods | [Identification] Take 1.5 mL of this product and add to it slowly of 2.5 mL cooled hydroiodic acid drop wise; rapidly filter it and dry it under reduced pressure with the residue being unstable dark purple crystalline solid which has an unpleasant smell. Dissolving it in chloroform will form a red solution.
[Check] Optical absorption, take certain amount of this product, pour through the stream of dry nitrogen for 15 minutes and immediately place it in 1cm absorption cuvette with water as a blank. Measure the absorbance of 275 nm according to spectrophotometric method (Chinese Pharmacopoeia 1990 edition of Appendix page 24) with a value being not higher than 0.30; and then further measure absorbance at wavelength of 285nm and 295nm with its ratio over that of the absorbance of 275nm being not greater than 0.65 and 0.45, respectively. Also note that, there should be no maximal absorption in the 270~350nm range. The water content should not exceed 0.2% (Chinese Pharmacopoeia 1990 edition of Appendix 55).
Dimethyl sulfone take diphenyl methane to make a 0.025% acetone solution as the internal standard solution; accurately weigh appropriate amount of dimethyl sulfoxide and dilute to 0.050% solution of internal standard solution as the control solution; in addition, accurately weigh certain amount of this product and dilute to 50% solution with internal standard solution as the test solution. According to gas chromatography protocol (Chinese Pharmacopoeia 1990 edition, second volume, appendix page 31(3)), use 20 mM of 10% (coating concentration) polyethylene glycol as the fixer liquid; (the theoretical plate number calculated from dimethyl should be greater than 1500, and the separation degree of dimethyl sulfoxide peak and the internal standard peak should be greater than 2). The measured ratio of dimethyl sulfone peak area and diphenyl methane peak area of the sample solution at a column temperature of 150 °C should not be greater than that of the control solution.
| | Reference quality standards | For external use-DMSO quality of technical indicators
Content, GC,%, ≥ 99.9 GB/2307-1986) Gas Chromatography
IR identification, qualified EP
Chemical identification, qualified EP
Crystallization point, °C ≥ 18.3 GB/T7533-1993
The refractive index of 1.478-1.479 GB/T6488-1986
Moisture,%, KF, ≤ 0.2 GB/T6283-1986
UV extinction values, 275nm ≤ 0.3 GB 9721-1988
285nm ≤ 0.2
295nm ≤ 0.2
Heavy metals, PPm ≤ 10 GB/7532-1987
Specific gravity (20 ℃), 1.100-1.104 GB/4472-1984
Acidic value (0.1N/ml NaOH/50g), ≤ 5.0 GB/9736-1988
Packaging: PE plastic bucket
Anhydrous-Technical index of DMSO quality
Content (DMSO), GC%, ≥ 99.9 GB/2307---1986) Gas Chromatography
Crystal point, °C, ≥ 18.35 GB/T7533-1993
Acidic value mgKOH/g, ≤ 0.001 GB/T9736-1988
Refractive index (20 °C), 1.478-1.479 GB/6488---1986
Moisture,%, KF, ≤ 0.001 GB/6283--1986
Evaporation residue, PPM, ≤ 5. GB/T3209----82
Heavy metals,%, ≤ 10. GB/T7532----87
Package: PE plastic bucket
| | Toxicity | GRAS (FEMA).
| | Limited use | FEMA (mg/kg) bakery products, non-alcoholic beverages, breakfast cereals, cold drinks, sauces, meat products, dairy products, soft candy, soups, sweet sauces; 0.1 to 5.0 in average; Spices: 100-1000.
| | Recovery process | We should take recycling into account when using DMSO as a solvent in order to reduce the production cost. We can take advantage of DMSO’s property of being easily soluble in water, benzene, and toluene to generate association complex to separate it from other impurities using ether extraction or reverse extraction, and further apply vacuum distillation to get pure product. For example: for the mixture between DMSO and the inorganic salt, use toluene to extract DMSO out, and then further add water to the extraction liquid for reverse extraction of DMSO. The resulting DMSO aqueous solution is subject to vacuum distillation at reduced pressure ((1.33~2.00) × 103Pa to obtain the DMSO of over 99%. Upon the extraction with arene, first extract out the arene from reformate; further add water to the extracted liquid for reverse extraction to obtain the aqueous DMSO. Further get the pure DMSO product through vacuum distillation. In addition, there are also some other methods such as salting out with potassium dihydrogen phosphate, recycling method via methanol, ethanol extraction which can be adopted under certain conditions. For user of small quantities which is not easy to recycle can pile them up and commit to the production plant for recycling together.
The basic approach of DMSO recycling is through vacuum distillation with the requirement of two conditions, first, keep the temperature below 120 degrees, otherwise there will be security problems, and will also cause lower recovery. Another issue is the PH value of the system. An appropriate value is pH 8 which is suitable for both safety issue as well as the proper decomposition condition of DMSO. The tower I used is carbon steel with 7-8 trays and the reflux ratio being 3-2 which can generally reach 98-99% content. The device volume is 2000 L with a very good efficacy.
Semi-continuous vacuum distillation method for recovering the DMSO in ketoconazole production is as follows: (1) pour the raw material into the distillation column reactor, start heating when the liquid level reaches 1/3; ( 2) when the temperature of the top of rectification column top temperature reached 60 °C, turn on the internal reflux cooler as well as the vacuum system; (3)When the degree of vacuum reaches 0.1~0.15 × 10 Pa ↑, control the top of the distillation column of yielding material: when the top temperature reached 65 °C, the top of the rectification column mainly produces the semi-finished material; when the top temperature reached 80 °C, and also there was rapid increase in temperature of the top, the yielding material in top is DMSO; (4) When the temperature reaches 110 °C, vent and stop heating; (5) release the residue in the tower kettle.
| | Chemical Properties | It is colorless liquid, and is hygroscopic. It is almost odorless with a bitter taste. It can be dissolved in water, ethanol, acetone, ether, benzene and chloroform.
| | Uses | 1. It can be used for the extraction of arene, also as the reaction medium used for resins and dyes, and applied to acrylic polymerization and spinning solvent.
2. It can be used as an organic solvent, reaction medium and the intermediates of organic synthesis. It is highly versatile. This product has a highly selective extraction capacity, and can be used as the polymerization and condensation solvent of acrylic resin and polysulfone resin, as the polymerization and spinning solvent of polyacrylonitrile and cellulose acetate, as the extraction solvent for separating alkanes and arenes, and as the reaction medium for the arenes, butadiene extraction, acrylic fiber, plastic solvents, organic and synthetic dyes, and pharmaceuticals industries. In the field of medicine, dimethyl sulfoxide has anti-inflammatory and analgesic effect with a strong capability of penetration through the skin, and thus being able to dissolve certain drugs and boost their penetration into the human body to achieve the therapeutic purposes. Taking this carrier property of dimethyl sulfoxide can make it be used as pesticide additives. Add a small amount of dimethyl sulfoxide in some pesticides can facilitate the penetration of pesticides into the plant in order to improve the efficacy. dimethyl sulfoxide can also be used as the dye solvent, dye removing agent, and dye carrier for the synthetic fibers. It can also be used as the absorbent of recycling acetylene and sulfur dioxide and also the modifiers of synthetic fiber, antifreeze agent and the capacitor dielectric, brake oil, and extractant of the rare metals.
3. It can be used as analytic solvents and fixing agent of gas chromatography as well as the solvent for analyzing UV spectra.
| | Production method | dimethyl sulfoxide is generally made by using dimethyl sulfide oxidation method. They have different production processes due to the different oxidants and oxidation types. 1. Methanol carbon disulfide method; take carbon disulfide and methanol as raw materials and use γ-Al2O3 as the catalyst; first synthesize the dimethyl sulfide, then have oxidation reaction with nitrogen dioxide (or nitrate) to obtain dimethyl sulfoxide. 2. The hydrogen peroxide method: take acetone as the buffer medium to have dimethyl sulfide be reacted with hydrogen peroxide. This method of dimethyl sulfoxide synthesis has a relative high cost and thus is suitable for large-scale production. 3. The method of nitrogen dioxide: have methanol and hydrogen sulfide dimethyl sulfide reacted in the catalysis of γ-alumina to obtain dimethyl sulfide; have sulfate reacted with sodium nitrite to generate nitrogen dioxide; dimethyl sulfide has gas-liquid phase reaction with nitrogen dioxide at 60-80 °C to produce crude dimethyl sulfoxide. Sometimes people also use oxygen for direct oxidation which also generates the crude dimethyl sulfoxide. Then after vacuum distillation, refine to get the finished product of dimethyl sulfoxide. This method is a relative advanced production method. 4. Dimethyl sulfate method: apply dimethyl sulfate to react with sodium sulfide to obtain dimethyl sulfide; sulfate has reaction with sodium nitrite to generate nitrogen dioxide; dimethyl sulfide is oxidized with nitrogen dioxide to generate the crude dimethyl sulfoxide, and then undergo neutralization and distillation to obtain refined dimethyl sulfoxide product. In addition, the anodic oxidation method can also produce dimethyl sulfoxide via dimethyl sulfide.
| | Description | First synthesized in 1866 by Alexander Zaytsev in the Russian
Empire, dimethyl sulfoxide is an organosulfur compound. This
colorless liquid is an important polar aprotic solvent that
dissolves both polar and nonpolar compounds and is miscible
in a wide range of organic solvents as well as water. | | Chemical Properties | Dimethyl sulfoxide occurs as a colorless, viscous liquid, or as colorless crystals that are miscible with water, alcohol, and ether. The material hasa slightly bitter taste with a sweet aftertaste, and is odorless, or has a slight odor characteristic of dimethyl sulfoxide.Dimethyl sulfoxide is extremely hygroscopic, absorbing up to 70% of its own weight in water with evolution of heat. | | Originator | Rimso,Research,US,1978 | | Occurrence | Reported found in apple, raspberry, cabbage, cucumber, onion, tomato, peppermint and spearmint oils, milk,
pork liver, beer, rum, cocoa, coffee, black tea, oatmeal, soybean, beetroot, parsnip root, watercress, sweet corn, malt, cooked shrimp
and oysters | | Uses | Suitable for HPLC, spectrophotometry, environmental testing | | Uses | A polar aprotic solvent used in polymerase chain reactions (PCR) and as a cryoprotectant vitrification agent for the preservation of cells, tissues and organs. | | Uses | DMSO is a polar aprotic solvent used in chemical reactions, in polymerase chain reactions (PCR) and as a cryoprotectant vitrification agent for the preservation of cells, tissues and organs. DMSO is used in cell freezing media to protect cells from ice crystal induced mechanical injury. It is used for frozen storage of primary, sub-cultured, and recombinant heteroploid and hybridoma cell lines; embryonic stem cells (ESC), and hematopoietic stem cells. DMSO is frequently used in the combinations with BSA or fetal bovine serum (FBS). | | Uses | Human and animal cell lines grown in culture are generally stored frozen. Freezing protects the cell line from changes due to genetic drift and minimizes risk of contamination. Liquid nitrogen used in conjunction with a cryoprotective agent such as DMSO is a widely used method for preserving cells. Without the presence of a cryoprotective agent, freezing is lethal to most mammalian cells. Damage is caused by mechanical injury by ice crystals, concentration of electrolytes, pH changes, and denaturation of proteins. These lethal effects are minimized by adding a cryoprotective agent which lowers | | Uses | Dimethyl sulfoxide (1-10%) has been shown to accelerate strand renaturation and is believed to give the nucleic acid thermal stability against depurination. As a PCR cosolvent, DMSO may help improve yields, especially in long PCR. | | Uses | Solvent for many organic Compounds including fats, carbohydrates, dyes, resins, and polymers. In organic reactions. As antifreeze or hydraulic fluid when mixed with water. To cryopreserve and store cultured cells.
Dimethyl Sulfoxide is a common aprotic organic solvent used to dissolve both polar and nonpolar compounds. Used primarily for NMR study in its deuterated form DMSO-d6 (D479382) due to its ability to dissolve most analytes as well as its easily identifiable NMR spectrum. | | Definition | ChEBI: A 2-carbon sulfoxide in which the sulfur atom has two methyl substituents. | | Production Methods | Dimethyl sulfoxide is prepared by air oxidation of dimethyl sulfide in the presence of nitrogen oxides. It can also be obtained as a by product of wood pulp manufacture for the paper and allied industries. | | Manufacturing Process | A current of oxygen at the rate of 370 ml/min was bubbled through a 30-cm
layer of dimethyl sulfide maintained at 26.5°C, thereby producing a gaseous
mixture containing the stoichiometric amount of oxygen required for the
oxidation of the sulfide to sulfoxide. Nitric oxide at the rate of 30 ml/min was
added to the gaseous mixture as it passed into the first of a series of four
reaction chambers, each consisting of a glass tube 4.3 cm in diameter and
100 cm in length. The reaction started immediately, the temperature of the reaction mixture reached a maximum of about 75°C in the first two tubes
where most of the reaction occurred, and the reaction slowed down in the last
two tubes. The crude, yellow product, which dropped from the tubes,
contained about 10% dimethyl sulfide, about 2% dissolved nitrogen dioxide,
about 2% methane sulfonic acid, and some water. The crude product was
refluxed at 100°C for 30 minutes and the escaping gas was passed into the
first reaction chamber. The dimethyl sulfide was removed by then heating the
product to 150°C, the methane sulfonic acid was neutralized by adding slaked
lime, and the dimethyl sulfoxide was distilled in vacuum. The yield of pure
dimethyl sulfoxide (BP 63°C at 6 mm Hg) was 85% of the theoretical yield
from the evaporated dimethyl sulfide. | | Brand name | Rimso-50 (Bioniche). | | Therapeutic Function | Topical antiinflammatory | | Aroma threshold values | Aroma characteristics at 100%: fatty, oily, cheesy, over-cooked stir-fry, salty and garlic, mushroom, garlic
with a cracker and soy nuance | | Taste threshold values | Taste characteristics at 40 ppm: fatty, salty, garlic and cream corn | | General Description | A clear liquid, essentially odorless. Closed cup flash point 192°F. Vapors are heavier than air. Contact with the skin may cause stinging and burning and lead to an odor of garlic on the breath. An excellent solvent that can transport toxic solutes through the skin. High vapor concentrations may cause headache, dizziness, and sedation. | | Air & Water Reactions | Denser than water and miscible in water. | | Reactivity Profile | Dimethyl sulfoxide decomposes violently on contact with many acyl halides, aryl halides and related compounds such as phenyl and tolyl chloride, acetyl chloride, benzenesulfonyl chloride, benzoyl chloride, cyanuric chloride, phosphorus chloride, phosphorus oxychloride, and thionyl chloride [Chem. Eng. News 35(9):87 (1957)]. Reacts, possibly violently, with iodine pentafluoride [Chem. Eng. News 47(12):, 109(1969)]. Vacuum distillation from anhydrous magnesium perchlorate led to an explosion [MCA Case History 1187(1966)]. Violently reactive with fluorinating agents such as silver fluoride [Chem. Eng. News 44(24):7(1956)]. Can explode with sodium hydride [Chem. Eng. News 44(24):7(1966)]. Mixture with methyl bromide resulted in an explosion that shattered the apparatus [NFPA 491M, 1991]. Forms salts with perchloric acid that are explosive when dry [Chem. Abst. 44:p3935d (1950)]. Decomposes when heated above normal boiling point. | | Health Hazard | The acute toxicity of DMSO by all routes of exposure is very low. Inhalation of
DMSO vapor can cause irritation of the respiratory tract, and at higher
concentrations may cause vomiting, chills, headache, and dizziness. The material is
only slightly toxic by ingestion and may cause vomiting, abdominal pain, and
lethargy. Dimethyl sulfoxide is relatively nontoxic by skin absorption, but can cause
itching, scaling, and a transient burning sensation. Dimethyl sulfoxide can increase
the tendency for other chemicals to penetrate the skin and so increase their toxic
effects. Contact of DMSO liquid with the eyes may cause irritation with redness,
pain, and blurred vision.
Chronic exposure to dimethyl sulfoxide can cause damage to the cornea of the eye.
Dimethyl sulfoxide has not been found to be carcinogenic or to show reproductive or
developmental toxicity in humans | | Flammability and Explosibility | Combustible when exposed to heat or flame (NFPA rating = 1). Carbon dioxide or dry chemical extinguishers should be used to fight DMSO fires. | | Chemical Reactivity | Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent. | | Pharmaceutical Applications | Dimethyl sulfoxide is a highly polar substance that is aprotic, therefore lacking acidic and basic properties. It has exceptional solvent properties for both organic and inorganic components, which are derived from its capacity to associate with both ionic species and neutral molecules that are either polar or polarizable. Dimethyl sulfoxide enhances the topical penetration of drug sowing to its ability to displace bound water from the stratumcorneum;this is accompanied by the extraction of lipids and configurational changes of proteins.
the solvent is diluted. Increases in drug penetration have been reported with dimethyl sulfoxide concentrations as low as 15%,but significant increases in permeability generally require concentrations higher than 60–80%. Furthermore, while low molecular weight substances can penetrate quickly into the deep layers of the skin, the appreciable transport of molecules with a molecular weight of more than 3000 is difficult.
Dimethyl sulfoxide is now incorporated into a number of regulated products for healthcare and drug delivery applications, including stabilizing product formulations, sustained-release applications, and for the delivery of medical polymers.
The use of dimethyl sulfoxide to improve transdermal delivery has been reported for diclofenac, ciclosporin, timolol, and a wide range of other drugs. Dimethyl sulfoxide has also been used in the formulation of an injection containing allopurinol. It has also been investigated for use in an experimental parenteral preparation for the treatment of liver tumors.
In paint formulations of idoxuridine, dimethyl sulfoxide acts both as a solvent to increase drug solubility and a means of enabling penetration of the antiviral agent to the deeper levels of the epidermis.
Dimethyl sulfoxide has also been investigated as a potential therapeutic agent in conditions such as scleroderma, interstitial cystitis,(12) rheumatoid arthritis, and acute musculoskeletal injuries, and as an analgesic.(13–17) It has also been recommended for the treatment of anthracycline extravasation(18–21) and has been investigated as a potential cryoprotectant. | | Industrial uses | Dimethyl sulfoxide, a product of an oxidation reaction on dimethyl sulfide, contains a very
polar sulfoxide functional group. This highly polar functional group enables DMSO
to form complexes with many metal ions, to act as a reaction medium for synthetic
reactions, and to dissolve a large number of organic resins and polymers.
Dimethyl sulfoxide is more viscous than
many organic solvents and has a rather high surface tension value of 42.3
dynes/cm. The most outstanding property of DMSO is its very high polarity
character.
Dimethyl sulfoxide is used as a reaction solvent in the polymerization of
acrylonitrile with a vinyl monomer (e.g., styrene). Reaction of diisocyanates and
polyols or polyamines dissolved in DMSO yield solutions of the polyurethanes.
Mixtures of DMSO and water are used as a spinning solvent for polymer fibers.
Polyaryl ether polymers are formed from DMSO solutions containing the reactants
bis(4,4-chlorophenyl sulfone) and the disodium salt of dihydroxyphenol.
Dimethyl sulfoxide is a favored solvent for displacement reactions in synthetic chemistry. The rates of reaction in DMSO are many times faster than in an alcohol
or aqueous medium. Dimethyl sulfoxide is the solvent of choice in reactions where
proton (hydrogen atom) removal is the rate determining step. Reactions of this type
include olefin isomerizations and reactions where an elimination process produces
an olefin. Another application that uses DMSO is its use as an extraction solvent to
separate olefins from saturated paraffins. Several binary and ternary solvent
systems containing DMSO and an amine (e.g., methylamine), sulfur trioxide,
carbon disulfide/amine, or sulfur trioxide/ammonia are used to dissolve cellulose,
and act as spinning baths for the production of cellulose fibers. Organic fungicides,
insecticides, and herbicides are readily soluble in DMSO. Dimethyl sulfoxide is
used to remove polymer residues from polymerization reactors. | | Biological Activity | Solvent with wide ranging applications in biological research. | | Biochem/physiol Actions | Dimethyl sulfoxide (DMSO) is a low molecular weight, less hydrophilic and a freely permeable cryoprotectant. It is a polar aprotic solvent used in polymerase chain reactions (PCR) and as a cryoprotectant and vitrification agent for the preservation of cells, tissues and organs. | | Safety Profile | Slightly toxic by
ingestion. Moderately toxic by intravenous
and intraperitoneal routes. Human systemic
effects by intravenous route: nausea or
vomiting and jaundice. Experimental
teratogenic and reproductive effects. A
skin and eye irritant. Questionable
carcinogen with experimental tumorigenic
data. Human mutation data reported. Can
cause an anaphylactic reaction. Corneal
opacity reported only in rabbits, dogs, and
pigs. It freely penetrates the skin and may
carry dissolved chemicals with it into the
body. Combustible when exposed to heat or flame; can react with oxidizing materials. To
fight fire, use water, foam, alcohol foam,
CO2, dry chemical. Violent or explosive
reaction with many acyl, aryl, and nonmetal
halides (e.g., acetyl chloride, benzenesulfonyl
chloride, bromobenzoyl acetadde, cyanuric
chloride, iodine pentafluoride, Mg(ClO4)2,
CH3Br, NI04, oxalyl chloride, P203,
phosphorus trichloride, phosphoryl
chloride, silver fluoride, silver difluoride,
sodium hydride, sulfur dichloride, disulfur
dichloride, sulfuryl chloride,
tetrachlorosilane, thionyl chloride). Violent
or explosive reaction with boron compounds (e.g., borane,
nonahydrononaborate(2-) ion), 4(4'
bromobenzoyl)acetanilide, carbonyl
dhsohocyanate, dinitrogen tetraoxide,
hexachlorocyclotriphosphazine, copper +
trichloroacetic acid, metal alkoxides (e.g.,
potassium tert-butoxide, sodium
isopropoxide), trifluoroacetic acid
anhydride. Incompatible with magnesium
perchlorate, metal oxosalts, perchloric acid,
periodc acid, sulfur trioxide. Forms
powerfully explosive mixtures with metal
salts of oxoacids (e.g., aluminum
perchlorate, sodrum perchlorate, iron(IⅡ)
nitrate). When heated to decomposition it
emits toxic fumes of SOx | | Safety | Dimethyl sulfoxide has low systemic toxicity but causes local toxic effects. It is readily absorbed after injection or after oral or percutaneous administration and is widely distributed throughout the body. Dimethyl sulfoxide acts as a primary irritant on skin, causing redness, burning, itching, and scaling; it also causes urticaria. Systemic symptoms include nausea, vomiting, chills, cramps, andlethargy;dimethyl sulfoxidecanalsocauseincreases in intraocular pressure. Administration of dimethyl sulfoxide by any route is followed by a garlic-like odor on the breath.
Intravascular hemolysis and biochemical changes and reversible neurological deterioration have been reported following intravenous administration; however, it has been questioned whether these findings were directly attributable to dimethyl sulfoxide rather than to concomitant drug therapy or contaminants. One report describes massive intracranial hemorrhage associated with ingestion of dimethyl sulfoxide. Recently, a hypersensitivity reaction attributed to dimethyl sulfoxide has been reported.
In 1965, the FDA banned investigation in humans of dimethyl sulfoxide owing to the appearance of changes in the refractive index of the lens of the eye in experimental animals.However,in1966,the FDA allowed the study of dimethyl sulfoxide in serious conditions suchasscleroderma,persistent herpes zoster,and severe rheumatoid arthritis, and in 1968 permitted studies using short-term topical application of the solvent. By 1980, the FDA no longer specifically regulated investigations of dimethyl sulfoxide.
Dimethyl sulfoxide enhances the skin penetration of several drugs, which may result in producing the adverse effects associated with those drugs.
LD50 (dog, IV): 2.5g/kg
LD50 (rat, IP): 8.2g/kg
LD50 (rat, IV): 5.3g/kg
LD50 (rat, oral): 14.5g/kg
LD50 (rat, SC): 12g/kg
LD50 (mouse, IP): 2.5g/kg
LD50 (mouse, IV): 3.8g/kg
LD50 (mouse, oral): 7.9g/kg | | Veterinary Drugs and Treatments | Purported uses for DMSO are rampant, but the only FDA-approved
veterinary indication for DMSO is: “…as a topical application to
reduce acute swelling due to trauma” (Package Insert; Domoso?—
Syntex). Other possible indications for DMSO include: adjunctive
treatment in transient ischemic conditions, CNS trauma and cerebral
edema, skin ulcers/wounds/burns, adjunctive therapy in intestinal
surgeries, and analgesia for post-operative or intractable pain,
amyloidosis in dogs, reduction of mammary engorgement in the
nursing bitch, enhancement of antibiotic penetration in mastitis in
cattle, and limitation of tissue damage following extravasation injuries
secondary to chemotherapeutic agents.
DMSO’s effect on alcohol dehydrogenase, may make it useful in
the treatment of ethylene glycol poisoning,
but this has not been
sufficiently studied as of yet. DMSO’s attributes as a potential carrier
of therapeutic agents across the skin and into the systemic circulation
and its synergistic effects with other agents are potentially
exciting, but require much more study before they can be routinely
recommended.
While the potential indications for DMSO are many, unfortunately,
the lack of well-controlled studies leaves many more questions
than answers regarding this drug. | | First aid | If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit | | Environmental Fate | Most physiological properties of DMSO appear to be related to
its penetration properties, its potential to inhibit or stimulate
enzymes and to act as a free radical scavenger, and its ability to cause histamine release from mast cells. These properties are
largely based on DMSO’s chemical characteristics, including its
hydrogen bonding behavior, water affinity, ability to interchange
with water in membranes, and ability to react with
organic molecules. | | storage | Dimethyl sulfoxide is reasonably stable to heat,but upon prolonged reflux it decomposes slightly to methyl mercaptan and bismethylthiomethane. This decomposition is aided by acids, and is retarded by many bases. When heated to decomposition, toxic fumes are emitted.
At temperatures between 40 and 60°C, it has been reported that dimethyl sulfoxide suffers a partial breakdown, which is indicated by changes in physical properties such as refractive index, density, and viscosity.
Dimethyl sulfoxide should be stored in airtight, light-resistant containers. The PhEur 6.0 states that glass containers should be used. Contact with plastics should be avoided. | | Shipping | DMSO is not specifically cited in DOT’sPerformance-Oriented Packaging Standards. | | Purification Methods | This colourless, odourless, very hygroscopic liquid, is synthesised from dimethyl sulfide. The main impurity is water, with a trace of dimethyl sulfone. The Karl-Fischer test is applicable. It is dried with Linde types 4A or 13X molecular sieves, by prolonged contact and passage through a column of the material, then distilled under reduced pressure. Other drying agents include CaH2, CaO, BaO and CaSO4. It can also be fractionally crystallised by partial freezing. More extensive purification is achieved by standing overnight with freshly heated and cooled chromatographic grade alumina. It is then refluxed for 4hours over CaO, dried over CaH2, and fractionally distilled at low pressure. For efficiency of desiccants in drying dimethyl sulfoxide see Burfield and Smithers [J Org Chem 43 3966 1978, Sato et al. J Chem Soc, Dalton Trans 1949 1986]. [Reddy Pure Appl Chem 25 459 1969, Beilstein 1 IV 1277.] Rapid purification: Stand over freshly activated alumina, BaO or CaSO4 overnight. Filter and distil it over CaH2 under reduced pressure (~ 12 mm Hg). Store it over 4A molecular sieves. | | Toxicity evaluation | DMSO is naturally released in the environment, primarily by
the oxidation of dimethyl sulfide that is biologically produced
in soil, water, and vegetation. It is produced by phytoplankton,
and may be released during its production, transport, disposal,
and use as a solvent, medicinal analgesic, and other uses.
If released in water, it should disproportionate to dimethyl
sulfide and dimethyl sulfone, and may be reduced by reducing
agents that may occur in natural waters. In soil, DMSO is
rapidly reduced to dimethyl sulfide. In the atmosphere, DMSO
will exist primarily in the vapor phase, and will react with
photochemically produced hydroxyl radicals with a half-life of
w7 h. It also may be released during its production, transport,
disposal, and use as a solvent and medical analgesic.
A bioconcentration factor (BCF) of <1 was observed for
dimethyl sulfoxide, using orange-red killifish (Oryzias latipes)
which were exposed over an 8-week period. According to
a classification scheme, this BCF suggests that bioconcentration
in aquatic organisms is low. | | Incompatibilities | DMSO reacts violently with strong oxidizers, many acyl halides, boron hydrides, and alkali metals. DMSO can form explosive mixtures with metal salts of oxoacids (sodium perchlorate, iron(III) nitrate). | | Waste Disposal | Excess dimethyl sulfoxide and waste material containing this substance should be placed in an appropriate container, clearly labeled, and handled according to your institution's waste disposal guidelines. | | Regulatory Status | Included in the FDA Inactive Ingredients Database(IVinfusions,SC implants, and topical preparations). Available in the USA as a 50% solution for irrigation in the treatment of interstitial cystitis. Also available in Canada as a 70% solution for use as a topical antifibrotic, and in Germany as a topical gel containing 10% dimethyl sulfoxide for the treatment of musculoskeletal and joint disorders. Included in topical formulations of idoxuridine and diclofenac licensed in the UK. |
| | Dimethyl sulfoxide Preparation Products And Raw materials |
|